Hydroxyeicosatetraenoic Acids and Related
Mono-Oxygenated Oxylipins
The oxygenated metabolites or oxylipins derived from arachidonic and related polyunsaturated fatty acids are synthesised through a series of complex, interrelated biosynthetic pathways that is often termed the 'eicosanoid cascade'. Here, the linear hydroxyeicosatetraenes and related mono-oxygenated metabolites are described, together with octadecanoids derived from linoleate and comparable oxylipins from eicosapentaenoic (EPA) and docosahexaenoic (DHA) acids. While these are relatively simple in structure, they are precursors for families of more complex molecules such as the leukotrienes and lipoxins and the protectins, resolvins and maresins (or 'specialized pro-resolving mediators'). The two main enzymatic pathways for these eicosanoids utilize lipoxygenases (LOXs) or oxidases of the cytochrome P-450 family, although some occur as minor by-products of cyclooxygenases or when cyclooxygenase 2 is inhibited by aspirin, as discussed in our web page on prostanoids, which have ring structures in the centre of the molecule and are discussed on their own web page. Hydroperoxides can also be formed non-enzymatically (autoxidation) as discussed in our web page dealing with isoprostanes.
1. Lipoxygenases and Hydroxyeicosatetraenoic Acids
Lipoxygenases are a family of enzymes that are characterized as non-heme iron proteins or dioxygenases, which catalyse the abstraction of hydrogen atoms from bis-allylic positions (1Z,4Z-pentadiene groups) of polyunsaturated fatty acids followed by stereospecific addition of dioxygen to generate hydroperoxides. They occur widely in plants, fungi, a few prokaryotes (cyanobacteria and proteobacteria) and animals, but not in the archaea and most insects. The plant lipoxygenases have distinctive substrates and products, and they are described in our web page dealing with plant oxylipins rather than here, although interesting parallels can be drawn with the mechanisms and functions of the animal enzymes.
 Animal
lipoxygenases that utilize arachidonate as substrate are of great biological and medical relevance
because their metabolites serve in signalling or in inducing structural or metabolic changes in the cell.
They react primarily with unesterified arachidonic acid to yield hydroperoxides in characteristic positions and thence by downstream processing
the plethora of eicosanoids, each with its own functions, which are described in this and other web pages and include lipoxins, trioxilins,
leukotrienes, hepoxilins, eoxins and specialized pro-resolving mediators.
Of these, only the primary lipoxygenase oxylipins are discussed here.
Some lipoxygenase can react to a limited extent directly with phospholipids in membranes to give hydroperoxides and further metabolites that
perturb the membranes to induce structural changes in the cell as in the maturation of red blood cells.
Beyond their role in oxylipin synthesis, lipoxygenases and lipid hydroperoxides act more generally in cellular redox homeostasis
and can stimulate the formation of unwelcome secondary metabolites, which, for example, can attack low-density lipoproteins directly with major
implications for the onset of atherosclerosis.
Animal
lipoxygenases that utilize arachidonate as substrate are of great biological and medical relevance
because their metabolites serve in signalling or in inducing structural or metabolic changes in the cell.
They react primarily with unesterified arachidonic acid to yield hydroperoxides in characteristic positions and thence by downstream processing
the plethora of eicosanoids, each with its own functions, which are described in this and other web pages and include lipoxins, trioxilins,
leukotrienes, hepoxilins, eoxins and specialized pro-resolving mediators.
Of these, only the primary lipoxygenase oxylipins are discussed here.
Some lipoxygenase can react to a limited extent directly with phospholipids in membranes to give hydroperoxides and further metabolites that
perturb the membranes to induce structural changes in the cell as in the maturation of red blood cells.
Beyond their role in oxylipin synthesis, lipoxygenases and lipid hydroperoxides act more generally in cellular redox homeostasis
and can stimulate the formation of unwelcome secondary metabolites, which, for example, can attack low-density lipoproteins directly with major
implications for the onset of atherosclerosis.
General mechanisms: The nomenclature of animal lipoxygenases is based on the specificity of the enzymes with respect to the positions of the resulting hydroperoxides from the reaction with arachidonate (not the initial point of hydrogen abstraction); for example, 12-LOX oxygenates arachidonic acid at carbon-12, and the stereochemistry of the reaction can be specified, e.g., 12R-LOX or 12S-LOX, although most hydroperoxides from enzyme reactions have the S‑configuration. Where more than one enzyme has the same specificity, it may be named after the tissue in which it is found, and there are platelet, leukocyte and epidermal types of 12‑LOX.
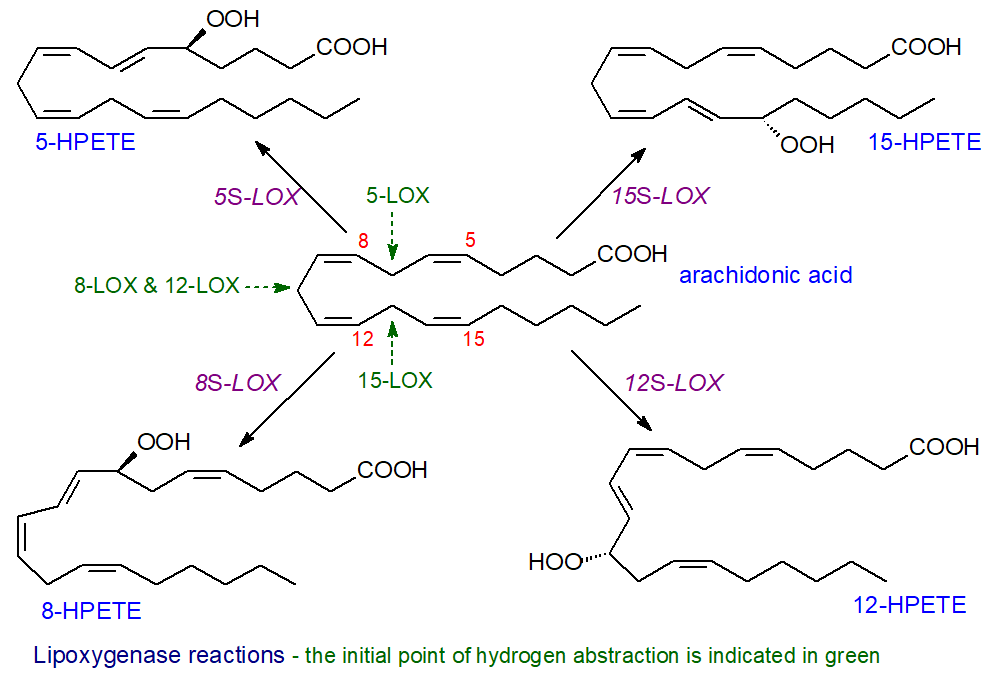 |
| Figure 1. Reactions of lipoxygenases with arachidonic acid. |
As the research in this area has developed, this simplistic nomenclature has become confusing as some enzymes can oxygenate more than one position, which can vary with the chain-length of the polyunsaturated substrate and the positions of the double bonds. Enzymes that act upon four different positions in arachidonic acid occur in animal tissues, i.e., 5‑LOX, 8-LOX, 12-LOX and 15‑LOX, although some of these have dual specificities, while many isoforms exist. In humans, there are now considered to be six main lipoxygenase family members (5‑LOX, 12‑LOX, 12/15‑LOX (15‑LOX type 1), 15‑LOX type 2, 12R‑LOX and epidermal LOX (eLOX-3)) with seven in mice. Orthologues of the same gene can have different positional selectivities in different species, and mice do not express a distinct 15‑LOX but rather a leukocyte-derived 12-LOX with some 15‑LOX activity, so it can be difficult to extrapolate from animal experiments to human conditions; the human enzymes only are discussed at length here.
Each of the lipoxygenase proteins in animal tissues has a single polypeptide chain with a molecular mass of 75-80 kDa. They have an N‑terminal 'β‑barrel' or 'PLAT' domain, which acquires the substrate, and a larger α-helical catalytic domain containing a single atom of non-heme iron, which is bound to four conserved histidine residues and to the carboxyl group of a conserved isoleucine at the C‑terminus of the protein. The PLAT domain anchors the otherwise cytosolic protein to membranes in response to intracellular calcium levels. For catalysis to occur, the iron component of the enzymes must be oxidized to the ferric state.
The enzymes enclose the fatty acid substrate within a tight channel with smaller channels that direct molecular oxygen toward the selected carbon to facilitate the formation of hydroperoxy-eicosatetraenes (HPETEs). In other words, the regiospecificity is regulated by the orientation and depth of substrate entry into the catalytic site, while stereospecificity is controlled by switching the position of oxygenation on the reacting pentadiene of the substrate at a single enzyme site, which is conserved as an alanine residue in S‑lipoxygenases and a glycine residue in the rarer R‑lipoxygenases. There is evidence that two amino acids opposite the catalytic iron ion determine the orientation of the substrate for entry into the enzyme channel. With 5‑LOX and 8‑LOX, the carboxyl group of arachidonic acid enters the catalytic site first, while with 12-LOX and 15-LOX, the ω‑terminus enters the site and assists the reaction. The N-terminal domains serve in membrane binding and regulation and are not utilized for catalysis.
Lipoxygenase action proceeds in four steps - hydrogen abstraction (1), radical rearrangement (2), oxygen insertion (3) and peroxy radical reduction (4), all occurring under strict steric control, as illustrated.
