Protectins, Resolvins and Maresins -
Specialized Pro-Resolving Mediators
 Oxygenated metabolites or oxylipins with highly specific stereochemical structures derived mainly
from eicosapentaenoic acid (20:5(n-3) or EPA), docosapentaenoic acid (22:5(n-3) or DPA) and especially docosahexaenoic acid
(22:6(n-3) or DHA) and termed the “(neuro)protectins, resolvins and maresins” (or collectively the - “specialized
pro-resolving mediators”) are reported to have potent anti-inflammatory and immunoregulatory actions at concentrations in the nanomolar
to picomolar range.
Their structures have been conserved in evolution from diatoms to flatworms to fish to humans, and they are present in all major organ systems.
Oxygenated metabolites or oxylipins with highly specific stereochemical structures derived mainly
from eicosapentaenoic acid (20:5(n-3) or EPA), docosapentaenoic acid (22:5(n-3) or DPA) and especially docosahexaenoic acid
(22:6(n-3) or DHA) and termed the “(neuro)protectins, resolvins and maresins” (or collectively the - “specialized
pro-resolving mediators”) are reported to have potent anti-inflammatory and immunoregulatory actions at concentrations in the nanomolar
to picomolar range.
Their structures have been conserved in evolution from diatoms to flatworms to fish to humans, and they are present in all major organ systems.
They are produced by dioxygen-dependent oxidation from the omega-3 essential fatty acids (EFA), which are the focus of considerable interest among nutritionists because of the perceived benefits for the health of consumers. Although the mechanisms by which the benefits of EFA are achieved are not entirely clear and evidently involve a multiplicity of factors, it seems likely that their oxygenated metabolites could play a significant part. The therapeutic potential of these oxylipins is appreciable, and it is hoped that their biosynthesis can be modulated by dietary supplementation with their fatty acid precursors as well as by direct administration of individual compounds. As research on this topic continues to develop rapidly, this account cannot be comprehensive or definitive. A further important caveat is that there is currently a debate on whether natural levels of these metabolites are sufficient to support some published claims regarding their biological properties, which may have been determined in tissue preparations in vitro.
Simple mono-oxygenated lipoxygenase and cytochrome P450 metabolites of EPA and DHA, as well as non-enzymic isoprostane analogues (neuroprostanes), which have related properties, are discussed elsewhere on this site for reasons of practical convenience. Oxylipins that may be produced by related dioxygenation reactions of arachidonic acid and other fatty acids of the omega-6 family are described in our web page on leukotrienes.
1. Definitions and Key Features
The most studied of the protectins and related oxylipins are derived from DHA and are dihydroxylated E,E,Z-docosatrienes, i.e., acyclic lipoxygenase metabolites containing a conjugated triene unit with an E,E,Z‑configuration and flanked by two secondary allylic alcohols (together with other non-conjugated cis-double bonds). The nomenclature can be confusing because it has changed as the science has developed, but three novel mediator super-families derived from EPA and DHA are now recognized. The trivial name 'neuroprotectin' was coined by Professor Charles N. Serhan and colleagues for one class of these molecules as they were first found in neuronal tissue, though the term 'protectin' was later preferred when it was realized that they occur in many more animal organs. Other oxygenated derivatives of DHA, DPA and EPA were named 'resolvins' or 'resolution-phase interaction products', because these compounds were first encountered in resolving inflammatory exudates (pus). Subsequently, a third group of oxylipins with comparable structural features but formed by different enzymatic routes were characterized and termed 'maresins'.
As the range of lipid mediators expanded with continuing research, they were collectively termed ‘Specialized Pro-resolving Mediators’ or ‘SPMs’ because of their intimate involvement in the resolution of inflammation, as discussed below. Oxylipins derived from EPA are designated as SPMs of the E series, while those formed from the precursors DHA and DPA are denoted as SPMs of the D series.
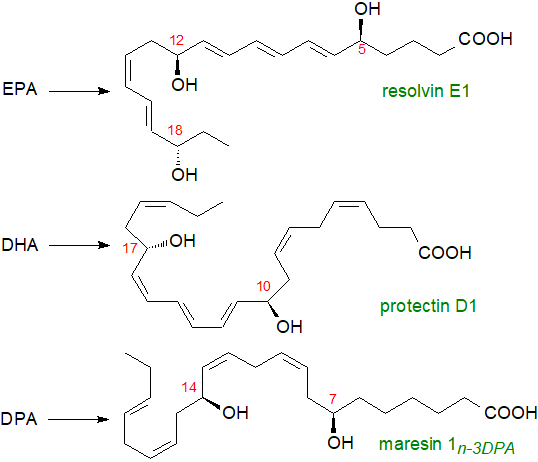 |
| Figure 1. Structures of representative SPMs and their precursors. |
The lipoxins, derived from arachidonic acid of the omega-6 family of polyunsaturated fatty acids, have long been considered to be a fourth class of SPMs and indeed were the first to be discovered, but this now appears to be in doubt as they cannot be detected in vivo and no receptor has yet been identified. They are discussed on this site in the web page dealing with the leukotrienes to which they are structurally and biosynthetically related.
It was long thought that resolution of inflammation was a passive process, occurring simply by the dilution of inflammatory signals from the site of injury or infection, but a concept has now emerged in which acute inflammation is initiated in animal tissues by prostaglandins and leukotrienes, before there is a switch in metabolism to the production of SPMs that end the inflammation. The acute inflammatory response is a coordinated process, and both initiation and resolution stages are required to maintain healthy tissues. SPMs are key factors in the latter "by limiting further neutrophil tissue infiltration and stimulating local immune cell (e.g., macrophage)-mediated clearance of apoptotic polymorphonuclear neutrophils, cellular debris, and microbes, as well as counter-regulating eicosanoid/cytokine production" (DOI). As uncontrolled or dysregulated inflammation is now widely recognized to be a unifying phenomenon in many chronic diseases, SPMs and derived molecules may have great therapeutic potential.
As with other oxylipins, the first step in the biosynthesis of the dihydroxylated metabolites of EPA or DHA is the release of the free acids from their link to phospholipids by the action of phospholipases. Thereafter, there are a number of common steps in the mechanism of oxygenation, starting with an antarafacial hydrogen abstraction at the C3 position in one of the cis,cis-1,4-diene moieties. Then, there is a stereo-selective insertion of molecular oxygen to form a carbon-oxygen bond in a hydroperoxide (Hp) intermediate, followed by a second hydrogen abstraction leading to an intramolecular nucleophilic attack by the oxygen atom in this intermediate to produce an epoxide. The final step is a hydrolase-assisted nucleophilic addition of water to a cis-double bond in the epoxide intermediate. Sequential actions of COX-2/aspirin, various lipoxygenases and CYP450 oxygenases are required as summarized in the following figure.
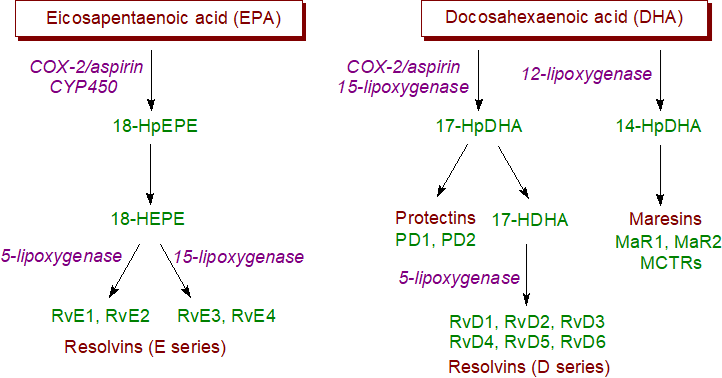 |
| Figure 2. Summary of the biosynthesis of some key SPMs. |
Although SPMs and SPM clusters were first found in bioactive amounts in human inflammatory exudates, they are now known to be present in most tissues and body fluids. Thus, many different molecular forms have been identified by lipidomic techniques in human peripheral blood and serum, lymph nodes, spleen, brain and breast milk, and analysis of these can potentially be indicative of disease status.
