Cardiolipin (Diphosphatidylglycerol)
Cardiolipin is the trivial but universally used name for a lipid that might be better termed 'diphosphatidylglycerol' or more precisely 1,3‑bis(sn‑3'-phosphatidyl)-sn-glycerol. It is a unique phospholipid with a dimeric structure in essence by comparison with other phospholipids, in that it has four acyl groups, five chiral centres and potentially two negative charges. The tetra-linoleoyl molecular species, important in heart mitochondria, is illustrated.

Cardiolipin is present universally in both prokaryotes and eukaryotes where it is an essential phospholipid component of membranes in those organelles that generate an electrochemical potential for substrate transport and ATP synthesis. Thus, it is found almost exclusively in mitochondria of eukaryotes and in certain membranes of bacteria (plasma membrane and hydrogenosomes) and a few Archaea (haloarchaea). Although it has sometimes been termed the 'signature lipid' of the mitochondrion in that its presence in the membranes of an organelle identifies that organelle as a mitochondrion, this may overstate the case as cardiolipin is reported to be a significant constituent of the membranes of peroxisomes (although it probably originates in mitochondria) and occasionally in other organelles and body fluids (although all analyses are not equally trustworthy). The trivial name 'cardiolipin' is derived from the fact that it was first found in animal hearts, where it is most abundant, but it can be found in mitochondria of all animal tissues and indeed of the eukaryotic kingdom.
1. Structure and Composition
Cardiolipin amounts to about 10% of the phospholipids of bovine heart muscle, and 20% of the phospholipids of the mitochondrial membranes in this organ, and it is a substantial component of the phospholipids of skeletal muscle, which has a high demand for energy production, but other organs tend to have much lower amounts. In general, the phospholipid composition of mitochondrial membranes is highly conserved from yeast to humans, with each lipid class having necessary functions in the assembly and activity of mitochondrial proteins, but especially those for oxidative phosphorylation.
 A common feature of this
lipid in a variety of very different organisms is a relatively simple fatty acid and molecular species composition,
leading to a high degree of structural symmetry.
As there are four fatty acyl groups in cardiolipin, the potential for complexity in the distribution within molecules is enormous,
yet the compositions can be remarkably simple and very different from those of other phospholipids.
In animals, these are relatively resistant to dietary manipulation and cardiolipin tends to contain 18-carbon fatty acids almost exclusively
with 80% of this typically as linoleic acid (18:2(n‑6)).
Among other tissues, testis cardiolipin is an exception in that it contains mainly palmitic acid,
while cardiolipin in the central nervous system contains a much wider range of fatty acids including palmitic, stearic, oleic,
arachidonic and docosahexaenoic acids (over 100 molecular species); lymphoblast cardiolipin contains only monoenoic fatty acids.
Most mammals have similar compositions, but bovine heart cardiolipin differs in that it is enriched in α‑linolenic acid
(18:3(n‑3)).
A common feature of this
lipid in a variety of very different organisms is a relatively simple fatty acid and molecular species composition,
leading to a high degree of structural symmetry.
As there are four fatty acyl groups in cardiolipin, the potential for complexity in the distribution within molecules is enormous,
yet the compositions can be remarkably simple and very different from those of other phospholipids.
In animals, these are relatively resistant to dietary manipulation and cardiolipin tends to contain 18-carbon fatty acids almost exclusively
with 80% of this typically as linoleic acid (18:2(n‑6)).
Among other tissues, testis cardiolipin is an exception in that it contains mainly palmitic acid,
while cardiolipin in the central nervous system contains a much wider range of fatty acids including palmitic, stearic, oleic,
arachidonic and docosahexaenoic acids (over 100 molecular species); lymphoblast cardiolipin contains only monoenoic fatty acids.
Most mammals have similar compositions, but bovine heart cardiolipin differs in that it is enriched in α‑linolenic acid
(18:3(n‑3)).
In some marine animals, cardiolipin contains appreciable amounts of docosa- or tetracosahexaenoic acids (22:6(n-3) (DHA) or 24:6(n‑3)), and zebrafish (Danio rerio) cardiolipin contains ~45% DHA (22:6), together with mainly stearic and oleic acids, while some molluscs can contain 80% DHA. 16:1 and 18:2 are the main fatty acids in cardiolipin from the fruit fly Drosophila melanogaster.
In the cardiolipin of higher plants, linolenic and linoleic acids predominate, and the most abundant molecular species contain 10 or 11 double bonds in total. Leaf and seeds of Arabidopsis thaliana contain 61 and 58% of 18:3 and 35 and 37% of 18:2, respectively, in their cardiolipin. This lipid from the yeast Saccharomyces cerevisiae differs in having mainly 16:1 and 18:1 fatty acids, while that from the fungus Aspergillus fumigatus contains 18:2, 16:0 and 18:1 fatty acids as the main components. Bacterial cardiolipin, such as that in Escherichia coli or Bacillus drentensis, contains predominantly saturated and monoenoic fatty acids with 14 to 18 carbons, while some bacteria contain mono- and dialk-1-enyl forms (plasmalogens) as well as tetra-acyl species; a sulfate-reducing bacterium, Desulfatibacillum alkenivorans produces molecules containing one (monoether/triester) to four (tetraether) ether-linked saturated straight-chain or methyl-branched alkyl chains.
Detailed lipidomic analysis of cardiolipin, including the positional distributions on the various glycerol moieties, was a technically daunting task before the advent of modern mass spectrometric methodology, but it has been accomplished for many different animals and data for bovine heart and rat liver are listed in Table 1.