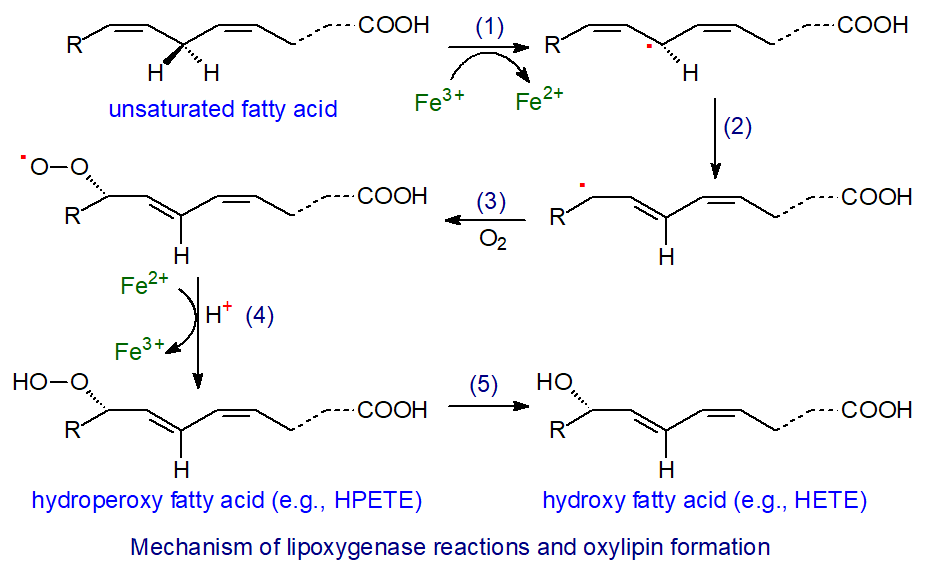 |
| Figure 2. Mechanism of lipoxygenase action and oxylipin biosynthesis. |
For example, in the action of 5-LOX, the first and rate-limiting step is the abstraction of a hydrogen atom from carbon 7 of arachidonic acid by non-heme ferric iron (Fe(III)), involving a proton-coupled electron transfer in which the electron is transferred directly to the iron and the proton is acquired simultaneously by the hydroxide ligand in a concerted mechanism to produce a substrate radical, while the iron atom is reduced to the ferrous state (Fe(II)). The cis-double bond in position 5 migrates to position 6 to form a more stable conjugated diene with a change to the trans-configuration before dioxygen is introduced opposite to the removed hydrogen (antarafacially) to generate a lipid peroxyl radical. Finally, the lipid peroxyl radical is reduced by Fe(II) and protonated to create a lipid hydroperoxide in another concerted reaction. In the process, the iron atom is re-oxidized to its ferric state for another round of catalysis. The oxylipin from the reaction illustrated is 5S‑hydroperoxy-6E,8Z,11Z,14Z-eicosatetraenoic acid (5‑HPETE).
HPETE in general have a short half-life and are rapidly metabolized to hydroxy-eicosatetraenes (HETE) with the same stereochemistry, often via reduction by the abundant and ubiquitous glutathione peroxidases (step 5). While they are primarily intermediates in the biosynthesis of other eicosanoids, HPETE can have some direct influence on tissue metabolism.
Alternatively, isomerization reactions of hydroperoxides can occur to synthesise leukotrienes and lipoxins via epoxy intermediates. Simplistically, the Fe2+ in the lipoxygenase cleaves the O-O bond in the hydroperoxide with transfer of the hydroxyl group to form Fe3+‑OH, before the residual alkoxyl radical is cyclized to an epoxy fatty acid.
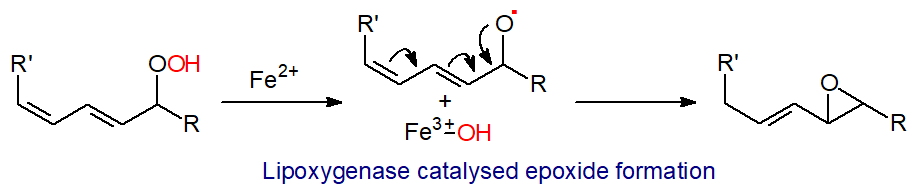 |
| Figure 3. Mechanism of lipoxygenase catalysed epoxide synthesis. |
Enzyme specificities: 5-LOX (ALOX5) is found only in cells derived from bone marrow (leukocytes, macrophages, etc.), and the products are the primary precursors for the leukotrienes and lipoxins and for resolvins. It is a cytosolic protein when intracellular calcium levels are low, but it becomes associated with the nuclear membrane when they are high or after phosphorylation. In contrast to other lipoxygenases, it requires the presence of a particular activator protein on the perinuclear membrane, five-lipoxygenase-activating protein (FLAP), which facilitates the transfer of arachidonic acid to the catalytic site on 5-LOX and may accomplish the functional coupling of phospholipase A2 (cPLA2) to 5-LOX at the membrane. It is noteworthy that both cPLA2 and 5‑LOX are Ca2+‑dependent. 5‑LOX and related enzymes are regulated by several factors that include the concentration and availability of the substrates, the redox state, intracellular Ca2+ concentrations, and phosphorylation-dephosphorylation by means of various protein kinases.
 8-, 12-
and 15-LOX operate in the same way to give analogous oxylipins and associate with membranes in a calcium-dependent process,
although they do not require accessory proteins.
15‑LOX exists in two isoforms, usually with one having a broader selectivity that depends on the animal
source, hence the superfluity of names (12S-LOX of mice, leukocyte-type 12S-LOX of rabbits, reticulocyte-type 15S-LOX of
rabbits, and human reticulocyte-type 15S-LOX), and it is expressed primarily in reticulocytes and macrophages on stimulation by
interleukins 4 and 13.
8-, 12-
and 15-LOX operate in the same way to give analogous oxylipins and associate with membranes in a calcium-dependent process,
although they do not require accessory proteins.
15‑LOX exists in two isoforms, usually with one having a broader selectivity that depends on the animal
source, hence the superfluity of names (12S-LOX of mice, leukocyte-type 12S-LOX of rabbits, reticulocyte-type 15S-LOX of
rabbits, and human reticulocyte-type 15S-LOX), and it is expressed primarily in reticulocytes and macrophages on stimulation by
interleukins 4 and 13.
15-LOX in humans makes 15S-HPETE mainly and is better termed 15‑LOX‑1 (alternatively, ALOX15 or 12/15‑LOX), but it can make some 12‑HETE, 8,15‑diHETE and eoxin A4 from arachidonic acid. It can oxidize linoleate to 13‑hydroperoxy-octadecadienoate (and in part to the 9-isomer), as well as reacting with α‑linolenic, γ‑linolenic, eicosapentaenoic and docosahexaenoic (DHA) acids. 15S‑HPETE is induced by the action of cytokines and is the precursor of the pro-resolution lipoxins, while with DHA, 15-LOX yields 17S‑HPDHA, a precursor of resolvins and protectins. Uniquely, 15‑LOX‑1 synthesises both pro- and anti-inflammatory molecules, and molecular genetics studies show that this feature is seen only in higher ranked primates and not in mammals ranked in evolution lower than gibbons, where the enzyme only the 12‑isomer results from arachidonate.
The second human arachidonate 15‑lipoxygenase has 40% homology with the first and is termed 15‑LOX‑2 (or ALOX15B). It produces 15-HETE exclusively from arachidonate and is expressed constitutively in macrophages, where it has been associated with cellular cholesterol homeostasis and is induced by hypoxia, and in the prostate gland, lung, skin and cornea. Like 15-LOX-1, it differs from the other lipoxygenases in that it can utilize most polyunsaturated fatty acids as substrates, both in unesterified state and bound to intact lipids, including phospholipids and cholesterol esters in membranes and lipoproteins. Hence the interest in the enzyme in autophagy, membrane disruption and disease states (asthma, psoriasis and atherosclerosis). Mouse skin has a lipoxygenase (8‑LOX) that is structurally related to 15-LOX-2, but generates 8S-HETE and 8S,15S-diHPETE from arachidonic acid. Some 15R-HETE comes from the action of COX-2 and aspirin.
12S-LOX (ALOX12) from human platelets and leukocytes was one of the first lipoxygenases to be characterized and takes part in the first step of maresin synthesis. A rather different enzyme is present in mammalian skin that unlike other lipoxygenases can produce the R‑enantiomer of its metabolites and not the usual S‑configuration. Indeed, 12R‑HETE was first characterized as a component of psoriatic lesions. One of the enzymes responsible is the second isoform of the human 15‑lipoxygenase (15-LOX-2), but there is also a 12R-LOX (ALOX12B) with distinctive functions in keratinocytes and certain other tissues in relation to linoleate metabolism and the formation of structural ceramides in the corneocyte envelope. As part of the process, eLOX3 in skin is a hydroperoxide isomerase (lipohydroperoxidase) that converts hydroperoxides to epoxy-alcohols and ketones (hepoxilin-like metabolites); enzymes related to this are common in aquatic invertebrates.
Hydroxy fatty acids from lipoxygenase reactions can be further oxidized to their keto analogues (cf., 5-oxo-eicosatetraenoic acid below) or to dihydroxy derivatives that include the leukotrienes discussed in a separate web page; some are conjugate with glutathione.
2. Cytochrome P450 Oxidases and Hydroxy/Epoxy-Eicosatetraenoic Acids
Arachidonic acid can be oxidized by several cytochrome P450 mixed-function oxidases to various HETE isomers (the name was coined to describe the first such enzyme to be characterized and was based on an unusual absorbance peak at 450 nm from its carbon monoxide-bound state). These enzymes are a superfamily of membrane-bound hemoproteins that catalyse the scission of the dioxygen bond in molecular oxygen and transfer a single atomic oxygen to a substrate carbon atom, i.e., they are monooxygenases, with the release of the other oxygen atom as water. The result is the introduction of either a hydroxyl or an epoxyl group into the molecule. The reaction is NADPH-dependent, requiring transfer of electrons from NADPH to the P450 heme iron (lipoxygenases use non-heme iron) for which a membrane-bound enzyme partner, NADPH-cytochrome P450 reductase, is essential in the endoplasmic reticulum (or functionally related enzymes in mitochondria).
 |
| Figure 4. Stoichiometry of CYP450 oxidase reaction. |
Cytochrome P450 oxidases are found in all mammalian cell types and indeed are to be ubiquitous in living organisms, although the number and distribution of each are characteristic both for cell type and species. They are located in the endoplasmic reticulum with a limited expression in mitochondria (and perhaps plasma membrane and nucleus) and predominantly in the liver but with significant levels in some extrahepatic tissues, including brain, kidney and lung. As well as generating HETE isomers, enzymes of this kind are part of the eicosanoid cascade in the metabolism of prostanoids, and they are utilized in cholesterol, steroid and vitamin metabolism as well as detoxification of lipophilic xenobiotics, including drugs and chemical carcinogens. Their nomenclature starts with the root 'CYP', followed by a number allocated to the family, a letter for subfamily and a gene-identifying number for isoforms.