It should be recognized that establishing the precise stereochemistries of the double bonds and hydroxyl groups in SPMs, features that are essential for their functions in cells, has required heroic efforts involving total synthesis of large numbers of possible isomers for structural comparisons with those formed naturally.
2. Protectins (Neuroprotectins)
In studies of metabolite formation from DHA in brain tissue in response to aspirin treatment, it was shown that new docosanoids were produced that were initially termed ‘neuroprotectins’, although the simpler term ‘protectins’ is now preferred. Like the leukotrienes, there are three double bonds in conjugation (so they are sometimes termed E,E,Z‑docosatrienes) with six double bonds in total. For example, protectin D1 is present in murine inflammatory exudates and lung, in peripheral human blood and exhaled breath condensates, and in a wide range of cell types. The biosynthetic pathway to neuroprotectin NPD1 or protectin D1, as established in murine brain tissue and in human leukocytes and lymphocytes, is illustrated.
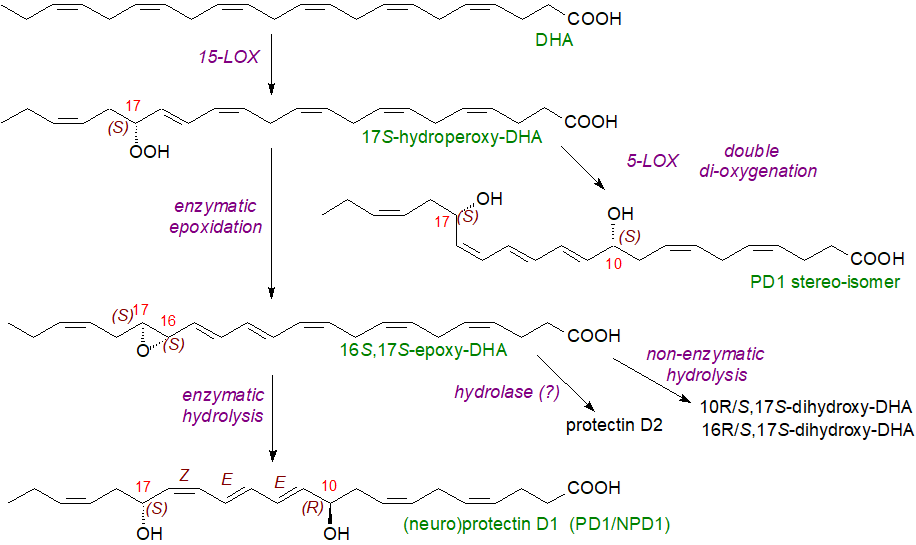 |
| Figure 3. Biosynthesis of protectins. |
Uniquely among SPMs, the initial product is 17S-hydroperoxy-DHA by reaction with 15-lipoxygenase (ALOX15), and this is converted first to a 16S,17S-epoxide and then to the 10,17‑dihydroxy-triene unit of 10R,17S-dihydroxy-docosa-4Z,7Z,11E,13E,15Z,19Z-hexaenoic acid, protectin D1 (PD1 or NPD1). Each step in the biosynthetic sequence is under precise stereochemical control by enzymes, and the structures of these DHA-derived mediators are highly conserved from fish to humans. Synthesis of PD1 is induced as a response to oxidative stress and/or activation of neurotrophins, and this highly stereospecific structure is necessary for its function.
Further oxygenation can occur, and 17S-hydroperoxy-DHA can react with 5-lipoxygenase to form the 10S,17S-dihydroxy metabolite in a double di‑oxygenation reaction, as illustrated, together with some of the 7,17-isomer. In the action of 5-LOX, the role of the accessory protein FLAP, which is required for leukotriene biosynthesis, has needed clarification, but it is now evident that it is necessary for the biosynthesis of most of the resolvins. The intermediate 16,17‑epoxy-docosatriene can undergo non-enzymatic hydrolysis to yield racemic 10R/S,17S-dihydroxy- and 16R/S,17S-dihydroxy-docosatrienes, but enzymatically controlled stereo- and regioselective nucleophilic addition of water at C16 by an as yet unidentified hydrolase produces protectin D2 (PD2) with 16S,17S-stereochemistry. Another metabolite 22-hydroxy-PD1 has been detected in human tissues and induces a significant response in cells in vitro.
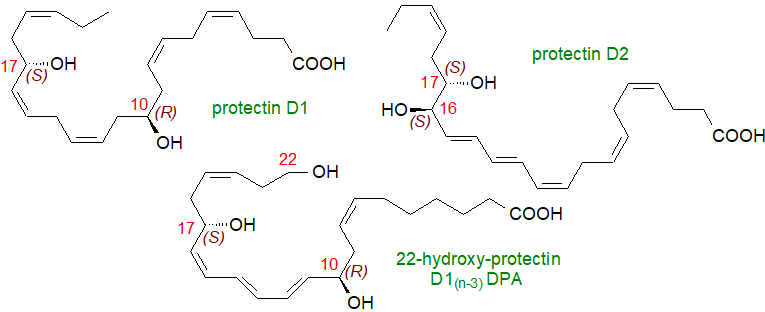
Both 22:5(n-3) and 22:5(n-6) fatty acids are good substrates for 15-lipoxygenase. Thus, 22:5(n-3) is first subjected to 17‑lipoxygenation and then via an epoxy intermediate to either 10,17-dihydroxy-7Z,11,13,15,19Z-docosapentaenoic acid (designated PD1n-3 DPA) or 16,17‑dihydroxy-7Z,10,13,14,19Z-docosapentaenoic acid (PD2n-3 DPA). 22:5(n-6) gives 17S-hydroxy-22:5(n‑6) and 10,17S-dihydroxy-22:5(n-6) as the main products. An omega-hydroxylated metabolite of PD1 (22‑OH‑PD1n‑3 DPA), produced by the action of a CYP1 monooxygenase, has been detected in human neutrophils and monocytes. All of these are potent anti-inflammatory agents. Sulfido-conjugates of protectins have been characterized and are discussed below.
PDX and poxytrins: Unfortunately, an isomer generated from DHA by the action of plant 15‑lipoxygenase
in a double di-oxygenation reaction was misidentified as PD1 and was made available commercially under that name,
leading to some erroneous reports in the literature.
This isomer (now designated ‘PDX’) differs in the geometry of double bonds in the conjugated triene, which is E,Z,E for PDX
and E,E,Z for PD1, and in the configuration of carbon 10, which is S in PDX and R in PD1.
On the other hand, PDX is now known to be a minor endogenous metabolite in murine peritonitis, and small amounts have been detected in
rat brain, both in free form and esterified to phospholipids.
Unfortunately, an isomer generated from DHA by the action of plant 15‑lipoxygenase
in a double di-oxygenation reaction was misidentified as PD1 and was made available commercially under that name,
leading to some erroneous reports in the literature.
This isomer (now designated ‘PDX’) differs in the geometry of double bonds in the conjugated triene, which is E,Z,E for PDX
and E,E,Z for PD1, and in the configuration of carbon 10, which is S in PDX and R in PD1.
On the other hand, PDX is now known to be a minor endogenous metabolite in murine peritonitis, and small amounts have been detected in
rat brain, both in free form and esterified to phospholipids.