Table 1. The main molecular species of mammalian cardiolipin. |
||||
| Molecular species* | Amount | |||
|---|---|---|---|---|
| Fatty acid 1A | Fatty acid 2A | Fatty acid 1B | Fatty acid 2B | (Mol%) |
| Bovine heart | ||||
| 18:2 | 18:2 | 18:2 | 18:2 | 48 |
| 18:3 | 18:2 | 18:2 | 18:2 | | 21 | |
| 18:2 | 18:3 | 18:2 | 18:2 | |
| 18:2 | 18:2 | 18:2 | 18:3 | |
| 18:2 | 18:2 | 18:3 | 18:2 | |
| 18:2 | 18:1 | 18:2 | 18:2 | 15 |
| 18:2 | 18:2 | 18:2 | 18:1 | |
| Rat liver | ||||
| 18:2 | 18:2 | 18:2 | 18:2 | 57 |
| 18:2 | 18:1 | 18:2 | 18:2 | 35 |
| 18:2 | 18:2 | 18:2 | 18:1 | |
| * Fatty acyl residues are designated as shown in the structure illustrated above. Data reproduced from Schlame, M. et al. Eur. J. Biochem., 212, 727-735 (1993); DOI, or see Emaus, K.J. et al. (2025) for a recent lipidomic study (DOI) or Weill, P.W. et al. for fatty acid compositional data in mice (DOI). |
||||
Until relatively recently, it was thought that cardiolipin was associated exclusively with the mitochondrial inner membrane in Eukaryotes with a higher abundance in the matrix leaflet. In bovine heart mitochondria, it represents about 25% of the total phospholipids, with most in the inner membrane, though approximately 4% is located in the outer membrane. This may be significant in that it may predominate at sites connecting the outer membrane with the inner membrane, where its unique physical properties are necessary to enable transport of lipids between them. Collapse of this locational asymmetry in a membrane may herald a signalling event. It is found mainly in the inner membranes of bacterial cells, but it is externalized in Gram-negative bacteria as they encounter host cells.
The highly specific location of cardiolipin is used as an argument in favour of the hypothesis that mitochondria are derived from prokaryotes, which lived inside a eukaryotic progenitor cell in symbiosis. If this did indeed occur, the function and importance of cardiolipin has changed during evolution, as mitochondria need a constant level of cardiolipin to operate efficiently, while prokaryotes may only require it in a few circumstances.
Physical properties. Even with four identical acyl residues, cardiolipin has two chemically distinct phosphatidyl moieties located at two chiral centres, one in each outer glycerol group, i.e., one is in a pro-R and the other in a pro-S position with respect to the central carbon atom of the glycerol bridge. These could in theory give rise to diastereomers, although natural cardiolipin has the R/R configuration. In consequence, the two phosphate groups have different chemical environments and thence separate 31P-NMR resonances; they are designated 1'-phosphate and 3'-phosphate with respect to the central glycerol. Each contains one acidic proton, but they have different levels of acidity with recent data suggesting a pK1 of 2.15, like that of phosphoric acid, while pK2 is only about one unit greater. The weaker acidity of the second phosphate is the result of formation of a stable intramolecular hydrogen bond with the central 2'‑hydroxyl group. Cardiolipin thus carries a double negative charge at physiological pH, and not a single charge in a tight bicyclic resonance structure as was once proposed.
Because of this unique structure, cardiolipin can yield micellar, lamellar and hexagonal states in aqueous dispersions, depending on pH and ionic strength. It probably exists mainly in the bilayer of natural membranes with its two phosphatidylglycerol moieties oriented perpendicular to the bilayer surface and the central glycerol group oriented at the water/membrane interface and parallel to it. As a cone-shaped structure, with the relatively small polar region at the top and the flexible acyl chains at the base of the "cone”, it exerts lateral pressure and segregates into regions or microdomains of locally high membrane curvature. It is located in and contributes to the highly curved regions in the inner mitochondrial membrane facing the cristae to maintain mitochondrial architecture and function; a deficiency of cardiolipin can lead to disintegration of the cristae and disruption of the membranes. The positively curved outer monolayer leaflet contains mainly phosphatidylcholine with lesser amounts of other phospholipids. In addition, the distribution of cardiolipin is determined by the lipid scaffolding proteins prohibitin-1 and prohibitin-2, which may facilitate the remodelling process as part of the biosynthetic machinery (see next section), while the interaction of cardiolipin with the mitochondrial fusion protein OPA1 controls the membrane-shaping mechanism and has a further influence.
There remains a tendency of cardiolipin to generate transient non-bilayer domains, which could have a profound influence on vital cellular processes. For example, the four linoleate residues in heart cardiolipin tend to spread laterally in a bilayer causing it to thin, and together with the compact head group, this leads to a hexagonal phase, which is responsible for its location in the inner mitochondrial membrane at contact sites with the outer mitochondrial membrane. In contrast, the mainly palmitate/vaccenate residues in cardiolipin from E. coli favour bilayers.
As the head-group glycerol of cardiolipin is shared by two phosphatidate moieties, its mobility is severely restricted, reducing its capacity for either intra- or inter-molecular interactions with other phospholipid head-groups. The secondary hydroxyl group of the central glycerol moiety is the sole source of hydrogen-bonding donor groups available for such interactions, so that bonding to other lipids is unlikely. It should be recognized that as the two phosphate groups are chirally distinct, there exist opportunities for differential metabolic reactions of each under both chiral and achiral conditions.