CYP450s have two key domains: a β-sheet-rich N-terminal domain and a larger helix-rich C-terminal catalytic domain. Those enzymes in the endoplasmic reticulum have a transmembrane helix in the N-terminal domain that is required for membrane anchoring, but this feature is not present in mitochondrial CYPs, which rely upon hydrophobic regions on the surface to bind to membranes. The catalytic domain contains the heme prosthetic group in a deep cavity, and variability in the structure of this site in each explains the flexibility for substrates and products. Access channels permit entry of substrates, and exit channels allow egress of the final metabolite. Three types of reaction have been observed in animal cells that lead to three families of eicosanoids, all requiring unesterified arachidonic acid as substrate, although appreciable amounts of the metabolites are eventually esterified.
Mid-chain HETE: Synthesis of mid-chain HETEs is accomplished by CYP1B1, CYP4A or CYP2B enzymes in reactions at bis‑allylic centres and is lipoxygenase-like in the nature of the ultimate HETEs, although not via hydroperoxy intermediates. Thus, these microsomal cytochrome P450 oxidases can react with arachidonic acid to give six regioisomeric cis,trans-conjugated dienols, i.e., with the hydroxyl group in positions 5, 8, 9, 11, 12 or 15. The mechanism probably involves bis-allylic oxidations at either carbon-7, 10 or 13, followed by acid-catalysed rearrangement to the cis,trans-dienol (two of the possible isomers are illustrated). 12R-HETE as opposed to the 12S-isomer is a major product of the reaction, and this was at one time though to be a distinguishing feature, but some other lipoxygenases are now known to synthesise the first of these.
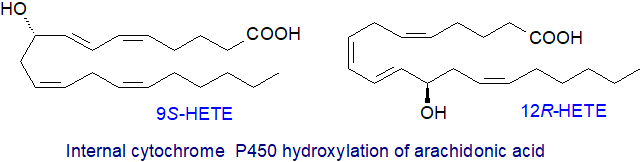 |
| Figure 5. Two metabolites from internal cytochrome P450 hydroxylation of arachidonic acid. |
Some CYP450 enzymes can introduce hydroxyl groups into positions 2 and 3 of fatty acids, while others can catalyse decarboxylation or form terminal alkenes and have biotechnological potential.
Omega-hydroxylated HETE: Secondly, there are ω- and (ω-1)-hydroxylases that introduce a hydroxyl group into positions 20 and 19, respectively, of arachidonic acid mainly, with no change in double bond position or geometry, although other enzymes are known that can react at positions 16, 17 and 18. The reaction is not confined to polyunsaturated fatty acids and was first observed with medium-chain saturated fatty acids, such as lauric (12:0), where it may play a part in oxidative catabolism. Some isoenzymes act only upon laurate, others with arachidonate, and some will utilize both fatty acids as substrates, but they have a high similarity in amino acid sequences although they have different physiological functions. In humans, the isoforms CYP4F2 and CYP4F3B are the main enzymes for ω‑hydroxylation of polyunsaturated fatty acids, including both arachidonic and eicosapentaenoic acids, while CYP4F3B, CYP4F11 and CYP4F22 perform (ω‑1)-hydroxylations. CYP4F3A inactivates leukotriene B4 as anti-inflammatory response, while CYP4F22 contributes to a healthy skin barrier. Both R- and S-enantiomers of the sub-terminal HETE with differing activities can occur.
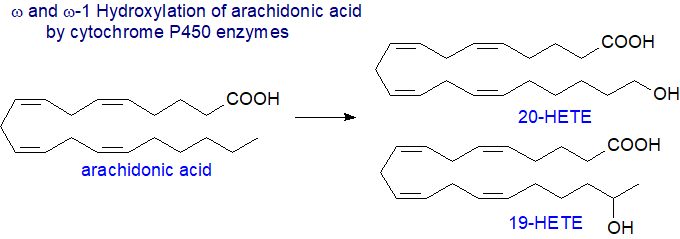 |
| Figure 6. Isomers from ω- and ω-1 hydroxylation by cytochrome P450 enzymes. |
20-HETE is metabolized by cyclooxygenases into a hydroxy analogue of prostaglandin H2 (20-OH PGH2), a vasoconstrictor that is further converted by isomerases into 20-OH PGE2 and 20-OH PGI2 (vasodilator/diuretic metabolites) or to 20‑OH thromboxane A2 and 20-OH PGF2α (vasoconstrictor-antidiuretic metabolites).
Epoxyeicosatrienoic acids: In the third series of reactions of CYP450 monooxygenases, epoxytrienoic acids (‘EET’) are synthesised from arachidonic acid, i.e., four cis-epoxyeicosatrienoic acids (14,15-, 11,12-, 8,9- and 5,6-EETs). Other than the 5,6-isomer, they are relatively stable molecules.
Several iso-enzymes of the cytochrome P450 epoxygenase exist, with CYP2C and CYP2J as the most active, and they can produce all four EET regioisomers, although one isomer tends to predominate in each tissue usually. For example, epoxygenases for 14,15-EET as the main isomer synthesise a significant amount of 11,12‑EET and a little 8,9-EET. The epoxygenase attaches an oxygen atom to one of the carbons of a double bond of arachidonic acid and then to a second to generate an epoxide with one fewer double bond. The enzymes are located both in the cytosol and the endoplasmic reticulum of endothelial cells, and they make use of arachidonic acid that is hydrolysed from phospholipids when the Ca2+-dependent phospholipase A2 is activated and translocated from the cytosol to intracellular membranes.
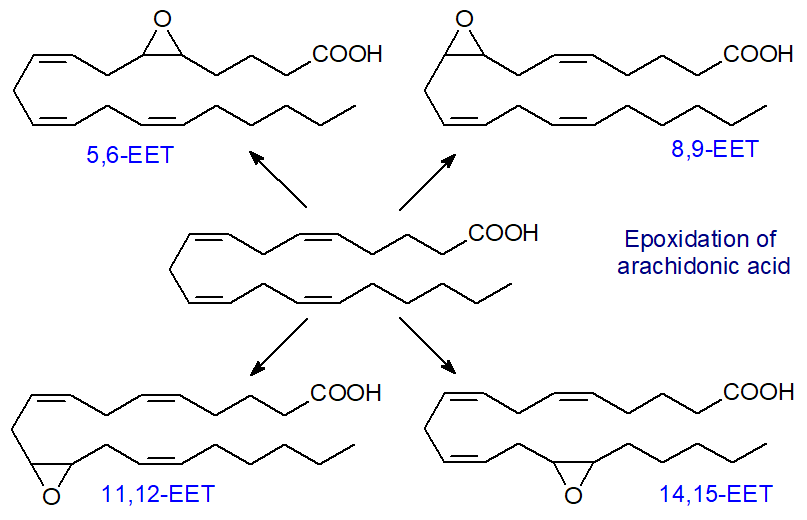 |
| Figure 7. Biosynthesis of epoxy-eicosanoids by CYP450 oxidases. |
The proportions of the various isomers depend on tissue and species, although the 11,12- and 14,15‑EET generally tend to predominate. In the rat, 14,15‑EET amounts to about 40% of those synthesised in the heart, while 11,12-EET represents 60% of those from the kidney. To add to the complication, each of these regioisomers is a mixture of R,S- and S,R-enantiomers, and each iso-enzyme gives variable proportions, differing even among regioisomers. Eight isomers can result, therefore, each having somewhat different responses in tissues. By the same means, adrenic acid (22:4(n-6)) can be converted to epoxy metabolites with 16,17‑epoxy-docosatrienoic acid as the most abundant isomer in liver.
The epoxygenases require the fatty acid substrate to be in the unesterified state, but their metabolites can be esterified later. Thus, significant amounts of epoxyeicosatrienes are found esterified to position sn-2 of phospholipids, including phosphatidylcholine, phosphatidylethanolamine and phosphatidylinositol, perhaps for a storage purpose that is available when a rapid response is required, and after neuronal, hormonal or chemical stimulation, free epoxyeicosatrienes can be released by phospholipase A2 . It is nonetheless possible that esterified epoxy-eicosanoids may have a function of their own within membranes (see below). The presence of esterified EETs in plasma suggests that some exchange between tissues is likely, although most are produced close to the site of action. In many tissues, the esterified epoxy-eicosanoids are almost identical in composition to those in the free state, so the conclusion must be that they result entirely from enzyme action. On the other hand, non-enzymic lipid epoxidation has been observed in erythrocytes in vitro, and some EETs with the epoxide group in both the cis- and trans-configurations may arise by this route.
EETs are rapidly metabolized in vivo to the corresponding dihydroxyeicosatrienoic acids (DHET) by epoxide hydrolases, of which at least five isoforms are known with different cellular locations and preferred substrates. The cytosolic or soluble (EPHX2 or sEH)) and membrane-bound (EPHX1) enzymes are of special importance, both in terms of oxylipin metabolism and for detoxification of xenobiotic epoxides. In humans, EPHX2 is widely expressed throughout the body and is a 62kDa enzyme composed of two domains separated by a short proline-rich linker in which the N‑terminal domain is a phosphatase towards lipid phosphates, while the C‑terminal domain is the epoxide hydrolase. The reaction is illustrated below for the conversion of 14,15-EET to 14,15-DHET, which proceeds with variable stereochemistry and gives both threo and erythro isomers, which may be good markers of oxidative stress.
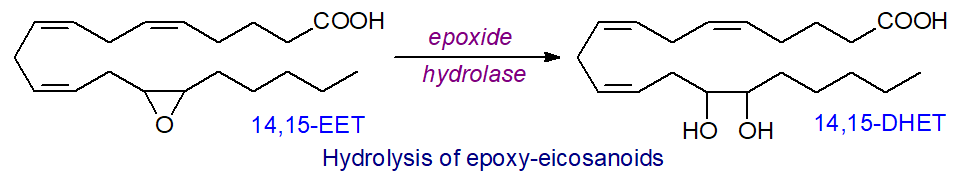 |
| Figure 8. Diol synthesis from epoxy-eicosanoids by epoxide hydrolase. |
This enzyme metabolizes 8,9-, 11,12- and 14,15-EET efficiently, but 5,6-EET is a poor substrate. It displays some enantioselectivity, and this may be a factor in determining the stereochemistry of the circulating epoxides. 11,12- and 14,15-EET can undergo partial β‑oxidation to C16 epoxy-fatty acids, or they can be elongated to C22, while 5,6- and 8,9-EET are substrates for cyclooxygenase. The gut microbiota in the colon has epoxide hydrolase activity distinct from that of the host so may influence host metabolism. While DHETs from EEts were once believed to be inert, they are now known have an influence on tissue metabolism in their own right.