Comparable oxylipins with the E,Z,E-conjugated triene motif between two secondary hydroxyl groups have been characterized that are related to leukotrienes (LTB4 and LTBX), and collectively, they have been named 'poxytrins' (PUFA oxygenated trienes). They are produced in animal tissues by sequential oxidation by 15-LOX and perhaps a second lipoxygenase, followed by reduction by cytosolic glutathione peroxidase.
Elovanoids: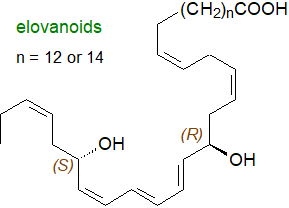 Very-long chain fatty acids 32:6(n-3) and 34:6(n-3) produced from DHA in brain and retina
by the action of elongases, mainly ELOV4, are converted to protectin analogues (hence the name
elovanoids) under conditions of oxidative stress, i.e., to
20R,27S-dihydroxydotriaconta-14Z,17Z,21E,23E,25Z,29Z-hexaenoic acid (ELV-N32) and
22R,29S-dihydroxytetratriaconta-16Z,19Z,23E,25E,27Z,31Z-hexaenoic
acid (ELV-N34).
They are low-abundance, high-potency, protective mediators that prevent retinal degeneration by enhancing the expression
of pro-survival proteins in cells subjected to oxidative stress.
In contrast to DHA per se, which is located in position sn-2 of phospholipids in these tissues, the precursors of the elovanoids
are located in position sn-1 with the implication that the stimulus for the release of the precursors and formation of the novel mediators
must be somewhat different from that of the protectins.
Very-long chain fatty acids 32:6(n-3) and 34:6(n-3) produced from DHA in brain and retina
by the action of elongases, mainly ELOV4, are converted to protectin analogues (hence the name
elovanoids) under conditions of oxidative stress, i.e., to
20R,27S-dihydroxydotriaconta-14Z,17Z,21E,23E,25Z,29Z-hexaenoic acid (ELV-N32) and
22R,29S-dihydroxytetratriaconta-16Z,19Z,23E,25E,27Z,31Z-hexaenoic
acid (ELV-N34).
They are low-abundance, high-potency, protective mediators that prevent retinal degeneration by enhancing the expression
of pro-survival proteins in cells subjected to oxidative stress.
In contrast to DHA per se, which is located in position sn-2 of phospholipids in these tissues, the precursors of the elovanoids
are located in position sn-1 with the implication that the stimulus for the release of the precursors and formation of the novel mediators
must be somewhat different from that of the protectins.
Aspirin-Triggered Protectins: A further route to protectin formation produces the 17R-epimer, i.e., 10R,17R-dihydroxy-docosa-4Z,7Z,11E,13E,15Z,19Z-hexaenoic acid and requires the intervention of aspirin (acetylsalicylic acid), the mode of action of which is described in our web page on prostaglandins. In brief, aspirin blocks the catalytic site of COX-1 by acetylating it irreversibly, but it can only partially block that of COX-2. The acetylated COX-2 acts in lipoxygenase-like manner comparable to 15‑LOX, but with the oxygen insertion in the R- rather than S‑configuration as is the case with lipoxygenases; the initial oxygenation reaction is not catalysed by unacetylated COX-2 or COX-1. Low-dose aspirin may jump start the resolution phase.
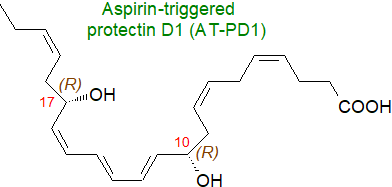
Production of 'aspirin-triggered' resolvins (see below) and then of protectins was first identified in resolving exudates and brain in mice and is now well established for human cells. Once more, the anti-inflammatory effects of the 10R,17R-isomer are dependent on the precise stereochemistry, as the 10S,17R‑dihydroxy-docosatriene is inert in vivo. Note that unlike aspirin, most non-steroidal anti-inflammatory drugs inhibit cyclooxygenases reversibly and can delay complete resolution.
Octadecanoids: Protectin-like metabolites can be produced from α-linolenic acid in animal tissues in vitro,
e.g., 9R,16S-dihydroxy-10E,12Z,14E-octadecatrienoic acid or linotrin 1, and
they are reported to occur naturally in plants.
While they are reported to have anti-inflammatory properties, they have not been detected in significant amounts in
vivo.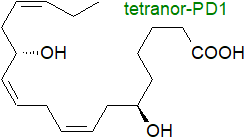
Catabolism: Protectin D1 is subjected to rapid β-oxidation from the carboxyl end of the molecule by cytochrome P450 enzymes with formation of 2,3‑dinor‑PD1 (C20) and 2,3,4,5‑tetranor-PDI (C18); this process may limit the concentration of PD1 in the systemic circulation and prevent its excretion in urine. In liver, these metabolites can be converted to glucuronide conjugates by specific uridine 5′‑diphospho-glucuronosyltransferase isoforms.
3. Resolvins
R-Resolvins (aspirin-triggered): Resolvins are produced from EPA, DPA and DHA by a trans-cellular mechanism, which is illustrated below for those derived from EPA. Biosynthesis of the 18R (illustrated) and 18S resolvins of the E series (name derived from EPA) has elements in common with the synthesis of the epi‑lipoxins, leukotrienes and of course the protectins.
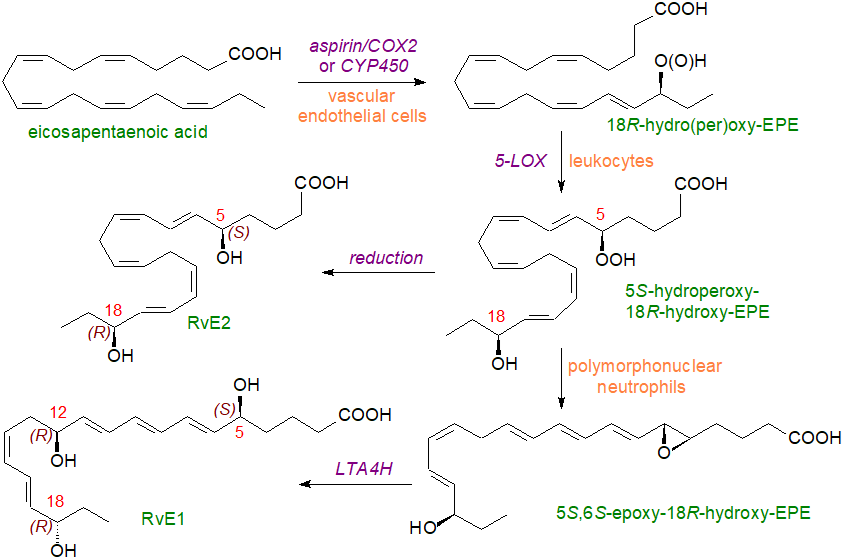 |
| Figure 4. Biosynthesis of the 18R-resolvins |
In vascular endothelial cells derived from blood vessels during inflammation, the cyclooxygenase enzyme COX-2 acetylated by aspirin introduces an 18R‑hydroperoxy group into the EPA molecule (see previous section); this can also be accomplished by a cytochrome P450 oxygenase. The product is reduced to the corresponding hydroxy compound and transferred to a neighbouring leukocyte before a 5S‑hydroperoxy group is introduced into the molecule by the action of 5‑lipoxygenase (ALOX5), as in the biosynthesis of the leukotrienes. A further reduction step produces 15S,18R‑dihydroxy-EPE or resolvin E2 (RvE2). Alternatively, the 5S‑hydroperoxy,18R-hydroxy-EPE intermediate is converted to a 5,6-epoxy fatty acid in polymorphonuclear neutrophils in humans and eventually to 5S,12R,18R-trihydroxy-6Z,8E,10E,14Z,16E-eicosapentaenoic acid or resolvin E1 (RvE1) by an enzyme required for the biosynthesis of leukotrienes in leukocytes, i.e., leukotriene A4 hydrolase (LTA4H).