2. Biosynthesis and Metabolism
Unique sets of enzymes and reactions are utilized for cardiolipin biosynthesis, and these are very different in eukaryotes and prokaryotes. With the former (yeasts, plants and animals), the biosynthetic pathway is like that of some other phospholipids in that it passes through the common intermediate phosphatidic acid, which is imported from the endoplasmic reticulum and transported to the inner mitochondrial membrane by certain protein complexes at contact sites between the two membranes. The first committed step in biosynthesis is formation of phosphatidylglycerol phosphate as an intermediate for subsequent production of the precursor phosphatidylglycerol. The second precursor is cytidine diphosphate diacylglycerol (CDP-DG) from a distinctive synthase in mitochondria, TAM41 in yeast or TAMM41 in animals, a member of the nucleotidyltransferase (NTase) superfamily, which differs structurally from the CDP-DG synthases in the endoplasmic reticulum. The cardiolipin synthase, a phosphatidyl transferase, then links phosphatidylglycerol to diacylglycerol phosphate from the activated phosphatidyl moiety of CDP-DG, with elimination of cytidine monophosphate (CMP).
 |
| Figure 1. Biosynthesis of cardiolipin by the eukaryotic route. |
The reaction requires a source of energy and the enzymes from all eukaryotes examined in detail need certain divalent cations (Mg2+, Mn2+ or Co2+) together with a high pH (8 to 9). In rat liver and in higher plants, the cardiolipin synthase is a member of the CDP-alcohol phosphatidyltransferase superfamily and resides in the inner mitochondrial membrane, while in yeast, it is part of a large protein complex in mitochondria. The enzymes for the synthesis of the precursors and of cardiolipin per se are located on the inner leaflet (matrix side) of the inner membrane, presumably close to each other and perhaps part of a single protein complex in yeast at least.
In prokaryotes such as bacteria, cardiolipin (diphosphatidylglycerol) synthase (CLS) catalyses a transfer of the phosphatidyl moiety of one phosphatidylglycerol to the free 3'‑hydroxyl group of another, with the elimination of one molecule of glycerol, via the action of one of two structurally related enzymes (depending on the organism), which are part of the phospholipase D superfamily. In effect, transphosphatidylation occurs with one phosphatidylglycerol acting as a donor and the other an acceptor of a phosphatidyl moiety. The reaction is energy independent, and the enzymes can operate in reverse under some physiological conditions to convert cardiolipin back to phosphatidylglycerol, so the biosynthesis of cardiolipin is regulated via that of phosphatidylglycerol.
 |
| Figure 2. Biosynthesis of cardiolipin by the prokaryotic route. |
There are in fact three cardiolipin synthases (ClsA/B/C) in the bacterium E. coli, with ClsA as the primary source during exponential growth. A second minor mechanism produces cardiolipin by condensation of phosphatidylglycerol and phosphatidylethanolamine with elimination of ethanolamine via the action of ClsB/C. There is a very different bifunctional cardiolipin/phosphatidylethanolamine synthase in Xanthomonas campestris, which is related to the phospholipase D superfamily and can synthesise cardiolipin from phosphatidylglycerol and CDP-diacylglycerol, but it also catalyses ethanolamine-dependent phosphatidylethanolamine formation. The Archaea have their own unique cardiolipin synthase, which utilizes archaetidylglycerol, a stereochemically distinct diether analogue of phosphatidylglycerol, as precursor
As eukaryotic cardiolipin synthase is a mitochondrial enzyme and mitochondria are believed to be phylogenetic derivatives of ancient prokaryotes, it may appear strange that there has been such a change in mechanism, but protein domain analyses indicate that both pathways evolved convergently. Surprisingly, Streptomyces coelicolor and other Actinomycetes use the eukaryote biosynthetic system, while the protozoan parasite, Trypanosoma brucei, utilizes the prokaryotic pathway.
Remodelling: The final fatty acid composition of cardiolipin in eukaryotes is attained by re-modelling on the matrix side of the mitochondrial inner membrane. This can be achieved in part by the coenzyme A (CoA)-dependent deacylation-reacylation cycle known as the 'Lands cycle' (after W.E.M. (Bill) Lands - see our web page on phosphatidylcholine), but the main mechanism is unique to cardiolipin. This makes use of a CoA-independent transacylation between different phospholipids in which an enzyme termed 'tafazzin' (TAZ), a phospholipid-lysophospholipid acyltransferase anchored to the membrane via a hydrophobic segment, plays a major part by converting monolysocardiolipin into cardiolipin and vice versa. Remodelling is necessary as the precursor phospholipid is very different in composition from the symmetrical molecules apparently required in the final product if it is to function correctly. There is evidence that the process is triggered by the assembly of the complexes of oxidative phosphorylation in mitochondria. While most research has dealt with heart mitochondria, most tissues need the ready availability of linoleate from other phospholipids.
 In animals, in the first step in remodelling,
a single acyl chain only is removed from newly synthesised cardiolipin with formation of monolysocardiolipin by a calcium-independent
phospholipase A2, of which iPLA2β appears most active (iPLA2γ cleaves oxidized fatty acids
from cardiolipin and is needed for bioenergetic purposes).
In yeast, the deacylation step of cardiolipin remodelling is catalysed by the lipase Cld1, and in higher eukaryotes, an enzyme ABHD18 is
now known to replicate all effects of Cld1 on cardiolipin metabolism, although rather than removing just one fatty acid, ABHD18 can deacylate
cardiolipin further, a function that is separate from remodelling and might lead to its complete breakdown.
In animals, in the first step in remodelling,
a single acyl chain only is removed from newly synthesised cardiolipin with formation of monolysocardiolipin by a calcium-independent
phospholipase A2, of which iPLA2β appears most active (iPLA2γ cleaves oxidized fatty acids
from cardiolipin and is needed for bioenergetic purposes).
In yeast, the deacylation step of cardiolipin remodelling is catalysed by the lipase Cld1, and in higher eukaryotes, an enzyme ABHD18 is
now known to replicate all effects of Cld1 on cardiolipin metabolism, although rather than removing just one fatty acid, ABHD18 can deacylate
cardiolipin further, a function that is separate from remodelling and might lead to its complete breakdown.