3. Oxo-Eicosatetraenoic Acids
 5-Oxo-6E,8Z,11Z,14Z-eicosatetraenoic acid
(5-oxo-ETE) is a metabolite of 5S‑hydroxy-6E,8Z,11Z,14Z-eicosatetraenoic acid (5‑HETE),
from oxidation by an NADP+-dependent 5‑hydroxy-eicosanoid dehydrogenase,
an enzyme found in the microsomal membranes of white blood cells (leukocytes), platelets, eosinophils and neutrophils (or by
transcellular biosynthesis from inflammatory cell-derived 5S-HETE).
The enzyme requires the presence of a 5S‑hydroxyl group and a
trans-6 double bond in the eicosanoid, and NADP+ is a cofactor.
Synthesis of the metabolite is stimulated during periods of oxidative stress, but some 5‑oxo-ETE may be formed directly from
5‑hydroperoxy-eicosatetraenoic acid, possibly by a non-enzymic route.
In neutrophils, a high proportion is rapidly incorporated into triacylglycerols.
5-Oxo-6E,8Z,11Z,14Z-eicosatetraenoic acid
(5-oxo-ETE) is a metabolite of 5S‑hydroxy-6E,8Z,11Z,14Z-eicosatetraenoic acid (5‑HETE),
from oxidation by an NADP+-dependent 5‑hydroxy-eicosanoid dehydrogenase,
an enzyme found in the microsomal membranes of white blood cells (leukocytes), platelets, eosinophils and neutrophils (or by
transcellular biosynthesis from inflammatory cell-derived 5S-HETE).
The enzyme requires the presence of a 5S‑hydroxyl group and a
trans-6 double bond in the eicosanoid, and NADP+ is a cofactor.
Synthesis of the metabolite is stimulated during periods of oxidative stress, but some 5‑oxo-ETE may be formed directly from
5‑hydroperoxy-eicosatetraenoic acid, possibly by a non-enzymic route.
In neutrophils, a high proportion is rapidly incorporated into triacylglycerols.
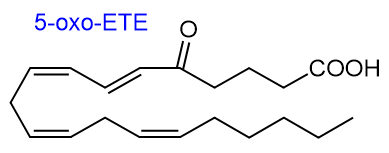
5-Hydroxyeicosanoid dehydrogenase can catalyse the reverse reaction, i.e., the reduction of 5-oxo-ETE, and this seems to be of special relevance in platelets to change its activity. Alternatively, a change can occur by reduction of the double bond in position 6, or by further oxidation either by lipoxygenases or by cytochrome P450 enzymes, the latter in positions 19 or 20.
All the HETE isomers can be converted to oxo-metabolites by hydroxy-eicosanoid dehydrogenases, and the 11-, 12-, 14- and 15‑isomers possess appreciable activity in tissues. 15S‑HETE and 11R-HETE are substrates for 15‑hydroxyprostaglandin dehydrogenase, the enzyme catalysing the first step of prostaglandin catabolism, to yield 15-oxo-ETE and 11‑oxo‑ETE, respectively, which mediate anti-proliferative properties in endothelial cells. As CoA esters, they can undergo up to four double bond reductions. It is now recognized that α,β‑unsaturated keto-eicosanoids generated in this way are electrophilic and have the potential to interact with nucleophilic centres in proteins and other molecules to modify their properties.
4. Mono-oxygenated Metabolites of EPA and DHA
Lipoxygenases and cytochrome P450 oxidases interact with the other essential polyunsaturated fatty acids of the omega-3 and omega-6 families, especially the former, to give comparable series of metabolites. Lipoxygenases have much the same positional specificities with eicosapentaenoic acid (EPA or 20:5(n-3)) as with arachidonic acid to yield hydroxy-eicosapentaenoic acids (HEPE), such as 5- and 12‑HEPE, but 18‑HEPE is derived from aspirin-acetylated COX-2 or by CYP2C8/CYP2J2. 5-Lipoxygenase generates 4- and 7‑hydroxy metabolites from docosahexaenoic acid (22:6(n‑3) or DHA), while 12-lipoxygenase generates 11- and 14-hydroxy metabolites, and 15‑lipoxygenase (15-LOX-2) introduces a 17‑hydroxyl group. These can react further to generate the protectins, resolvins and maresins or 'specialized pro-resolving mediators', which have special importance in the resolution of inflammation and have their own web page.
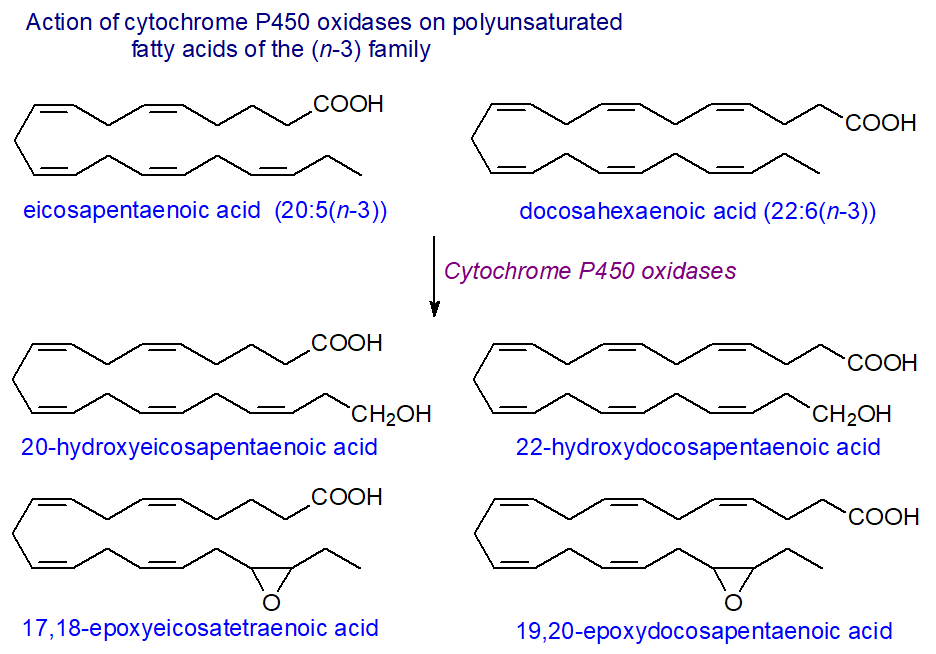
The lipoxygenase metabolites from arachidonate were soon documented, but it has taken longer to recognize the occurrence and properties of those from eicosapentaenoic (EPA) and docosahexaenoic (DHA) acids, e.g., the epoxides derived from various cytochrome P450 oxidases. Indeed, it is now evident that these n-3 polyunsaturated fatty acids, rather than arachidonic acid, are the preferred substrates for some of the enzyme isoforms, namely the CYP1A, CYP2C, CYP2J and CYP2E subfamily members, which then exhibit very different regio- and stereo-specificities. Human CYP1A1 acts mainly as a subterminal hydroxylase with arachidonate to give four different isomers, but with EPA, it generates mainly 17R,18S-epoxy-eicosatetraenoate with almost absolute regio- and stereo-selectivity, and with DHA, it epoxidizes the n-3 double bond and produces 19,20‑epoxy-docosapentaenoate. Other isoforms of the cytochrome P450 enzymes react with an n‑3 double bond in the same manner, some much more rapidly than with arachidonate as substrate, but CYP2C9 is an exception and oxidizes EPA to 14,15-epoxy-ETE mainly and DHA to 10,11‑epoxy-DPE.
 |
| Figure 9. Action of cytochrome P450 oxidases on EPA and DHA. |
The CYP4A/CYP4F subfamilies generate 20-hydroxy-eicosatetraenoic acid from arachidonate in mammals, and they hydroxylate the terminal methyl group in EPA and DHA at the same rate. In addition, human endothelial cells with upregulated COX-2 and treated with aspirin convert EPA to 18R‑hydroxyeicosapentaenoic acid with anti-inflammatory properties. 4‑Oxo‑DHA is present in plasma of rats fed DHA and is a potent anti-tumour agent (antiproliferative) against breast cancer, possibly because it can undergo the Michael reaction; it is also a PPARγ agonist.
By competing with arachidonate, EPA and DHA may modify the action of the various HETE metabolites, but the oxygenated EPA and DHA compounds have biological properties of their own. For example, 17,18-epoxyeicosatetraenoic acid generated in the gut is an anti-allergy molecule, while significant amounts of DHA epoxides, mainly 7,8‑epoxydocosapentaenoic acid, are present in the central nervous system of rats, where they ameliorate inflammatory pain. It has been suggested that such EPA and DHA metabolites may be responsible for some of the benefits associated with dietary n‑3 fatty acid intake.
5. Octadecanoids
Linoleate (9,12-18:2) hydroperoxides are produced in animal tissues by all the enzymes used in eicosanoid generation, including lipoxygenases, cyclooxygenases (COX-2) and cytochrome P450 enzymes, with first hydroperoxy-octadecanoids (HPODEs) and then hydroxy-octadecanoids (HODEs), although their primary source in plasma is not known. They can be catabolized by the same enzyme as with eicosanoids, i.e., 15-hydroxy-prostaglandin dehydrogenase, to keto derivatives. While similar reactions occur with both α- and γ‑linolenic acids in vitro and their metabolites have been detected in plasma and some other tissues at low levels, relatively little appears to be known of their significance in vivo. Autoxidation of linoleate gives similar molecules but with more variable stereochemistry.