There is a resolvin E3 (RvE3), 17R,18R-dihydroxy-5Z,8Z,11Z,13E,15E-EPE, which is synthesised by eosinophils via the 12/15‑LOX pathway, and perhaps surprisingly, this has been detected in marine microalgae together with 14S/R,17R,18R-trihydroxy-eicosatetraenoic acid. Resolvin E4 (RvE4), also synthesised by the 12/15‑LOX pathway, is 5S,15S-dihydroxy-6E,8Z,11Z,13E,17Z-eicosapentaenoic acid, and this can be subjected to omega-oxidation in human neutrophils to yield the 20-hydroxy metabolite.
DHA is converted to 17R-resolvins by an aspirin-triggered COX-2 mechanism like the previous (in the absence of aspirin, COX-2 in human microvascular endothelial cells converts DHA to 13S-hydroxy-DHA). Thence, enzymatic epoxidation generates either 7S,8‑epoxy or 4S,5‑epoxy intermediates, which are acted upon by 5‑LOX with the aid of the 5-LOX-activating protein (FLAP - see again our web page on leukotrienes) to yield the resolvins. The former synthesises the aspirin-triggered resolvins D1 and D2 illustrated, while the latter produces the aspirin-triggered resolvins D3 and D4 with all containing a 17R‑hydroxyl group. AT-RvD1 is 7S,8R,17R-trihydroxy-docosa-4Z,9E,119E,13Z,159E,19Z-hexaenoic acid.
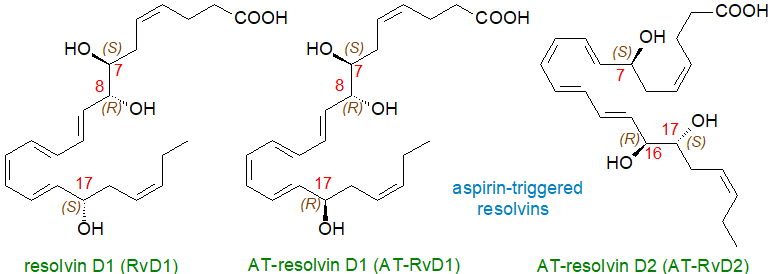 |
| Figure 5. Structures of resolvin D1 and aspirin-triggered resolvins D1 and D2. |
S-Resolvins: The highly specific stereochemistry of resolvin E1 is required for activation of a ligand-specific receptor and thence to enable it to act down to picomolar concentrations, but epimeric 18S-resolvins are produced in vivo by related biosynthetic pathways, with the first step catalysed by 15-LOX as for protectins, and these induce their own distinctive responses. Similarly, the precursor 18R- and 18S-hydroxy-EPEs have been shown to be anti-inflammatory in vitro.
In an alternative reaction in the absence of aspirin in human whole blood, isolated leukocytes and glial cells, 15-lipoxygenase (ALOX15) generates 17S‑hydroxy-DHA as the initial product. This is converted to 7S-hydroperoxy,17S-hydroxy-DHA by the action of a lipoxygenase, and thence via epoxy intermediates to resolvin (RvD1 or 7S,8R,17S‑trihydroxy-docosa-4Z,9E,11E,13Z,15E,19Z-hexaenoic acid) and epimeric resolvin D2 (RvD2 or 7S,16R,17S-trihydroxy-docosa-4Z,8E,10Z,12E,14E,19Z-hexaenoic acid), i.e., all contain a 17S‑hydroxyl group. An epimer of RvD2 (17R-RvD2) has been isolated from human macrophages, monocytes and saliva and is a potent proresolving mediator.
A further lipoxygenase-generated intermediate from 17S-hydroxy-DHA is transformed via an epoxide 4S,5S-epoxy-17S-hydroxy-DHA to resolvins D3 and D4 (4S,11R,17S- and 4S,5R,17S-trihydroxydocosahexaenoic acids, respectively) in human neutrophils, while M2 macrophages produce resolvin D4 and a cysteinyl-resolvin isomer (see below). 17R- and 17S‑hydroxy-DHA have anti-inflammatory properties of their own although they have generally been viewed simply as pathway markers and have been found in blood samples. Two further resolvins designated RvD5 and RvD6 (7S,14S- and 4S,17S‑dihydroxydocosahexaenoic acids, respectively), with a stereoisomer in the cornea termed R,R-RvD6, and other related oxylipins have now been characterized.
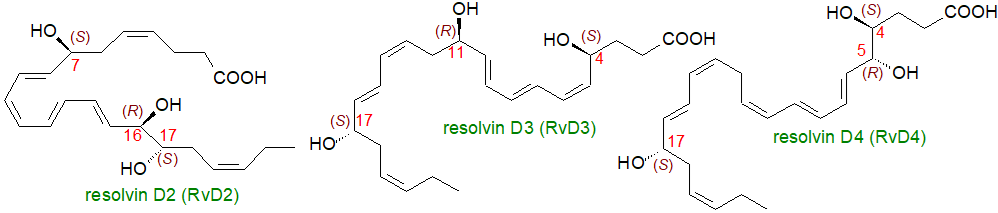 |
| Figure 6. Structures of resolvins D2, D3 and D4. |
Docosapentaenoic acid (22:5(n-3) or DPA) can be converted to resolvins, first by 17-lipoxygenation to 17-hydroperoxy-8Z,10Z,13Z,15E,19Z-docosapentaenoic acid followed by a 5-lipoxygenase-like reaction to yield three products, of which the most abundant is 7,8,17-trihydroxy-9,11,13,15E,19Z-docosapentaenoic acid (designated RvD1n-3 DPA). Four further metabolites of DPA have a hydroxyl group in position 13 and have been designated as 13‑series resolvins (RvTs), and RvT1 is 7,13,20-trihydroxy-8,10,14,16Z,18-docosapentaenoic acid. Biosynthesis of these molecules is initiated in the vascular endothelium when COX-2 converts docosapentaenoic acid to 13-hydroperoxy-7Z,10Z,14E,16Z,19Z-docosapentaenoic acid, which is rapidly hydrolysed to the 13R intermediate for donation to neutrophils where it is converted to RvT1. Sulfido-conjugates of resolvins are discussed below.
The marine diatom (microalgae) Cylindrotheca closterium produces appreciable amounts of SPMs, including RvE3 and novel structurally related eicosanoids derived from EPA, including 14S/R,17R,18R-trihydroxy-eicosatetraenoic acid, which are inflammation-resolving and anti-inflammatory in animals.
Catabolism: Resolvin E1 is eventually rendered inert in tissues by oxidation by the 15-hydroxyprostaglandin dehydrogenase common to the catabolism of many oxylipins, then proceeds via at least four pathways, including conversion to 18- and 12‑oxo‑RvE1 prior to further oxidation. RvD1 is deactivated by oxidation to 8- or 17-oxo-RvD1 as the first step in catabolism, while RvD4 is oxidized to 17‑oxo‑RvD4.