In contrast, tafazzin protects cardiolipin from such degradation. In heart and yeast mitochondria, tafazzin is known to transfer acyl groups bidirectionally from phosphatidylcholine to monolysocardiolipin, the latter lacking one of the four acyl groups, and the continuing reactions promote molecular symmetry among the molecular species of fully acylated cardiolipin with the appearance of a specificity for linoleate groups (and with lysophosphatidylcholine as an intermediate). The cyclic mechanism does not require coenzyme A esters of fatty acids, it is reversible, and only a trace level of either lysophosphatidylcholine or monolysocardiolipin need be present.
From a mathematical model based on the assumption that different molecular species have different free energies, it has been concluded that the characteristic acyl distributions in cardiolipin could arise from phospholipid transacylations in the tafazzin domain, even if tafazzin itself does not have substrate specificity. The suggestion is that sufficient energy differences arise from the packing properties of the entire lipid assembly in the membrane to enable tafazzin to catalyse the remodelling of cardiolipin by combinations of forward and reverse transacylations and create an equilibrium distribution of acyl groups. As the shape of tetralinoleoyl-cardiolipin is such that it fits the geometry of negatively curved monolayers particularly well, this structure is favoured. Alternatively, it has been suggested that the membrane environment might be a source of specificity in the tafazzin-catalysed reaction or that the latter could be controlled by the pool of phospholipid acyl chains in a tissue. It has been established that plasmenylcholine, which is abundant in linoleate in heart mitochondria, is a substrate for tafazzin and may be needed for remodelling cardiolipin.
This may be the first CoA-independent phospholipid transacylase to have been identified, and the reaction is quite different from the cycle of acylation and deacylation for remodelling of the more conventional phospholipids. Unlike the latter in which only position sn-2 is modified, all four positions in cardiolipin are affected.
In an alternative remodelling process via the Lands cycle, at least two acyltransferases can reacylate monolysocardiolipin in mammalian cells, including acyl-CoA:lysocardiolipin acyltransferase (ALCAT1) and monolysocardiolipin acyltransferase (MLCLAT1). ALCAT1 is an acyl-CoA-dependent acyltransferase that is located on to the mitochondria-associated membrane (MAM), a sub-compartment of the endoplasmic reticulum, and transfers acyl groups from linoleoyl-CoA or oleoyl-CoA preferentially to phospholipids that are biosynthetic intermediates for cardiolipin, such as monolysocardiolipin, dilysocardiolipin and phosphatidylglycerol. Similarly, MLCLAT1-catalysed acylation of monolysocardiolipin has a higher affinity for linoleoyl-CoA than for other coA esters. A third enzyme related to MLCLAT1, the alpha subunit of trifunctional protein (αTFP), may be part of the process.
In yeast, tafazzin is a non-integral membrane protein located at the outer leaflet of the inner membrane and the inner leaflet of the outer membrane, so cardiolipin must be translocated to its final destination by means of one of three enzymes, a phospholipid scramblase (PLS3), mitochondrial creatine kinase or nucleoside diphosphate kinase, probably requiring protein aggregation to promote membrane domains. In S. cerevisiae, which does not synthesise polyunsaturated fatty acids, a single phospholipase A2 deacylates cardiolipin before tafazzin transfers oleate from mitochondrial phosphatidylcholine to form tetraoleoyl cardiolipin. As some remodelling of cardiolipin occurs in the that region of the endoplasmic reticulum closely associated with mitochondria and as cardiolipin is a significant component of peroxisomal membranes (7% in yeast), some intracellular transport mechanism must exist that probably involves membrane vesicles derived from mitochondria.
In addition to the biogenesis of new cardiolipin, this remodelling process may be utilized to repair oxidatively damaged cardiolipin or to change the molecular species of existing cardiolipin to suit some mitochondrial purpose. For example, cardiolipin remodelling is essential for the differentiation of myoblasts (myogenesis) in skeletal muscle into mature cells. Improper cardiolipin processing because of mutations or deficiencies in tafazzin and other relevant enzymes can lead to mitochondrial dysfunction and can contribute significantly to cardiovascular and other diseases. It should be noted that the fatty acid and molecular species compositions of cardiolipin in tissues other than heart can be very different and may need enzymes with differing specificities.
Catabolism: The half-life of cardiolipin is several-fold longer than that of other phospholipids, but catabolism can occur by the action of phospholipase A2 (or ABHD18) to remove fatty acyl groups, possibly after peroxidation as part of the process of apoptosis (see below). There is also a mitochondrial phospholipase D, which hydrolyses cardiolipin to phosphatidic acid (and phosphatidylglycerol) and in so doing promotes the fusion of mitochondria, a reaction that may be highly relevant under conditions of oxidative stress; the cardiolipin phospholipase HSD10 mediates the degradation of the oxidized lipid. With all phospholipases, the rate of hydrolysis is significantly higher in the 3'‑phosphatidyl moiety.
3. Function
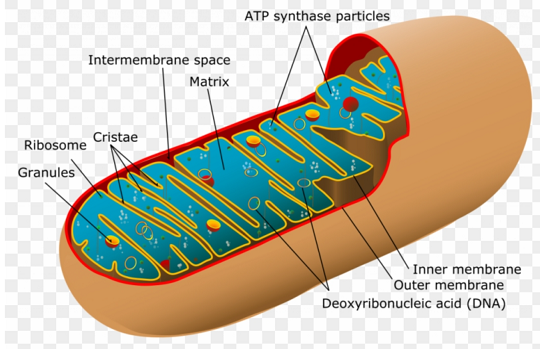 |
| Schematic mitochondrion - courtesy of Clipartmax |
As cardiolipin is the characteristic lipid component of mitochondria, its function in this organelle is clearly crucial in all tissues and organisms. Mitochondria have unique double bilayer membranes, the outer and inner mitochondrial membranes, and these enclose two aqueous compartments, the intermembrane space and the matrix. Cardiolipin is located mainly on the inner of these membranes, where it interacts with many integral membrane proteins in a manner that is determined by its physical and structural properties, but it also regulates endoplasmic reticulum-mitochondrial contact sites. In summary, it is involved in the biogenesis, morphology and stability of mitochondrial cristae, in mitochondrial quality control and dynamics through fission and fusion, and in innumerable aspects of mitochondrial metabolism. Bioenergetic processes, such as the tricarboxylic acid cycle and β‑oxidation of fatty acids, are performed within the matrix.