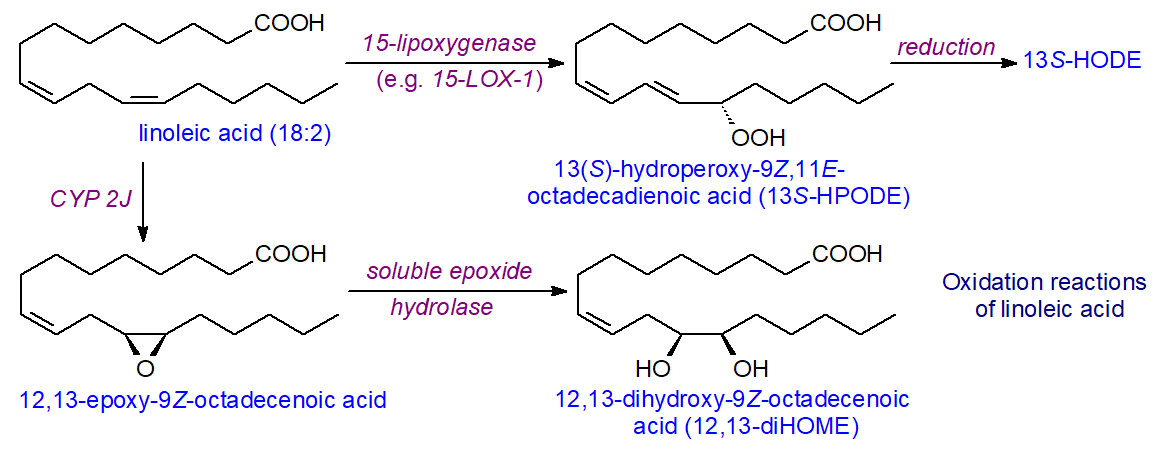
The action of lipoxygenases upon linoleic acid in plant tissues is discussed in the web page on plant oxylipins, but this fatty acid is acted upon by lipoxygenases in animal tissues in the same way to synthesise 9- and 13-hydroperoxy- and thence hydroxy-octadecadienoic acids of defined stereochemistry. 13S‑Hydroperoxy-9Z,11E-octadecadienoic acid (13S‑HPODE) is generated by the action of 15‑lipoxygenase (15‑LOX‑1) on linoleic acid, and this is reduced to the hydroxy compound, while oxo-, epoxy- and epoxy-keto-octadecenoic acids result from further reactions, as illustrated. 12R-LOX reacts readily with linoleate to produce 9R‑HPODE.
 |
| Figure 10. Some reactions of oxidases with linoleic acid. |
Enzymes of the cytochrome P450 family make a further contribution, and linoleic acid is a substrate for CYP epoxygenases, e.g., CYP2C9 in human liver, to yield the linoleic epoxides 9,10- and 12,13‑epoxyoctadecenoic acids (9(10)-EpOME and 12(13)-EpOME, respectively), which are sometimes termed leukotoxins (although this name has been applied also to very different microbial metabolites). Epoxide hydrolase can then metabolize them to the 9,10- and 12,13‑diols, 9,10-DiHOME and 12,13-DiHOME, respectively.
Linoleate metabolites were first found in patients with burns and inflammatory diseases, adult respiratory distress syndrome and chronic obstructive pulmonary disease (COPD), and the diols can cause mitochondrial-mediated cell death, although they can be detoxified by conversion to the glucuronides. On the other hand, 12,13-dihydroxy-9Z-octadecenoate (12,13-diHOME) synthesised in adipose tissue has beneficial properties (see below). As linoleic acid is a major unsaturated fatty acid in animal tissues, appreciable amounts of these hydroxy and hydroperoxy metabolites can accumulate and influence inflammatory diseases. Indeed, linoleate metabolites are by far the most abundant oxygenated fatty acids in both free and esterified state in human plasma and in the brain of rat pups. Other octadecanoids result from bacteria in the gut.
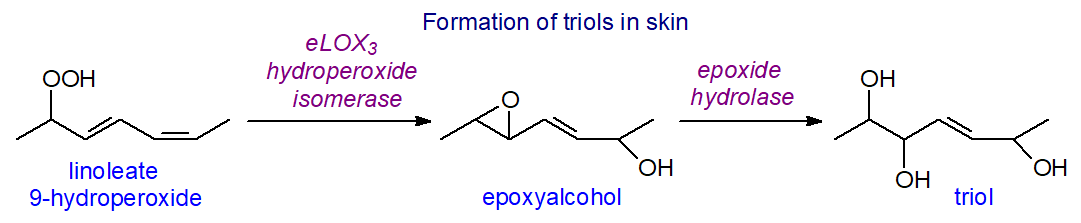
Sebaleic acid (5,8,-181:2) in sebum is oxidized by 5-LOX in neutrophils to 5-oxo-ODE, a potent chemoattractant that may be involved in inflammatory skin disease, and to 5-hydroxy-(6E,8Z)-octadecadienoic acid (5-HODE) and other metabolites. A further interesting observation is that one of the unique ceramides of skin, O‑linoleoyl-ω-hydroxyacyl-sphingosine, is a substrate for 12R‑LOX with 9R‑hydroperoxy-linoleoyl-ω-hydroxyceramide as the product. This in turn can be converted to hepoxilin-like compounds, i.e., with an epoxyl group, by an enzyme epidermal lipoxygenase 3 (eLOX-3), while trihydroxy compounds, e.g., octadec-9R,10S,13R-trihydroxy-11E-enoate (tri-HOMEs), may be formed subsequently by the action of an epoxide hydrolase, such as the human soluble enzyme. 9R,10S,13R-Trihydroxy-11E-octadecenoate is an important oxylipin in porcine and human epidermis, where it interacts with the ceramides to assemble the waterproof barrier, while in the lung, tri-HOMEs are synthesised by a mechanism that starts with a 13S‑hydroperoxide from the action of 15-lipoxygenase and proceeds via an epoxide intermediate.
 |
| Figure 11. Biosynthesis of octadecanoid triols and other oxylipins in skin from linoleate. |
6. Esterified Oxylipins
Most eicosanoid-generating enzymes require free fatty acids as the substrate, and they are unable to oxidize intact phospholipids, although 15‑LOX in human monocytes is an exception, while in platelets, 12-HETE-lysophosphatidylethanolamine is produced by direct action of 12-LOX. However, arachidonoyl-carnitine and arachidonoyl-CoA can as substrates for mammalian ALOX isoforms and are oxygenated with high positional specificity, although the latter reacts more slowly than the free acids. Further, free 5-, 12- and 15-HETEs can be esterified to phospholipids in tissues, often with some selectivity, as can hydroxydocosapenta- and hexaenoic acids, but octadecanoids are preferentially esterified to neutral lipids. All mammalian long-chain acyl-CoA synthetase isoforms have the capacity to activate HETE for further esterification through the action of membrane-bound O‑acyltransferases (thromboxanes may be an exception). The mechanisms for these esterification processes are discussed in our web page on oxidized phospholipids. It is evident that many of these reactions depend on particular cell types and lipids, and that cell compartmentalization is a significant factor as eicosanoids of exogenous origin and those generated endogenously are sensed differently. In contrast, non-enzymatic oxidation (autoxidation) of polyunsaturated fatty acids occurs when they are esterified by the initial steps described in our web page on isoprostanes.
15-HETE is selectively esterified to phosphatidylinositol in lung and kidney epithelial cells and in aortic endothelial cells, while 12-HETE occurs predominantly in phosphatidylcholine in microsomal membranes. In neutrophils, 5-HETE is incorporated mainly into phosphatidylethanolamine plasmalogens and phosphatidylcholine, while three phosphoinositides linked to 12‑hydroxyeicosatetraenoic acid have been detected in thrombin-activated platelets. More than 90% of the EETs in most plasma and organ tissues are esterified to position sn‑2 of glycerophospholipids, and in aortic endothelial cells, 20‑HETE is present in an esterified state in several phospholipid classes.
Many of these esterified lipoxygenase and oxidase metabolites of phospholipids remain within the membranes where they may serve for storage to be released on appropriate stimulation, possibly into other cellular compartments with different roles from their unesterified equivalents. On the other hand, such oxidized lipids have the potential to perturb membrane structures and carry out secondary oxygenations that could induce unwanted changes in cells, such as by an involvement in ferroptosis. Oxidation of low-density lipoprotein by this means has been implicated in the initiation of atherosclerosis, and there are suggestions that cholesteryl arachidonate in the lipoprotein LDL is a good substrate for 15‑LOX‑1 (and for autoxidation), and that the products are a causative factor in this disease (see our web page on cholesterol).
Phospholipids containing EET are substrates for the production of lipid mediators such as 2‑epoxyeicosatrienoyl-sn-glycerols, comparable to the endocannabinoid 2‑arachidonoylglycerol and to the anandamide analogue (and discussed further under this topic). Kidney and spleen, for example, synthesise sn-2-glycerol derivatives esterified with 11,12-EET or 14,15-EET, which are endocannabinoids and exert effects by their interaction with the CB1 and CB2 receptors, while phospholipids containing EET are probable substrates for synthesis of EET-ethanolamide in the liver and kidney. Endocannabinoids such as anandamide and synaptamide can be converted directly to various oxygenated derivatives, which can sometimes initiate greater responses than their precursors.
7. Tissue Responses to HETE
Numerous hydroxyeicosatetraenoic acids and related compounds have now been discovered and most of these have some biological activity, in vitro at least, and primarily in signalling. They modulate ion transport, vascular tone, renal and pulmonary functions, and growth and inflammatory responses through both receptor and non-receptor mechanisms. Their release is stimulated by the action of growth factors and cytokines, and they attain physiological concentrations in tissues that are much higher than those of prostanoids. This is a field that is still developing rapidly, and it is evident that the picture is complex and far from complete. A given eicosanoid of this kind can have differing functions in different cell types, and as this may be opposed or modified by another eicosanoid, the balance between them in a cell may be critical. As animal models can have very different isoforms of enzymes, it is often difficult to translate experiments with other species to human conditions. It is not possible to give a comprehensive picture of this complexity here, as this would require a substantial tome, so only a few of best known and studied are described briefly below.