4. Maresins and Related Docosanoids
Alternative oxygenation reactions occur in human macrophages and platelets in which dihydroxy-mediators are formed from DHA termed maresins (‘macrophage mediator in resolving inflammation’) such as maresin 1 (MaR1) illustrated, i.e., 7R,14S‑dihydroxy-docosa-4Z,8E,10E,12Z,16Z,19Z-hexaenoic acid. Biosynthesis is initiated in macrophages by 14‑lipoxygenation of DHA by the action of human 12‑lipoxygenase (ALOX12 or ALOX15), producing the hydroperoxy intermediate 14S‑hydroperoxydocosa-4Z,7Z,10Z,12E,16Z,19Z-hexaenoic acid, which is subsequently converted to 13S,14S-epoxy-maresin. This undergoes enzymatic hydrolysis via an acid-catalysed nucleophilic attack by water at carbon-7, resulting in the introduction of a hydroxyl group at that position and double-bond rearrangement with the characteristic stereochemistry of maresin 1 (some of the required enzymes have yet to be identified).
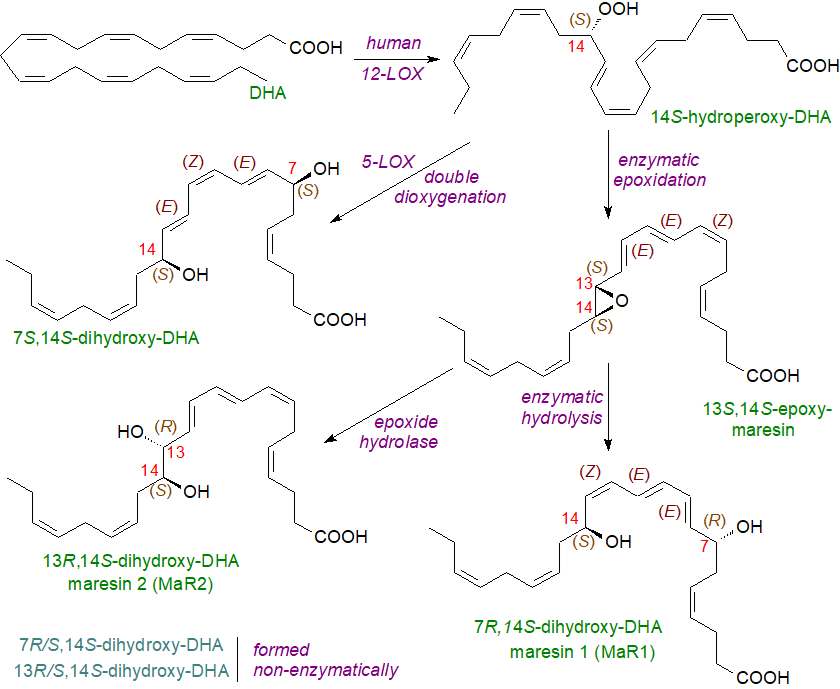 |
| Figure 7. Biosynthesis of maresin 1. |
13S,14S-Epoxy-maresin (13,14-eMar), which has activities of its own, is the precursor for 13R,14S‑dihydroxy-docosahexaenoic acid, designated maresin 2 (MaR2), via the action of the soluble epoxide hydrolase, and this metabolite is a potent anti-inflammatory and pro-resolving agent. MaR1 can be produced by circulating neutrophil–platelet aggregates in which DHA is converted to 13S,14S‑eMaR by the platelet 12‑lipoxygenase, before the intermediate is donated to neutrophils for the hydrolysis step. Analogous oxygenated compounds with anti-inflammatory properties are formed from 22:5(n‑3) and 22:5(n‑6) fatty acids with those derived from the former designated MaR2n‑3 DPA and MaR3n‑3 DPA. MaR1 has been found in primitive invertebrates suggesting that its structure and function have been conserved in evolution.
 Maresin-like di-oxygenated metabolites of DHA produced by sequential oxidation
by 12-LOX and enzymes of the cytochrome P450 family have been shown to occur in macrophages, i.e.,
14S,21S‑dihydroxy-docosa-4Z,7Z,10Z,12E,16Z,19Z-hexaenoic
acid and its epimers.
Their synthesis is induced by wounding, and they have been shown to promote the healing of wounds.
Similar 14,22‑dihydroxy-metabolites are synthesised by a related mechanism in leukocytes and platelets, and they induce comparable
responses.
14S,20R‑dihydroxy-DHA is produced by the action of 12/15‑lipoxygenase in eosinophils and has been detected
in inflammatory exudates, while 22-hydroxy-MaR1 is produced by neutrophils and 14‑oxo-MaR1 by macrophages in inflammation induced
by bacterial infection; 22‑hydroxy-MaR1 retains the actions of its parent.
Maresin-like di-oxygenated metabolites of DHA produced by sequential oxidation
by 12-LOX and enzymes of the cytochrome P450 family have been shown to occur in macrophages, i.e.,
14S,21S‑dihydroxy-docosa-4Z,7Z,10Z,12E,16Z,19Z-hexaenoic
acid and its epimers.
Their synthesis is induced by wounding, and they have been shown to promote the healing of wounds.
Similar 14,22‑dihydroxy-metabolites are synthesised by a related mechanism in leukocytes and platelets, and they induce comparable
responses.
14S,20R‑dihydroxy-DHA is produced by the action of 12/15‑lipoxygenase in eosinophils and has been detected
in inflammatory exudates, while 22-hydroxy-MaR1 is produced by neutrophils and 14‑oxo-MaR1 by macrophages in inflammation induced
by bacterial infection; 22‑hydroxy-MaR1 retains the actions of its parent.
5. Sulfido-Conjugates of Specialized Pro-Resolving Mediators
Sulfido-peptide conjugated mediators with some structural similarity to the cysteinyl-leukotrienes and with novel properties are now known to be produced from SPMs in macrophages, and they have been termed protectin, resolvin and maresin conjugates in tissue regeneration (PCTR, RCTR and MCTR, respectively). While 13S,14S‑epoxy-maresin, produced as described above, is a lipid mediator in its own right, it is the precursor of a family of further metabolites, i.e., 13‑glutathionyl,14-hydroxy-docosahexaenoic (MCTR1), 13‑cysteinylglycinyl,14-hydroxy-docosahexaenoic (MCTR2) and 13‑cysteinyl,14-hydroxy-docosahexaenoic (MCTR3) acids. Indeed, it is now evident that the same enzymes are used for the biosynthesis of these two oxylipin mediator families. In brief, 13S,14S-epoxy-maresin is acted upon by the leukotriene C4 synthase or by glutathione S-transferase Mu 4 to produce MCTR1, which is converted to first to MCTR2 by a γ‑glutamyltransferase and thence to MCTR3 by a dipeptidase.
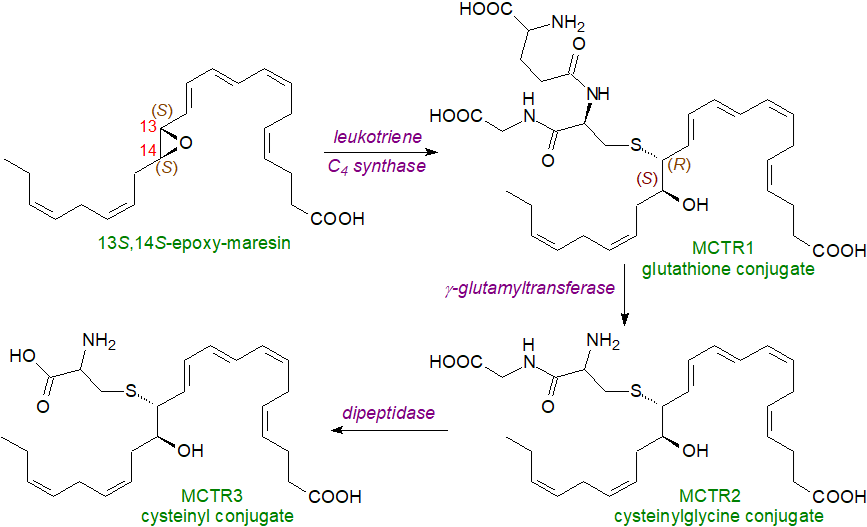 |
| Figure 8. Proposed biosynthetic route to sulfido-conjugated mediators. |
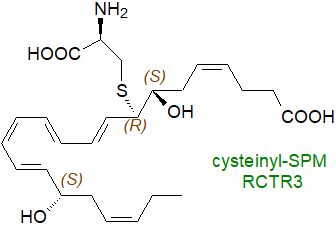 Sulfido-conjugates are produced by resolvins (RCTR1 and 4S,5R-RCTR1, RCTR2 and RCTR3) and protectins
(PCTR1, PCTR2 and PCTR3) in the same way, and those of the 17-series are produced by human leukocytes and are abundant in
lymphatic tissue.