Protein binding: Cardiolipin has a potent binding capacity for many structurally unrelated proteins, so its structure must be adapted to differing protein surfaces. Although no characteristic sequence motif has been identified in proteins, there are three key regions: (i) phosphate binding patches (enriched in basic amino acids, e.g., Arg, His and Lys), (ii) hydroxyl binding patches (amino acids with mixed aromatic/polar characters, e.g., Trp and Tyr), and (iii) acyl binding patches (large hydrophobic amino acids, e.g., Ile, Leu and Val). In general, the head group of cardiolipin interacts strongly with certain amino acid residues via electrostatic forces and hydrogen bonds and with water molecules, while the acyl chains retain their flexibility and interact through van der Waals forces with several protein surface sites. Among many proteins known to bind in this way, there are the haemoprotein, cytochrome c, the cell permeable peptide, elamipretide, snake venom cardiotoxins, α-synuclein, the ABC transporter ABCB10, and the plasma protein β2 glycoprotein I.
As discussed above, cardiolipin has a role in the lateral organization of mitochondrial membranes by promoting clusters and non-bilayer structures, while the charge on the head group may facilitate conformational changes to proteins with structural and functional consequences and can thus may modulate the activity of some membrane proteins. Although we may not yet know why the characteristic fatty acid and molecular compositions are necessary, modifications of these compositions by dietary means can have consequences for mitochondrial efficiency.
Oxidative phosphorylation: The interactions between cardiolipin and enzymes have been studied intensively for the cytochrome bc1 complex of the respiratory chain, which couples electron transfer between ubiquinol and cytochrome c to the translocation of protons across the lipid bilayer. In this instance, one cardiolipin molecule is bound close to the site of ubiquinone reduction where it ensures the stability of the catalytic site as well as taking part in proton uptake. Such interactions with cardiolipin are required to stimulate many enzymes, but especially those for oxidative phosphorylation and photo-phosphorylation, which result in ATP production.
In animals, the respiratory chain consists of four enzymes (NADH dehydrogenase, succinate dehydrogenase, cytochrome bc1 complex and cytochrome c oxidase), which are organized in large complexes, designated complexes I to IV and constituting a supramolecular network or 'respirasome'. Cardiolipin has been described as a "glue” that binds components of the mitochondrial respiratory chain into an integrated system to provide efficient transfer of electrons and protons. For example, cardiolipin has been identified as an integral component in crystals of mitochondrial complex III, complex IV where it may have a role in proton translocation, complex I and possibly complex II, and the ADP-ATP-carrier; it is known to be necessary for the stability of the quaternary protein structure of complex IV. In the respiratory chain, these assemble into large super-complexes, in which complex I binds a dimer of complex III and several copies of complex IV and together may incorporate as many as 200 cardiolipin molecules. In yeast, the related enzymes are part of a single functional unit, components of which contain tightly bound cardiolipin. This is an essential constituent of the interface between the complex and its membrane environment or between subunits within the complex, and removal of cardiolipin leads to break-up of the complex and loss of functionality. When linoleate is displaced experimentally by docosahexaenoate in mitochondrial cardiolipin in the rat, the activities of the respiratory complexes are greatly reduced, apparently by preventing the formation of phospholipid domains that are regulatory.
| Mitochondrial electron transport chain (Image courtesy of Fvasconcellos and Wikipedia). |
Cardiolipin is critical for the degree of oligomerization and the degree of order in ATP synthase assemblies. It has further links to energy metabolism in that it is required for optimal stimulation of the enzyme pyruvate dehydrogenase and thence to the generation of acetyl-CoA and the operation of the tricarboxylic acid cycle, while there is a requirement for cardiolipin for the stability and activity of the mitochondrial calcium uniporter, which controls calcium uptake by mitochondria and coordinates cytosolic signalling events with mitochondrial bioenergetics.
Three molecules of cardiolipin are tightly bound to uncoupling protein 1 (UCP1), an integral membrane protein found in the mitochondrial inner membrane of brown adipose tissue that is crucial for thermogenesis. Also, in relation to energy metabolism, cardiolipin anchors two kinases, mitochondrial creatine kinase and nucleoside diphosphate kinase, to the inner and possibly the outer mitochondrial membranes where they come in contact. Together with the ADP-ATP- and phosphate-carrier proteins, it facilitates the transport of solutes between the intra-membrane and matrix spaces of mitochondria.
Mitophagy and apoptosis: Cardiolipin has been implicated in the processes of mitophagy (mitochondrial degradation) and of apoptosis (programmed cell death) in animal cells through its interactions with a variety of death-inducing proteins. When mitophagy is warranted, cardiolipin is externalized by crossing the inner mitochondrial membrane, the intermembrane space and then the outer mitochondrial membrane in a process mediated by nucleoside diphosphokinase (NDPK-D) and/or phospholipid scramblase 3. Once externalized, it binds to microtubule-associated protein 1 light chain 3, which is part of the autophagy machinery, and acts as a signal for the process. Cardiolipin exposed in this way in mitochondria acts in a pro-inflammatory manner as a platform for inflammasome localization and stimulation.