HETEs: 5S-Hydroxy-6E,8Z,11Z,14Z-eicosatetraenoic acid (5S-HETE) is the precursor of the leukotrienes and lipoxins, but it has some functions in its own right, although these can be difficult to disentangle from those of its metabolites. Like its metabolite 5-oxo-HETE, 5S‑HETE activates neutrophils and monocytes, and it is known to stimulate proliferation of cancer cells in a similar manner to certain leukotrienes with increased amounts present in brain tumours; 5‑LOX inhibitors are preventive.
5-Oxo-6E,8Z,11Z,14Z-eicosatetraenoic acid is a chemo-attractant for eosinophils and neutrophils, and it also takes part in actin polymerization, calcium mobilization, integrin expression and degranulation. Its signalling functions are mediated via a Gi/o-coupled receptor ('OXE'), leading to increased intracellular calcium concentrations and inhibition of cAMP production. By increasing the number of dermal fibroblasts, it promotes wound healing, but this is inhibited by the action of ceramide 1-phosphate on its receptor (OXER1 in mice). In contrast, it stimulates the proliferation of prostate tumour cells, and it is a mediator in asthma and other allergic diseases. Efforts are underway to find inhibitors of the OXE receptor of potential clinical value.
 Arachidonate
8S-lipoxygenase and the derived 8S-hydroxy-5Z,9E,11Z14Z-eicosatetraenoic
acid (8S-HETE) have only been found in the skin of mice.
It is a potent activator of the peroxisome proliferator-activated receptor PPARα, it is an anti-tumorigenic agent towards skin cancer,
and it promotes wound healing in the cornea.
In contrast, a human orthologue of this enzyme (15‑LOX‑2) is found in skin, sebaceous glands and prostate tissue but produces
15S-HETE.
Arachidonate
8S-lipoxygenase and the derived 8S-hydroxy-5Z,9E,11Z14Z-eicosatetraenoic
acid (8S-HETE) have only been found in the skin of mice.
It is a potent activator of the peroxisome proliferator-activated receptor PPARα, it is an anti-tumorigenic agent towards skin cancer,
and it promotes wound healing in the cornea.
In contrast, a human orthologue of this enzyme (15‑LOX‑2) is found in skin, sebaceous glands and prostate tissue but produces
15S-HETE.
12S-Hydroxy-5Z,8Z,10E,14Z-eicosatetraenoic acid (12S-HETE) is the precursor of the hepoxilins but has functions of its own. In nervous tissue, it modulates membrane properties and stimulates melatonin synthesis, while together with 15S‑HETE, it serves as a secondary messenger in synaptic transmission and is involved in learning and memory processes; increased levels are found in Alzheimer's disease, and it is the main isomer identified during brain ischemia. 12S‑HETE can either stimulate or inhibit aggregation in platelets, depending on species and circumstances, and it can cause constriction of blood vessels but inhibition of thromboxane (TxA2)-induced platelet aggregation while stimulating lipoxin synthesis. The serum level of 12‑HETE has been shown to be elevated in individuals with coronary heart disease, and in patients threatened with thoracic aortic dissection, it is an endogenous ligand for the BLT2 receptor on macrophages that can activate a signalling cascade and lead to release of inflammatory cytokines; this can eventually trigger vascular degeneration, dissection even and rupture. In leukocytes, it promotes chemotaxis and induces the synthesis of heat-shock protein. In addition, plasma concentrations of 12-HETE correlated well with the severity of the disease in COVID-19.
Particular attention has been devoted to how 12S‑HETE suppresses cancer cell apoptosis and promotes cancer cell invasion, motility, and tumour angiogenesis. It inhibits the adhesion of cancer cells to endothelial cells, and this has been linked to metastasis in cancer of the prostate with mediation via cell surface signalling and activation of protein kinase C; it promotes the proliferation of ovarian and other cancer cells by various mechanisms. Extracellular vesicles derived from platelets promote tumour metastasis because they deliver 12‑lipoxygenase to cancer cells where free and phospholipid-esterified 12S‑HETEs are generated. A deficiency in platelet 12-LOX is reported to exacerbate inflammation and disease severity during SARS-CoV-2 infection.
The enantiomeric compound 12R-HETE is a factor in the pathophysiology of psoriasis and related skin diseases, although it is crucial for the development of normal skin and it may have a function in the eye. 12S‑Hydroxy-8,10,14-eicosatrienoic acid (12S‑HETrE), derived from dihomo-γ-linolenic acid (20:3(n‑6)), has been shown to provide protection against thrombotic-mediated events in vivo and has a potential therapeutic role in providing cardioprotection by interacting with the prostacyclin receptor (IP).
The hydroperoxide precursors of the various HETE isomers tend to be less studied, but 12S-hydroperoxy-ETE is reported to be a key player in oxidative stress in platelets and is known to stimulate the metabolism or arachidonate and other polyunsaturated fatty acids by activating phospholipase A2 (cPLA2) and cyclooxygenase (COX-1), the first enzymes required for prostanoid production.
15S-Hydroxy-5Z,8Z,11Z,13E-eicosatetraenoic acid (15S-HETE) is a precursor of the lipoxins and is synthesised by two lipoxygenases in human tissues, one of which (15-LOX-1) is related structurally to the 12-lipoxygenase of leukocytes and is unusual in that it yields some 12‑HETE in addition to the 15‑isomer. The second isoform of the enzyme was first found in the epidermis, although it is now known to exist in other tissues. 15S‑HETE has been implicated in cell differentiation, inflammation, asthma and carcinogenesis. It plays a dual role in inflammatory responses as it can exert anti-inflammatory effects by inhibiting the 5-lipoxygenase pathway to reduce the production of pro-inflammatory leukotrienes, but in adipocytes in vitro, it can stimulate the production of pro-inflammatory cytokines and monocyte chemoattractant protein (MCP). In atherogenesis, there is an accumulation of 15‑HETE in human carotid plaques, and this may assist in the induction of atherothrombotic events by increasing platelet aggregation and thrombin generation. 15‑HETE contributes to the development of Hodgkin lymphoma, and colorectal, lung and many other cancers, but that from 15‑LOX‑2 activates PPARγ, a nuclear transcription factor involved in epithelial differentiation, which may explain an anti-proliferative action on prostate cancer cells. 15-LOX-1 is a feature of the processes of apoptosis, autophagy and ferroptosis, and reduced levels of this enzyme in some cancers lead to decreased activity of PPARγ, resulting in a halt to apoptosis and enhanced cell proliferation.
Of the terminal and near-terminal HETE isomers (cytochrome P450 metabolites), 20-HETE is regarded as a critical regulator of multiorgan homeostasis as it maintains vascular tone by promoting vascular smooth muscle cell contraction and proliferation and migration of vascular and endothelial cells, acting via the G protein receptor GPR75. It may prevent septic shock and multi-organ failure induced by bacterial lipopolysaccharides, and it stabilizes blood pressure and exerts a beneficial effect in terms of insulin secretion; in the kidney, it is anti-hypertensive by blocking re-absorption of sodium by inhibiting the Na+‑K+‑ATPase. In contrast, excessive 20-HETE synthesis has been associated with metabolic disorders, which include some renal diseases, hypertension, obesity, diabetes, and non-alcoholic fatty liver disease, so it has sometimes been considered to be a pro-inflammatory agent. It may be a factor in rheumatoid arthritis, and production is increased after the onset of both ischemic and hemorrhagic stroke.
Although 20‑HETE may promote tumour growth, 8- and 11‑HETE are anti-tumour agents. EPA and DHA are potent inhibitors of the biosynthesis of 20‑HETE, suggesting that this may be a partial explanation for the physiological role of omega-3 fatty acids, although other HETE isomers may act in opposition to it, and 18- and 19‑HETE induce vasodilatation by inhibiting 20‑HETE. Together with 16- and 17‑HETE, they induce re-uptake of sodium in the kidney, while 16‑HETE inhibits neutrophil adhesion so may be relevant to inflammation. On the other hand, high 19-HETE concentrations have been correlated with cardiovascular events.
 3-Hydroxy-eicosanoids, e.g. 3R-hydroxy-HETE or
3R-hydroxy-5Z,8Z,11Z,14Z-eicosatetraenoic acid from pathogenic fungi may take part in the
inflammatory processes associated with infections by such organisms, as together with down-stream metabolites from their hosts, they are
strong pro-inflammatory lipid mediators.
As they are synthesised during the reproductive phase of growth, yeast and fungi presumably require them for their own internal purposes.
3-Hydroxy-eicosanoids, e.g. 3R-hydroxy-HETE or
3R-hydroxy-5Z,8Z,11Z,14Z-eicosatetraenoic acid from pathogenic fungi may take part in the
inflammatory processes associated with infections by such organisms, as together with down-stream metabolites from their hosts, they are
strong pro-inflammatory lipid mediators.
As they are synthesised during the reproductive phase of growth, yeast and fungi presumably require them for their own internal purposes.
Metabolites of EPA and DHA: The EPA lipoxygenase metabolite 5-hydroxy-eicosapentaenoic acid (5-HEPE) enhances the induction of regulatory T cells (Tregs) that modulate the immune system and prevent autoimmune disease, and it can increase insulin secretion from pancreatic beta cells in mice. Both 5- and 12‑HEPE are induced in brown fat when exposed to cold, and they have been termed 'batokines' or 'lipokines' that improve glucose metabolism by promoting glucose uptake into adipocytes and skeletal muscle through triggering an insulin-like intracellular signalling pathway. 12-HEPE is the main oxylipin produced by platelets in the presence of EPA, and it inhibits agonist-stimulated platelet aggregation with benefits towards cardiovascular protection by reducing thrombus formation. Apart from being a precursor of E‑series resolvins, 18‑HEPE per se has cardioprotective properties and inhibits metastasis in a cancer model. Various HEPE isomers have been detected in psoriatic arthritis, where they may be anti-inflammatory.