RCTR3 is 8R-cysteinyl-7S,17S-dihydroxy-4Z,9E,11E,13Z,15E,19Z-docosahexaenoic
acid, for example, i.e., it is derived from the epoxy intermediate 7S,8S‑epoxy-17S-hydroxy-DHA with the sulfur atom
of cysteine attached to carbon 8.
Sulfido-conjugates are produced by resolvins (RCTR1 and 4S,5R-RCTR1, RCTR2 and RCTR3) and protectins
(PCTR1, PCTR2 and PCTR3) in the same way, and those of the 17-series are produced by human leukocytes and are abundant in
lymphatic tissue.
RCTR3 is 8R-cysteinyl-7S,17S-dihydroxy-4Z,9E,11E,13Z,15E,19Z-docosahexaenoic
acid, for example, i.e., it is derived from the epoxy intermediate 7S,8S‑epoxy-17S-hydroxy-DHA with the sulfur atom
of cysteine attached to carbon 8.
Macrophages produce many such metabolites including a protectin analogue PCTR1 derived from 16S,17S-epoxy-protectin, in which the sulfur atom is attached to carbon 16, and PCTR1 in turn gives rise to PCTR2 and PCTR3. These oxylipins have been detected in lymph nodes, serum and milk in humans, and they regulate the ability of the system to clear bacteria and to repair and regenerate damaged tissues. They have been detected in mice and primordial organisms such as Planaria.
6. Specialized Pro-Resolving Mediators and Inflammation
Note! A review by a multi-author/national group has cast doubt upon many aspects of this topic. This may mean that I have to re-evaluate what follows, but I do not plan to make significant changes before I see how the debate develops (Schebb, N.H. and 18 others. Formation, signaling and occurrence of specialized pro-resolving lipid mediators - what is the evidence so far? Front. Pharmacol., 13, 838782 (2022); DOI). An important point of contention is whether these mediators can be detected naturally in tissues at levels that have the effects claimed in cells in vitro, and there is a report that they are barely detectable in human leukocytes, which are considered to be the main source (Kahnt, A.S. et al. Formation of lipoxins and resolvins in human leukocytes. Prostaglandins Other Lipid Mediators, 166, 106726 (2023); DOI).
The protectins, resolvins and maresins (SPMs) are distinctive oxylipins with highly stereospecific structures, and they are endogenous local mediators that are strongly pro-resolution and immunoregulatory at picomolar to nanomolar concentrations. This stereospecificity is required for their functions, and unnatural stereoisomers tend to be much less efficient. As part of the molecular mechanisms that contribute to removal of inflammatory cells and restoration of tissue integrity once the need for the inflammatory response is over, SPMs assist in the resolution of inflammation, once thought to be a passive process, and they are strong attenuators of oxidative stress (strictly speaking, pro-resolution is not identical to anti-inflammation). In that it controls many aspects of both the innate and adaptive arms of the immune response, the nervous system is a central player in stimulating SPM production with neurotransmitters such acetylcholine being of particular relevance. It is noteworthy that SPMs are not immunosuppressive. Several SPMs may take part in the response to an inflammatory challenge, but each will have its own time-course for biosynthesis and insertion into the process, and the following discussion is necessarily simplistic and can only touch upon what is an increasingly complex and rapidly developing topic.
Each SPM is believed to interact with its own G protein coupled receptor, which can evoke rapid intracellular signalling and long-term actions by regulating the expression of genes necessary for the resolution of inflammation, although the evidence is contested, especially for lipoxins (Table 1). Depending on the cellular context, each receptor may interact with more than one SPM, so RvD1 acts with DRV1 for homeostatic functions and with ALX/FPR2 for anti-neutrophil actions in resolving inflammation, while for maresin 1, RORα is a nuclear receptor and LGR6 is a plasma membrane G protein coupled receptor. It is noteworthy that SPMs can also antagonize pro-inflammatory receptors, and RvE1, MaR1 and 22-OH-MaR1 are all competitive antagonists of the leukotriene B4 receptor BLT1. On the other hand, it has been suggested from studies with pure synthetic SPMs and macrophages that most SPMs are positive allosteric modulators of prostaglandin E2 receptor subtype 4 (EP4), which enhances coupling of EP4 to the Gs signalling pathway and enables it to couple to Gi-proteins, but this has been criticised as the levels of SPMs utilized experimentally to achieve such pro-resolving properties exceed by orders of magnitude the concentrations recent studies suggest may be present endogenously.
Table 1. Possible receptors for SPMs in mice and humans. |
|
| SPM | Receptor |
| RvD1 | ALX/FPR2 and DRV1 (GPR32) |
| RvD2 | DRV2 (GPR18) |
| RvE1 and RvE2 | ERV1 (ChemR23) and BLT1 |
| Protectins (PD1) | GPR37 |
| RvD1n-3PDA | ALX/FPR2 and GPR32/DRV1 |
| RvD5n-3PDA | GPR101 |
| Maresin 1 |
LGR6 |
Acute inflammation in response to infection or tissue damage is an essential defensive process that is usually characterized by heat, redness, swelling and pain at a simple observational level, and by oedema, accumulation of neutrophils and then of phagocytes such as monocytes and macrophages at a cellular level. The latter are the first line of defence of the innate immune system to defeat pathogens and toxins traveling through the body, and they protect each organ by engulfing and killing invading pathogens and clearing debris from within cells. Failure to achieve proper resolution can lead to chronic inflammation with consequences that can include tissue damage and impairment including extensive collagenous scars as part of the pathogenesis of various disorders. Leukotrienes (especially LTB4) and prostaglandins (PGE2 and PGD2) derived from arachidonic acid are important in the early stages of the inflammatory process by stimulating the migration of neutrophils to the affected tissue, and biosynthesis of these prostaglandins is critical for resolution of inflammation as they stimulate the induction of the 15‑lipoxygenases that are necessary for subsequent production of other SPMs. Efficient resolution of inflammation can be beneficial towards atherosclerosis and other chronic autoimmune diseases (see next section).
Early in the acute inflammatory response, the basis is laid for a dynamic and detailed programmed biosynthesis of SPMs, i.e., a change in lipid mediator production in which arachidonic acid metabolism switches from the production of leukotrienes to that of SPMs. Local mobilization of the SPM precursor DHA occurs before the autacoid protectins, resolvins and maresins are produced at the site of inflammation to help tissues to return to health by promoting resolution of inflammation through recruitment of non-inflammatory monocytes while limiting that of proinflammatory granulocytes. After this switch in the nature of the mediators formed, macrophages and mast cells can remove excess neutrophils together with cellular debris via draining lymph nodes without leaving remnants of the host defences or of the invading microorganisms or other inflammatory initiators, which might prolong inflammation. It is evident that the presence of aspirin uniquely facilitates this resolution phase at local sites of inflammation by enhancing the conversion of the omega-3 fatty acids EPA and DHA to 18R- and 17R-oxygenated products, respectively, i.e., precursors of resolvins of the E and D series, which carry anti-inflammatory signals.