When the metabolism of the cells is impaired by a continued accumulation of mitochondrial fragments, it may be necessary to initiate the elimination of the entire cell by a process of apoptosis, and this can involve externalization of oxidized cardiolipin as a signalling molecule involved in innate immunity as a damage-associated molecular patterns (DAMP). Oxidation of the fatty acyl chains is catalysed mainly by cardiolipin-bound cytochrome c acting as a peroxidase after the enzyme undergoes a structural reconfiguration that changes its function from that in the respiratory complex. Peroxidized cardiolipin breaks one of the coordination bonds in cytochrome c, transforming it into a peroxidase, and so establishes a "vicious cycle” of further cardiolipin oxidation in which this enzyme reacts only with cardiolipin, but not with other more abundant phospholipids, causing oxidation, loss of molecular symmetry and then hydrolysis of the product cardiolipin hydroperoxides. In consequence, the cytochrome c is released into the intermembrane space, while the oxidized cardiolipin is translocated to the outer mitochondrial membrane, and with a synergistic effect of Ca2+ ions, it participates in the formation of the mitochondrial permeability transition pore that facilitates egress of pro-apoptopic factors from mitochondria into the cytosol where they triggers apoptosis (see our web page on oxidized phospholipids). Only those cardiolipins containing mono-oxygenated linoleate have been identified as predictive biomarkers of apoptosis.
 During this process,
cardiolipin has a role in the anchoring, translocation and embedding of caspases, a family of protease enzymes necessary for apoptosis, in the
mitochondrial membrane and thereby causes further release of apoptotic factors
into the cytosol to influence inflammation and the immune response.
As a result, the cellular concentration of cardiolipin decreases rapidly while some monolyso-cardiolipin may accumulate.
Although the mechanisms are very different, there are parallels with how phosphatidylserine
trigger apoptosis.
During this process,
cardiolipin has a role in the anchoring, translocation and embedding of caspases, a family of protease enzymes necessary for apoptosis, in the
mitochondrial membrane and thereby causes further release of apoptotic factors
into the cytosol to influence inflammation and the immune response.
As a result, the cellular concentration of cardiolipin decreases rapidly while some monolyso-cardiolipin may accumulate.
Although the mechanisms are very different, there are parallels with how phosphatidylserine
trigger apoptosis.
In contrast, there is a school of thought that the initial oxidation of a cardiolipin molecule triggers a chain of anti-apoptotic reactions, which hinder generation of reactive oxygen species and further oxidation of cardiolipin in the entire mitochondrion. It is only when oxidation is more advanced that the pro-apoptotic reactions take over.
Cytochrome c and cardiolipin oxidized in this way are responsible for the oxidative cleavage of plasmalogens (see our web page on ether lipids) with potential implications for eicosanoid signalling, and for the pathologies of ischemic tissue damage, stroke, chronic inflammation, aging, and age-related degenerative diseases such as Alzheimer's. There tends to be a reduction in the relative concentration of tetralinoleoyl cardiolipin with aging, which may lead to mitochondrial dysfunction, although cytoplasmic droplets within mitochondria can be induced to release linoleate for cardiolipin synthesis in these circumstances.
Other metabolic effects: Many other instances of the participation of cardiolipin in the crystal structures and thence the functions of proteins have been documented. In animal tissues, cardiolipin may be a cofactor for cholesterol translocation from the outer to the inner mitochondrial membrane, and in steroidogenic tissues, it induces mitochondrial cholesterol side chain cleavage and is a potent stimulator of steroidogenesis. Cardiolipin may take part in the import of proteins into mitochondria, and it can behave as a molecular chaperone to promote folding of mitochondrial proteins. It binds to the DNA in eukaryotic chromatin (the material of which chromosomes are composed), and indeed all of this lipid in chromatin is bound to DNA, where both have a common 'interphosphate' structural motif, and so it may regulate gene expression. Synthesis of cardiolipin de novo is required for activation of T cells, and those with a deficiency fail to respond to pathogens and are not able to adapt to nutrient stress.
Plants and bacteria: In higher plants and photosynthetic bacteria, cardiolipin is crucial for maintaining mitochondrial ultrastructure and for stabilizing the respiratory complex I/complex III super-complexes, and it is an integral constituent of the photosystem II complexes, which are responsible for oxidative processes, while facilitating adaptation to environmental stresses. It enables mitochondrial fission through stabilization of fission-related protein complexes. In eubacteria, it has a role in oxidative phosphorylation, but can be replaced by other phospholipids in selected mutants at least.
Cardiolipin is an integral component of several prokaryotic enzyme systems, including those for cell division, energy metabolism and membrane transport. Its propensity to form non-bilayer structures enables it to participate in dynamic protein-lipid membrane domains of higher curvature such as those where bacteria divide. The cytoplasmic membranes of bacteria are thought to contain micro-domains of cardiolipin (and of phosphatidylethanolamine), which assemble spontaneously because of the intrinsic physical properties of the lipid and seem be located where there is intense phospholipid biosynthesis so may be relevant to other cellular processes, including cell division and sporulation. In E. coli cells depleted of phosphatidylethanolamine, the cardiolipin synthase flips its catalytic globular domain to supply cardiolipin to the periplasmic leaflet of the inner membrane. The distinctive physical properties of the lipid are essential for osmoregulation here, where it regulates the osmosensory proline transporter, and it aids the transportation of lipopolysaccharide across the cell wall of to maintain the barrier to the environment.
With some bacterial pathogens, free sphingosine is reported be bactericidal by binding of the protonated NH2 group to the negatively charged head-group of cardiolipin in their plasma membranes, thereby promoting rapid permeabilization of the membrane and bacterial cell death.