It is noteworthy that while 17-hydroxy-DHA derived from the action of 15-LOX is often considered simply as a precursor of the specialized pro-resolving mediators (SPMs), there is evidence that it (rather than the latter) alleviates the sensitivity to heat pain and osteoarthritis pain in humans. Its metabolite, 17-oxo-DHA, contributes to the resolution of inflammation by enhancing efferocytosis, i.e., the clearance of apoptotic granulocytes by phagocytes, through stimulation of the biosynthesis of the SPM resolvin D2 (RvD2). In brain, 7S‑HDHA is a high-affinity PPARα ligand that stimulates the growth of neurons and regulates the expression of genes associated with their morphology. In obese mice, 17‑HDHA attenuated inflammation in adipose tissue and improved insulin sensitivity and glucose tolerance.
Epoxyeicosatrienoic acids (EETs): The various EETs are crucial autocrine and paracrine effectors in the cardiovascular and renal systems that are anti-inflammatory and largely beneficial. As the regioisomers and enantiomers have many similar metabolic and functional properties, epoxyeicosatrienoic acids have often been treated as a single class of compounds, although as knowledge has expanded this view is no longer justifiable. 11,12-EET in particular induces distinctive responses in tissues. Because of the anti-hypertensive, anti-fibrotic and anti-thrombotic properties of EETs, their presence in red blood cells has implications for the control of circulation and the physical properties of the circulating blood. Both cis- and trans-EETs are synthesised and stored in erythrocytes, and they are produced and released in response to a low oxygen concentration as during exercise. 11,12‑EET enhances the process by which immature precursor cells develop into mature blood cells (hematopoiesis) and in their further development (engraftment) in mice and zebrafish in vitro.
In the kidney, EETs modulate ion transport and gene expression to cause vasodilation, and in other tissues, they exert beneficial effects on insulin resistance and obesity-associated diseases, they alleviate inflammatory pain, neuroinflammation and neuroinflammatory diseases, and they improve lung function and wound healing. By direct and indirect anti-inflammatory actions in the heart, they are cardioprotective and alleviate cardiomyopathy and cardiac remodelling while regulating cardiac electrophysiology. There is a suggestion that signalling by 11,12-EET may be a factor in the regulation of the response to DNA damage, and it is reported to ease the symptoms of pulmonary fibrosis. In the central nervous system, epoxyeicosanoids regulate the release of neurohormones and neuropeptides, and by reducing the long-term damage associated with central neurologic insults, they may be beneficial towards neurologic diseases including Parkinson's disease, Alzheimer's disease and dementia.
As significant amounts of EETs are incorporated into phospholipids from which they are rapidly released in the presence of Ca2+ ionophores, it has been suggested that they may take part in those signal transduction processes mediated by phospholipases. In non-failing human hearts, one isoform of phospholipase A (cPLA2ζ) channels arachidonic acid into protective EETs, whereas in failing hearts, opening of the mitochondrial permeability transition pore increases the activity of a second isoform of phospholipase A (cPLA2γ) that channels arachidonic acid into toxic HETEs.
For some purposes, epoxy-eicosanoids would be expected to require cell-surface receptors, and GPR132 has been identified tentatively as a potentially low-affinity EET receptor with physiological relevance in hematopoiesis (a high-affinity receptor has yet to be identified), but they can also make use of intracellular mechanisms, i.e., direct interaction with ion channels, signalling proteins or transcription factors. They may act as intracellular mediators in responses elicited upon some hormone-receptor interactions, including those with epidermal growth factor, vascular endothelial growth factor and insulin binding. Through interactions with select binding pockets, fatty acid binding proteins (FABP3 and FABP5) modulate signalling of EETs in synapses in the brain. Their concentrations are controlled by soluble epoxide hydrolases, and it is hoped that inhibitors of these will be developed with therapeutic potential against some debilitating inflammatory diseases.
 17,18-Epoxyeicosatetraenoic acid is the main epoxide regio-isomer synthesised
from eicosapentaenoic acid and has anti-allergy and anti-inflammatory properties by activating the receptor GPR40;
it may have therapeutic properties in the skin and intestines.
In sensory neurons, it acts through the prostacyclin receptor (IP) to sensitize the transient receptor potential vanilloid 1 (TRPV1).
Acting via the sphingosine-1-phosphate receptor 1, it mediates the atheroprotective effect of eicosapentaenoic acid.
It is a vasodilator and may be responsible for some of the beneficial effects of dietary omega-3 fatty acids.
While 19,20-epoxy-docosapentaenoic acid derived from DHA has been shown to have positive effects in tissues,
much less is known of the other oxygenated metabolites of EPA and DHA, although they can act in opposition to HETE
isomers and may be important in the cardiovascular system and as anti-cancer agents.
Until recently, EETs were believed to be relatively benign molecules, but it has now been demonstrated in mice that they are
stimulants for the release of primary cancer tumours from dormancy, for promoting their growth and for triggering metastasis; their dihydroxy
metabolites have even stronger effects upon cancer progression.
17,18-Epoxyeicosatetraenoic acid is the main epoxide regio-isomer synthesised
from eicosapentaenoic acid and has anti-allergy and anti-inflammatory properties by activating the receptor GPR40;
it may have therapeutic properties in the skin and intestines.
In sensory neurons, it acts through the prostacyclin receptor (IP) to sensitize the transient receptor potential vanilloid 1 (TRPV1).
Acting via the sphingosine-1-phosphate receptor 1, it mediates the atheroprotective effect of eicosapentaenoic acid.
It is a vasodilator and may be responsible for some of the beneficial effects of dietary omega-3 fatty acids.
While 19,20-epoxy-docosapentaenoic acid derived from DHA has been shown to have positive effects in tissues,
much less is known of the other oxygenated metabolites of EPA and DHA, although they can act in opposition to HETE
isomers and may be important in the cardiovascular system and as anti-cancer agents.
Until recently, EETs were believed to be relatively benign molecules, but it has now been demonstrated in mice that they are
stimulants for the release of primary cancer tumours from dormancy, for promoting their growth and for triggering metastasis; their dihydroxy
metabolites have even stronger effects upon cancer progression.
The dihydroxyeicosatrienoic acids (DHETr) from epoxide hydrolases are pro-inflammatory in general, and they have been associated with colonic inflammation in obese mice and with osteoarthritis in the knee in humans. Of special note, elevated concentrations of 11,12-diHETrE in neonatal cord blood have been correlated with the severity of the symptoms of autism spectrum disorders after birth and with impaired adaptive functioning, especially in girls. Acting via ferroptosis, dihydroxy-metabolites of dihomo-γ-linolenic acid (20:3(n-6)) are reported to cause neurodegeneration, while 19,20‑dihydroxydocosapentaenoic acid has a role in the development of diabetic retinopathy. As dihydroxy metabolites may be factors in psychiatric and neurological disorders by weakening the blood brain barrier, the soluble epoxide hydrolase (EPHX1) is a therapeutic target for such conditions as well as by assisting in cardiac recovery after ischemia. Inhibitors of the enzyme are undergoing clinical trials for several inflammatory conditions, including cancer.
Octadecanoids: The oxidized linoleate metabolites 13S-HODE and 9S-HODE are atherogenic through the induction of pro-inflammatory cytokines and production of foam cells from macrophages by activation of PPARs, e.g., PPARγ, and other receptors. 9S‑HODE but not 13S-HODE is a high-affinity ligand of GPR132 and is proinflammatory with the first of these a marker for oxidative stress. Both (and their keto metabolites) contribute to the process of pain perception and itchiness in skin by interactions with the main receptors involved in the pain circuit, e.g., TRPV1. In a Drosophila model, 9S‑HODE regulates the FOXO family of transcription factors. HODE are found at increased levels in psoriatic skin, and they contribute to hepatic injury, including non-alcoholic steatohepatitis, possibly by non-enzymatic formation of potentially toxic adducts with proteins. As the actions of octadecanoids on the regulation of inflammation are of relevance to the metabolic processes associated with atherogenesis and cancer, they are attracting increasing interest.
In contrast, there is evidence that a 15-LOX metabolite 13S-HPODE induces apoptosis in colon cancer cells. 13S-HODE in the brains of rat pups is reported to increase axonal outgrowth cortical neurons in males significantly, but not in females where linoleic acid per se acts in this way. In general, in brain, HODE regulate pain thresholds, inflammation, neurotransmission and the response to ischemic brain injury.
Epoxy-octadecenoic acids (epOMEs) are metabolites of linoleate from the cytochrome P450 monooxygenase pathway that are cancer-promoting in the colon via mechanisms that involve the gut microbiota. In insects, they take part in the resolution of cellular and humoral immune reactions. 12,13-EpOME is present in plasma of healthy humans, and it is involved in the regulation of monocyte differentiation during inflammation.
At high levels in humans, the dihydroxy-metabolites (DiHOMEs) are vascular permeability and cytotoxic agents associated with multiple organ failure, adult respiratory distress syndrome, and sepsis in burn patients, although they are not implicated in colon cancer. In severe burn injury, they drive immune cell dysfunction through hyperinflammatory neutrophilic and impaired monocytic actions, so inhibition of soluble epoxide hydrolase may be a promising therapeutic strategy. 9,10-DiHOME produced by commensal bacteria in the intestines facilitates regulatory T cell differentiation and may be a biomarker for colitis. 12,13‑Dihydroxy-9Z-octadecenoic acid (12,13‑diHOME) causes increased sensitivity to inflammatory pain and has been associated with the development of paediatric asthma, and by promoting inflammatory macrophages, it reduces the ability to respond to microbes and allergens. In contrast, it is reported to reduce the risk of myocardial infarction by improving cardiac systolic and diastolic functions. Both 9,10- and 12,13-diHOME are reported to be biomarkers for small versus large vessel stroke, together with blood brain barrier and neurovascular-glial disruption and temporal lobe atrophy. Elevated 12,13-diHOME levels occur in maternal and umbilical cord blood during preeclampsia, while it has been correlated (with noradrenaline) with the occurrence of acute myocardial infarction and cognitive decline in patients with type 2 diabetes mellitus.
On the other hand, synthesis of 12,13‑diHOME is induced by cold with the effect of stimulating brown adipose tissue by promoting the uptake of fatty acids. As plasma levels are increased by exercise when the source is probably brown adipose tissue, 12,13-diHOME has been termed an exercise-stimulated lipokine (or batokine), which triggers an increase in fatty acid oxidation and uptake in skeletal muscle that results in improved whole-body metabolic homeostasis and may be cardioprotective. It has been detected in human milk where it may influence infant adiposity, while 9,10-DiHOME, 12,13-DiHOME, and the α-linolenic acid metabolites 9,10-DiHODE and 12,13-DiHODE are potential drivers of obesity in adults.