 It
has become apparent that a novel aspect of the resolution process is that SPMs are able to induce changes
in the phenotype of macrophages toward a pro-resolution state.
During inflammation, polymorphonuclear neutrophils are produced which are generally beneficial in countering
disease by removing invading pathogens by phagocytosis, but in the longer term or if malfunctioning, they may eventually cause
trauma and cellular damage through infiltration into tissues.
The resolvins may regulate and even inhibit such harm, and in so doing, they oppose some of the pro-inflammatory prostanoids.
A cluster of SPMs, including RvD3, RvD4 and RvD6, are produced in human blood to target leukocytes at the single-cell level, directly
inducing extracellular signalling in human neutrophils and monocytes to promote host defence by enhancing phagocytosis and killing
bacteria.
It
has become apparent that a novel aspect of the resolution process is that SPMs are able to induce changes
in the phenotype of macrophages toward a pro-resolution state.
During inflammation, polymorphonuclear neutrophils are produced which are generally beneficial in countering
disease by removing invading pathogens by phagocytosis, but in the longer term or if malfunctioning, they may eventually cause
trauma and cellular damage through infiltration into tissues.
The resolvins may regulate and even inhibit such harm, and in so doing, they oppose some of the pro-inflammatory prostanoids.
A cluster of SPMs, including RvD3, RvD4 and RvD6, are produced in human blood to target leukocytes at the single-cell level, directly
inducing extracellular signalling in human neutrophils and monocytes to promote host defence by enhancing phagocytosis and killing
bacteria.
The resolvins RvE1, RvE2 and RvE3 have separate but coordinated anti-inflammatory properties and take part in the resolution of inflammation through the release of interleukin (IL)-10, the expression of its receptor, and phagocytosis. Nanomolar concentrations of resolvin E1 derived from EPA dramatically reduce dermal inflammation, peritonitis, dendritic cell migration, lung damage from smoking and interleukin production. In a model of periodontitis, RvE1 alone of the SPMs was able to reduce local inflammation by suppressing superoxide production directly in primary neutrophils. RvE1 blocks excessive platelet aggregation, it inhibits certain human pathogens by enhancing phagocytosis by polymorphonuclear leukocytes, and it is reported to be more effective in reducing inflammatory pain than morphine. RvE2 reduces joint pain in arthritis, and RvD5 has beneficial properties in an experimental model of this disease.
RvD1 is reported to have anti-inflammatory, antioxidant, anti-fibrosis, anti-apoptotic and anti-tumour properties by binding to its receptors, and it has therapeutic potential in liver diseases. RvD2 and RvD3 derived from DHA have regulatory actions on neutrophil trafficking in the picogram range in vivo by stimulating resolution and enhancing innate host defence mechanisms to clear invading pathogens via a specific receptor. Thus, in experimental animals, RvD2 interacting with the receptor DRV2 enhances killing and clearance of bacteria and ameliorates the symptoms of bacterial sepsis. It prolongs survival from severe burns and reduces colitis, and it targets myogenic cells and improves muscle regeneration in mouse models of Duchenne muscular dystrophy.
In general, RvD3 and RvD4 appear late in resolution suggesting that they have a special role in the last stages of the process. RvD3 is protective of tissues, and in uninjured lungs, it may regulate tissue tone. RvD4 has pro-resolving actions in Staphylococcus aureus infections, and it mitigates lipopolysaccharide-induced lung injury in mice. It is a powerful stimulant for the clearance of apoptotic cells by skin fibroblasts, and it is produced in mouse bone marrow and lung in response to ischemic injury with reductions in the concentrations of pro-inflammatory eicosanoids with amelioration of thrombosis. Similarly, RvD5 controls E. coli and S. aureus infections and lowers the antibiotic requirements for bacterial clearance. Although contradictory results have been obtained in administering resolvins to treat sepsis in some experimental models, this may have been because the appropriate receptors were down regulated.
Protectins operate in the same way as the resolvins in brain tissue. Thus, (N)PD1 is anti-inflammatory by promoting resolution of neuroinflammation and nerve regeneration. It is protective in animal models of stroke and of Alzheimer's disease, and in a mouse model of epilepsy, considerable improvements in the condition were observed after treatment with PD1. In non-neuronal tissues, it promotes apoptosis of T cells, it is beneficial towards asthma in nanogram amounts, and it is reported to have therapeutic potential against virus infections by inhibiting viral replication. PD1 binds to retinal pigment epithelial cells in a highly selective manner to protect from apoptosis induced by oxidative stress. Protectins synthesised in white adipose tissue are anti-inflammatory in obesity and diabetes, and both protectins and maresins reduce neuropathic pain in experimental animals.
 The
poxytrin PDX has properties that differ from those of PD1.
It may have antithrombotic potential as an inhibitor of platelet activation against thromboxane A2-induced
aggregation, it may inhibit the inflammation associated with cyclooxygenase (COX), and it has been reported to inhibit influenza virus
replication by targeting its RNA metabolism.
The
poxytrin PDX has properties that differ from those of PD1.
It may have antithrombotic potential as an inhibitor of platelet activation against thromboxane A2-induced
aggregation, it may inhibit the inflammation associated with cyclooxygenase (COX), and it has been reported to inhibit influenza virus
replication by targeting its RNA metabolism.
Maresin 1 is a powerful regulator of the resolution of inflammation, tissue regeneration and pain to influence the progression of many disease states including lung, bone, vascular and metabolic diseases and bacterial infections. In general, maresins may be required for tissue regeneration and wound healing in the later stages of resolution (D-series resolvins also participate in skin repair after wounding). MaR1 stimulates osteoblast proliferation through leucine-rich repeat G protein-coupled receptor 6 (LGR6) and has the potential to have a beneficial impact upon such bone diseases as osteoporosis, osteoarthritis, rheumatoid arthritis and periodontitis. In studies with planaria, a primordial organism utilized for surgical injury experiments, maresins and RvE1 each markedly improved the rates of tissue regeneration. MaR1 is a key mediator of the switch to the macrophage M2 phenotype thereby stimulating macrophage phagocytosis and efferocytosis to enhance the clearance of inflammation without affecting the innate response, while it interferes with vanilloid receptor TRPV1 in dorsal root ganglion neurons and blocks capsaicin. After spinal cord injury, it was found to improve neurological recovery. The epoxy intermediate in maresin biosynthesis, 13S,14S‑epoxy-maresin, affects macrophages independently by inhibiting the production of inflammatory eicosanoids such as leukotrienes by direct inhibition of the LTA4 hydrolase. 7S,14R-diHDHA, a stereoisomer of maresin-1 produced by leukocytes, can assist mesenchymal stem cells to ameliorate some of the symptoms of diabetic mellitus.
At nanomolar concentrations, sulfido-conjugates derived from epoxy-maresin are reported to resolve E. coli infections, and in general they may constitute a novel signalling mechanism that contains infections, stimulates resolution of inflammation, and promotes the restoration of human tissues and those of experimental animals. By stimulating human macrophages, protectin and resolvin sulfido-conjugates limit bacterial infections in a dose-dependent manner, and RCTR1, RCTR2 and RCTR3 each exert potent anti-inflammatory and proresolving actions with human leukocytes. PCRT1 is an agonist of monocytes and macrophages and regulates key anti-inflammatory and pro-resolving processes during infection by bacteria. Inflammatory effects of cysteinyl leukotrienes in the lung are countered by cysteinyl maresins.