4. Cardiolipin in Disease
Barth syndrome: This is a human disease state (an infantile cardiomyopathy) linked to the X-chromosome that has been associated with marked abnormalities in remodelling and thence in the composition of cardiolipin, i.e., a decrease in tetra-linoleoyl species and an accumulation of monolysocardiolipin; determination of the relative amounts of these lipids in blood aids diagnosis of the disease. The metabolic defect is now known to be due to mutations in the tafazzin gene that lead to cardiolipin deficiency and the other lipid changes. It has been demonstrated that monolysocardiolipin binds to the proteins of oxidative phosphorylation with a much lower affinity than for cardiolipin per se, and in consequence, there are reductions in the efficiency of oxidative phosphorylation in mitochondria because of abnormalities in the expression, assembly and function of Complex I of the mitochondrial respiratory chain. An increase in the permeability of the mitochondrial membranes results in further metabolic responses that include the enzymes of the tricarboxylic acid cycle, together with morphological changes to the inner mitochondrial membrane that are ultimately manifested in patients as skeletal muscle weakness and dilated cardiomyopathy. Some comparable biochemical phenomena have been observed in yeast mutants that lack the tafazzin equivalent.
The leading hypothesis is that monolysocardiolipin accumulating in this way is in a complex with cytochrome c that acts as a peroxidase to oxidize polyunsaturated fatty acids in phospholipids. A mitochondria-targeted anti-peroxidase agent was found to inhibit this reaction and improve mitochondrial respiration in a Drosophila melanogaster model of the disease, and it is hoped that this offers a potential therapeutic approach.
Reduced cardiolipin levels may cause instability in the mitochondrial calcium uniporter and may be another relevant factor. In addition, a marked decrease in the plasmalogen plasmenylcholine with a high content of linoleate has been identified in Barth syndrome, and restoring this is reported to be beneficial towards cardiolipin concentrations in an experimental model in vitro. Clinical trials with elamipretide, a mitochondrial tetrapeptide that interacts with cardiolipin, have shown promise in alleviating some of the symptoms of the syndrome.
Other diseases: Reductions in the concentrations of cardiolipin or changes in its composition in heart mitochondria have been implicated in many other human diseases states, including heart failure, diabetes and cancer, although it is not clear whether these effects are symptoms or the cause. Although oxidation of cardiolipin is part of the normal process of apoptosis, there is evidence that the proximity of this lipid to highly reactive oxygen species can lead to excessive peroxidation and oxidative stress, especially in the ischemic heart and skeletal muscle or during aging. At the onset of diabetes, there is a dramatic decline in the content of myocardial cardiolipin, and this is accompanied by extensive remodelling to yield molecules enriched in docosahexaenoic acid, which is highly susceptible to oxidation. The enzyme acyl-CoA:lysocardiolipin acyltransferase 1 (ALCAT1), an acyltransferase upregulated by oxidative stress, catalyses this remodelling and promotes lipid hydroperoxide formation that leads to a vicious cycle of oxidative stress, cardiolipin depletion and mitochondrial dysfunction in age-related diseases.
Malfunctions of cardiolipin metabolism in brain mitochondria have been implicated in neurodegenerative diseases, including Alzheimer’s and Parkinson’s diseases, and with the latter, oligomers of the α-synuclein protein bind to cardiolipin in mitochondrial membranes to disrupt their integrity and impair their capacity to operate effectively. After cardiac arrest, brain-type cardiolipins enriched in polyunsaturated fatty acids appear in plasma and may serve as a marker of mitochondrial/tissue injury in adult brain. Similar findings have been obtained in studies of traumatic brain injury.
The presence of antibodies to cardiolipin in plasma of patients with various diseases in which tissue damage occurs can be a danger signal for the immune system. T cells responsive to cardiolipin or oxidized cardiolipin may serve in immune surveillance during infection and tissue injury, while antibodies to cardiolipin are used in diagnostic tests after unexplained venous or arterial thrombotic episodes or recurrent miscarriages. In macrophages in vitro at least, it has been demonstrated that unsaturated cardiolipins compete with lipopolysaccharides for binding to Toll-like receptor 4 (TLR4) and prevent its activation, while saturated cardiolipins are TLR4 agonists and induce a pro-inflammatory response.
The bacteria responsible for syphilis release cardiolipin from mitochondria in their hosts, and this is believed to be the main factor that contributes to the production of anti-cardiolipin antibodies in the disease for which cardiolipin is widely used as an antigen in tests. It was this property that led to the first isolation of cardiolipin by Mary Pangborn in 1942, followed by the establishment of its structure by LeCocq and Ballou in 1964.
5. Related Lipids
Animals and higher plants contain only cardiolipin per se, but structural analogues, such as phosphatidylglycerophosphoglycerol, D‑glucopyranosylcardiolipin, D-alanylcardiolipin, L-lysylcardiolipin and the phosphatidylglycerol acetal of plasmenyl diphosphatidylglycerol, have been found in both Gram-positive and Gram-negative bacteria, and for example, lysyl-cardiolipin is a major constituent of the membranes of Listeria where it can represent up to 30% of the total phospholipids. In these lipids, which are obviously related to the complex lipoamino acids derived from phosphatidylglycerol, the alanyl, lysyl and glucosyl residues are linked to the 2'-hydroxyl on the central glycerol moiety. They are synthesised by analogous enzymes and have comparable functions. Some of the Archaea contain true cardiolipin analogues.
 |
| Figure 3. Unusual cardiolipins or cardiolipin analogues from bacteria. |
In the glucosylcardiolipin from Geobacillus stearothermophilus in which glucose is linked to the free hydroxyl of the central glycerol moiety (molecules with 1 to 5 glucose molecules are known), the main fatty acids present have iso-and anteiso-methyl branches (14:0 to 19:0), while fatty acids with a terminal cyclohexyl group are the main components in the lipid from the thermophilic bacterium Alicyclobacillus acidoterrestris, which also contains an O-acyl glycosylated cardiolipin.