Hepoxilin-like triHOMEs are important in skin, where they are vital for epidermal barrier assembly (and some argue for mammalian survival), but their levels are dysregulated in asthma and chronic obstructive pulmonary disease (COPD). In psoriasis, 9,10‑epoxy-13-hydroxy-octadecenoate and 9,10,13-trihydroxy-octadecenoate contribute to itch and pain.
Oxidized phospholipids: Although studies are at a relatively early stage, it is apparent that esterified HETEs have their own functions in relation to apoptosis, immune regulation, signalling and blood coagulation. Oxidation of cardiolipin by an enzyme of the cytochrome c family is an significant event in triggering mitochondrial apoptosis, while phosphatidylserine containing oxidized fatty acids is externalized to the surface of cells and is a signal for engulfment and digestion of apoptotic cells. Phosphatidylethanolamines containing arachidonic and adrenic acids that are oxidized by 15-LOX are pro-ferroptotic signals, but oxidized phosphatidylcholine is reported to be anti-inflammatory and protective in the lung. 12S‑HETE-lysophospholipids react selectively with certain human monocytes to generate tumour necrosis factor α (TNFα) and thence initiate a key signalling pathway; they may serve as biomarkers for age-related diseases and could potentially be used as targets for therapeutic intervention. Indeed, dysregulation of the metabolism of any of these molecules may have implications for human health. Further effects are discussed in our web page on oxidized phospholipids, including those from non-enzymatical reaction.
8. Fungal and Bacterial Enzymes
Certain fungi and yeasts synthesise 3R- and/or 3S-HETE and 3,18-di-HETE, when supplied with exogenous arachidonic acid from their animal hosts during infection. The biochemical mechanism is unclear, and there are reports that implicate lipoxygenases, cyclooxygenases or mitochondrial β‑oxidation. With some pathogenic fungi, the 3-hydroxyeicosanoids produced in infected cells (illustrated above) can be acted upon by the host COX-2 enzyme to form a family of 3-hydroxy-prostaglandins, which are at least as active as the normal compounds. Aspergillus fumigatus has two lipoxygenase homologues of human ALOX15 and ALOX5, termed LoxA and LoxB, of which the latter is secreted extracellularly and yields 13‑hydroxyoctadecadienoic acid (13‑HODE) from linoleate; other fungi are now know to synthesise many different octadecanoids from C18 precursor fatty acids.
Some fungi have peroxygenases that introduce oxygen atoms into non-activated carbon-hydrogen bonds of aliphatic and aromatic compounds. As they only require H2O2 for catalysis and not cofactors and complex regeneration systems, they have great biotechnological potential.
Prokaryotic LOX-encoding genes are best known from Pseudomonas aeruginosa (Gram-negative bacteria) with the capacity to produce 15S‑hydroxyeicosatetraenoic acid (15S‑HETE), but these are distant evolutionarily from human LOX. This species has a cytochrome P450 enzyme, CYP168A1, which is a subterminal fatty acid hydroxylase that can make 19-HETE from arachidonic acid. Many other bacteria are now known to have lipoxygenases, and some of these can utilize oleic, linoleic or polyunsaturated fatty acids as substrates with high positional and stereochemical specificity, while others, including Rhodococcus sp. and Bacillus megaterium, have monooxygenases of the P450 family for a wide range of oxylipins.
In the gut, bacteria produce 12Z- and 11E-10-hydroxyoctadecenoic acids (HOME) and many other octadecanoids by enzymatic oxidation of linoleic acid, mainly by various hydratases, and these can eventually be absorbed by the intestines and appear in plasma. Conjugated linoleic (CLA) and linolenic acids are metabolized in the same way. Such fatty acid hydratases are present in bacteria but not in Eukaryotes, and these oxidize fatty acids by adding the elements of water to double bonds, with flavin adenine dinucleotide (FAD) as a cofactor, presumably incidental to the detoxification of environmental toxins as their primary purpose. For example, oleate hydratases, such as that from Staphylococcus aureus, catalyse the hydroxylation of oleic acid to 10R-hydroxy-stearic acid, a reaction that may prove useful in industry.
 |
| Figure 12. Hydroxylation of oleic acid by a bacterial hydratase. |
9. Analysis
The remarkable array of eicosanoids and related oxygenated metabolites in animal tissues provides a daunting task for analysts, partly because of their sensitivity to any manipulation at low natural concentrations. Fortunately, many primary standards, including deuterated molecules, are now available commercially to assist in the task of comparison, calibration or validation, although gaps remain. Selective extraction, concentration and derivatization steps are required, followed by gas chromatography or high-performance liquid chromatography linked to mass spectrometry. The topic has been reviewed by Schebb, N.H. and ~100 others (2025), DOI, where recommendations for best practice are set out. Equally authoritative for the analysis of octadecanoids is Quaranta, A. and others (2022); DOI.
Recommended Reading
- Aranda, C., Carro, J., Gonzalez-Benjumea, A., Babot, E.D., Olmedo, A., Linde, D., Martinez, A.T. and Gutierrez, A. Advances in enzymatic oxyfunctionalization of aliphatic compounds. Biotechn. Adv., 51, 107703 (2021); DOI.
- Benatzy, Y., Palmer, M.A. and Brüne, B. Arachidonate 15-lipoxygenase type B: Regulation, function, and its role in pathophysiology. Front. Pharm., 13, 1042420 (2022); DOI.
- Biringer, R.G. The enzymology of human eicosanoid pathways: the lipoxygenase branches. Mol. Biol. Rep., 47, 7189-7207 (2020); DOI.
- Biringer, R.G. A review of non-prostanoid, eicosanoid receptors: expression, characterization, regulation, and mechanism of action. J. Cell Commun. Signal., 16, 5-46 (2022); DOI.
- Chrisnasari, R., Hennebelle, M., Vincken, J.P., van Berkel, W.J.H. and Ewing, T.A. Bacterial lipoxygenases: Biochemical characteristics, molecular structure and potential applications. Biotech. Adv., 61, 108046 (2022); DOI.
- Eccles, J.A. and Baldwin, W.S. Detoxification cytochrome P450s (CYPs) in families 1-3 produce functional oxylipins from polyunsaturated fatty acids. Cells, 12, 82 (2023); DOI.
- Hajeyah, A.A., Griffiths, W.J., Wang, Y., Finch, A.J. and O’Donnell, V.B. The biosynthesis of enzymatically oxidized lipids. Front. Endocrinol., 11, 591819 (2020); DOI.
- Hou, Y.-N., Liu, S.-J., Chen, F., Wang, S., Song, S.-Y., Yang, W.-Q. and Li, L.-M. 20-HETE: Its potential role in physiological and pathophysiological processes. Biochem. Pharm., 242, 117238 (2025); DOI.
- Lin, L., Dai, F., Wei, J.J. and Chen, Z. Biological roles of 5-oxo-6,8,11,14-eicosatetraenoic acid and the OXE receptor in allergic diseases: Collegium Internationale Allergologicum Update (2024). Int. Arch. Allergy Immunol., 185, 301-310 (2024); DOI.
- McReynolds, C., Hammock, B. and Morisseau, C. Regulatory lipid vicinal diols counteract the biological activity of epoxy fatty acids and can act as biomarkers and mechanisms for disease progression. Pharmacol. Therapeut., 248, 108454 (2023); DOI.
- Mendoza, S.R., Zamith-Miranda, D., Takács, T., Gacser, A., Nosanchuk, J.D. and Guimarães, A.J. Complex and controversial roles of eicosanoids in fungal pathogenesis. J. Fungi, 7, 254 (2021); DOI.
- Meng, Y.-W. and Liu, J.-Y. Pathological and pharmacological functions of the metabolites of polyunsaturated fatty acids mediated by cyclooxygenases, lipoxygenases, and cytochrome P450s in cancers. Pharmacol. Therapeut., 2561, 108612 (2024); DOI.
- Oliw, E.H. Thirty years with three-dimensional structures of lipoxygenases. Arch. Biochem. Biophys., 752, 109874 (2024); DOI.
- Perepechaeva, M.L. and Grishanova, A.Y. The role of arachidonic acid metabolizing cytochromes P450 in the control of cardiovascular functions. Biochem.-Moscow, B. Biomed. Chem., 18, 192-213 (2024); DOI.
- Revol-Cavalier, J., Quaranta, A., Newman, J.W., Brash, A.R., Hamberg, M. and Wheelock, C.E. The octadecanoids: synthesis and bioactivity of 18‑carbon oxygenated fatty acids in mammals, bacteria, and fungi. Chem. Rev., 125, 1-90 (2025); DOI.
- Schebb, N.H. and ~100 others. Technical recommendations for analyzing oxylipins by liquid chromatography-mass spectrometry. Sci. Signal., 18, 678 (2025); DOI.
- Shi, Z.Q., He, Z.W. and Wang, D.W. CYP450 epoxygenase metabolites, epoxyeicosatrienoic acids, as novel anti-inflammatory mediators. Molecules, 27, 3873 (2022); DOI.
- Singh, N.K. and Rao, G.N. Emerging role of 12/15-Lipoxygenase (ALOX15) in human pathologies. Prog. Lipid Res., 73, 28-45 (2019); DOI.
- Wang, B., Wu, L.J., Chen, J., Dong, L.L., Chen, C., Wen, Z., Hu, J., Fleming, I. and Wang, D.W. Metabolism pathways of arachidonic acids: mechanisms and potential therapeutic targets. Signal Transd. Targ. Ther., 6, 94 (2021); DOI.
- Zhou, M.Z., Li, J.D., Xu, J.Y., Zheng, L.F. and Xu, S.T. Exploring human CYP4 enzymes: Physiological roles, function in diseases and focus on inhibitors. Drug Discovery Today, 28, 103560 (2023); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: January 2026 | ||
© The LipidWeb is open access and fair use is encouraged - but not text and data mining, AI training, and similar technologies (text scraping).