In Halobacterium salinarum (Archaea), osmotic shock induces synthesis of an even more complex lipid consisting of sulfo-triglycosyl-diether esterified to the phosphate group of phosphatidic acid, which has been termed 'glycocardiolipin', although the use of the term ‘cardiolipin’ in this instance seems something of a misnomer as a glucose unit takes the place of the central glycerol. Morganella morganii in the human gut microbiome contains an unusual phospholipid that resemble conventional cardiolipins but with diethanolamine replacing the central glycerol; palmitic acid is the main component in position 1 of the glycerol moiety and position 2 is occupied by monoenoic and cyclopropanoid fatty acids. The presence of this organism has been correlated with and the incidence of major depressive disorder in the human host.
6. Analysis
Cardiolipin elutes close to phosphatidylglycerol and phosphatidic acid in many chromatographic systems, but it can be resolved with care. Modern liquid chromatography-mass spectrometry and 'shotgun lipidomics' techniques are proving to be sensitive, but as such methods do not distinguish between the two chiral glycerol moieties, detailed analyses of molecular species and of the positional distributions of the fatty acid components are still difficult problems technically. A method that utilizes high-performance liquid chromatography linked to electrospray mass spectrometry has been developed to separate cardiolipin and monolysocardiolipin, while determining the composition of each, as an aid to the diagnosis of Barth syndrome.
Recommended Reading
- Carney, O.S. and others. A review of disorders of cardiolipin metabolism: Pathophysiology, clinical presentation and future directions. Mol. Gen. Metab., 145, 109184 (2025); DOI.
- Chai, Z.D. and others. Cardiolipin's multifaceted role in immune response: a focus on interacting proteins. Front. Immun., 16, 1680326 (2025); DOI.
- Christie, W.W. and Han, X. Lipid Analysis - Isolation, Separation, Identification and Lipidomic Analysis (4th edition), 446 pages (Oily Press, Woodhead Publishing and now Elsevier) (2010) - see Science Direct.
- Dong, C., Lv, D.P., Dong, Y.Y., Zhang, Z.H., Li, Q.C. and Chen, Z. Advances in cardiolipin analysis: applications in central nervous system disorders and nutrition interventions. Biomolecules, 16, 71 (2026); DOI.
- Dowhan, W. and Bogdanov, M. Eugene P. Kennedy's legacy: defining bacterial phospholipid pathways and function. Front. Mol. Biosci., 8, 666203 (2021); DOI.
- Fox, C.A. and Ryan, R.O. Studies of the cardiolipin interactome. Prog. Lipid Res., 88, 101195 (2022); DOI.
- Fuentes, J.M. and Morcillo, P. The role of cardiolipin in mitochondrial function and neurodegenerative diseases. Cells, 13, 609 (2024); DOI.
- Jiang, Z.T., Shen, T., Huynh, H., Fang, X., Han, Z. and Ouyang, K.F. Cardiolipin regulates mitochondrial ultrastructure and function in mammalian cells. Genes, 13, 1889 (2022); DOI.
- Kagan, V.E. and others. Anomalous peroxidase activity of cytochrome c is the primary pathogenic target in Barth syndrome. Nature Metab., 5, 2184-2205 (2023); DOI.
- Li, Z.Z., Xiao, H.X., Hu, J.J., Xie, W., Wang, Z.X., Pan, Y.P., Li, X.H. and Yu, X.F. The mechanisms and implications of cardiolipin in the regulation of cell death. Cell Biochem. Function, 43, e70066 (2025); DOI.
- Luévano-Martínez, L.A. and Duncan, A.L. Origin and diversification of the cardiolipin biosynthetic pathway in the Eukarya domain. Biochem. Soc. Trans., 48, 1035-1046 (2020); DOI.
- Maguire, J.J. and others. Known unknowns of cardiolipin signaling: The best is yet to come. Biochim. Biophys. Acta, Lipids, 1862, 8-24 (2017); DOI - and other articles in this special journal issue on "Lipids of Mitochondria".
- Pennington, E.R., Funai, K., Brown, D.A. and Shaikh, S.R. The role of cardiolipin concentration and acyl chain composition on mitochondrial inner membrane molecular organization and function. Biochim. Biophys. Acta, Lipids, 1864, 1039-1052 (2019); DOI.
- Shilovsky, G.A., Putyatina, T.S., Ashapkin, VV., Yamskova, O.V., Lyubetsky, V.A., Sorokina, E.V., Shram, S.I., Markov, A.V. and Vyssokikh, M.Y. Biological diversity and remodeling of cardiolipin in oxidative stress and age-related pathologies. Biochemistry (Moscow), 84, 1469-1483 (2019); DOI.
- Slavetinsky, C., Kuhn, S. and Peschel, A. Bacterial aminoacyl phospholipids – Biosynthesis and role in basic cellular processes and pathogenicity. Biochim. Biophys. Acta, Lipids, 1862, 1310-1318 (2017); DOI.
- Venkatraman, K., Lee, C.T. and Budin, I. Setting the curve: the biophysical properties of lipids in mitochondrial form and function. J. Lipid Res., 65, 100643 (2024); DOI.
- Xu, Y., Phoon, C.K.L., Ren, M.D. and Schlame, M. A simple mechanistic explanation for Barth syndrome and cardiolipin remodeling. J. Inher. Metab. Dis., 45, 51-59 (2022); DOI - and other articles in this special journal issue.
- Yoo, Y., Yeon, M., Yoon, M.S. and Seo, Y.K. Role of cardiolipin in skeletal muscle function and its therapeutic implications. Cell Commun. Signal., 23, 36 (2025); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: February 2026 | ||
© The LipidWeb is open access and fair use is encouraged - but not text and data mining, AI training, and similar technologies (text scraping).