Although the specialized pro-resolving mediators are produced locally in many different tissues to terminate inflammation, they reach the circulation and are found in human peripheral blood, suggesting that they may act as anti-inflammatory signals in tissues other than those in which they originate. It has been claimed that proresolving mediators are present at bioactive levels in human breast milk and in human and rat placentas, so it is possible that they may regulate normal physiological development as well as pathophysiological processes. It is hoped that measurement of the levels of these mediators in infected patients will provide a better understand of their inflammation-resolution status and might predict outcome or whether therapeutic treatments are likely to be successful.
7. Specialized Pro-Resolving Mediators and Disease
There are many reports that imbalances between specialized proresolving and proinflammatory mediators leading to impaired resolution are associated with several human diseases. Thus, it is now well established that administration of protectins, resolvins and maresins in vivo and in vitro in animal models can aid the process of recovery from inflammation without compromising host defences by causing immune suppression, and there is a view that promoting resolution instead of inhibiting inflammation may be a way forward in treating many infections. It is claimed that SPMs have considerable potential for therapeutic intervention in acute inflammation or chronic inflammatory diseases, and they have been tested in a wide range of experimental models, including peritonitis, periodontitis, colitis, arthritis, psoriasis, dry eye, inflammatory pain, cardiovascular disease (including atherosclerosis), depression, neurodegenerative disorders, and asthma and other lung ailments (dependent on age, sex and race). Synthetic lipoxins administered at levels orders of magnitude higher than may be present naturally may also be anti-inflammatory.
There is evidence that SPMs can cross the blood-brain barrier, and they hold promise for neuroprotection by altering the expression of pro-inflammatory genes and promoting resolution of neuroinflammation. In particular, RVD1 and its epimer, aspirin-triggered RvD1 (AT‑RvD1), are reported to be beneficial at nanomolar and micromolar concentrations on many neurological conditions, including experimental models of traumatic brain injuries, pain, depression, and Alzheimer's and Parkinson's diseases. Elovanoids have been reported to counteract the expression of genes for β‑amyloid protein in a model of Alzheimer's disease, and they protect the retinal pigment epithelial cells and sustain photoreceptor cell integrity in the retina, e.g., during age-related macular degeneration; they are neuroprotective in experimental ischemic stroke and in a traumatic brain injury model in rats. In Parkinson's disease, data suggest that SPMs inhibit microglial activation and decrease induced markers of inflammation.
Inflammation in the tumour microenvironment is a defining property of cancers that is exacerbated by carcinogens. By stimulating the resolution of inflammation, SPMs are anti-cancer agents in that they suppress tumour growth and enhance therapy by stimulating clearance of tumour debris by macrophages. In particular, the aspirin-triggered specialized proresolving mediators (AT‑SPMs), such as aspirin-triggered resolvins (AT-RvDs) and lipoxins (AT‑LXs), are potent anti-tumour agents and may provide an explanation for the broad efficacy of aspirin in the treatment of many cancers (in addition to its inhibition of prostaglandin biosynthesis). SPMs may re-programme tumour-associated macrophages to an anti-tumour phenotype. As SPMs operate at much lower concentrations than aspirin, it is hoped that they may be a non-toxic route to harnessing aspirin as an anticancer agent so reducing carcinogen-induced morbidity and mortality. Another therapeutic approach may be to inhibit the soluble epoxide hydrolase, as this stimulates the production of SPMs.
 In
relation to cardiovascular disease, it has been reported that the deleterious effects of leukotriene LTB4
resulting from an excessive inflammatory response are countered by the presence of specialized proresolving mediators like RvD1
in experimental models of atherosclerosis, and it has been suggested that this may be a new therapeutic approach to limiting the progression
of atherosclerotic plaques and promoting plaque stability.
Similarly, RvE1 controls vascular inflammation and protects against atherosclerosis by modifying the uptake of oxidized LDL
while enhancing phagocytosis of macrophages.
During blood coagulation, a characteristic SPM cluster has been detected that consists of resolvin El (RvE1), RvD1, RvD5, lipoxin B4 and maresin 1
at bioactive concentrations (0.1 to 1 nM), together with eicosanoids, such as prostaglandins, thromboxanes and leukotrienes.
Studies with animal models suggest that supplementation with RvD2 may be beneficial towards cardiovascular and metabolic diseases,
and in particular, it limits the brain damage caused by myocardial infarction and reverses the resultant neurological dysfunction.
In
relation to cardiovascular disease, it has been reported that the deleterious effects of leukotriene LTB4
resulting from an excessive inflammatory response are countered by the presence of specialized proresolving mediators like RvD1
in experimental models of atherosclerosis, and it has been suggested that this may be a new therapeutic approach to limiting the progression
of atherosclerotic plaques and promoting plaque stability.
Similarly, RvE1 controls vascular inflammation and protects against atherosclerosis by modifying the uptake of oxidized LDL
while enhancing phagocytosis of macrophages.
During blood coagulation, a characteristic SPM cluster has been detected that consists of resolvin El (RvE1), RvD1, RvD5, lipoxin B4 and maresin 1
at bioactive concentrations (0.1 to 1 nM), together with eicosanoids, such as prostaglandins, thromboxanes and leukotrienes.
Studies with animal models suggest that supplementation with RvD2 may be beneficial towards cardiovascular and metabolic diseases,
and in particular, it limits the brain damage caused by myocardial infarction and reverses the resultant neurological dysfunction.
RvD5n-3 DPA and PD1n-3 DPA are protective against intestinal inflammation and intestinal diseases including inflammatory bowel disease; the latter promotes resolution of neuroinflammation and may be beneficial in epilepsy. RvD1 produced by goblet cells of the eye interacts with its receptors ALX/FPR2 and GPR32 to set off a chain of reactions that result in mucin secretion into the tear film to protect the ocular surface and maintain a healthy interface between the eye and the environment. As examples of the therapeutic potential of SPMs, a phase III clinical trial of a RvE1 analogue against dry eye syndrome is underway, and NPD1/PD1 is in clinical development for neurodegenerative diseases. There is hope that SPMs may prove efficacious in treating the low-level inflammatory conditions that occurs in obesity, including insulin resistance, type 2 diabetes and non-alcoholic fatty liver disease, and they are considered to be candidate drugs for treating chronic inflammation and infection in patients with cystic fibrosis.
In general, specialized proresolving mediators are beneficial towards attack by bacteria, fungi, viruses and parasites, including such debilitating diseases as influenza and malaria, by limiting neutrophil influx, stimulating phagocytosis, killing bacteria, and clearance of apoptotic neutrophils and cellular debris by macrophages. As the actions of resolvin RvD1 are additive to those of antibiotics, they reduce the doses required to clear both Gram-positive and Gram-negative infections. SPMs synthesised during viral infections may control the magnitude and duration of the inflammatory response and stimulate host immunity to protect from organ damage. RvE1 and lipoxins applied topically were found to have benefits in the treatment of experimental periodontitis. Discovery of agonist antibodies for the pro-resolving receptors that induce endogenous resolution, such as those for ChemR23, add to the potential for novel therapeutic approaches to control excessive inflammation and cancer.
 As SPMs tend to be relatively unstable and are not available in sufficient quantities, it is unlikely that they will be used
directly in a clinical setting.
However, they are being used as templates for novel designer drugs or mimetics to treat inflammatory diseases, and hopefully these will reduce
the requirement for antibiotics if they can be delivered in humanized nano particles.
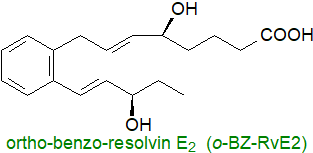
A synthetic RvE2 analogue, ortho-benzo-resolvin E2, replacing C9 to C15 with a benzene ring, was found to be
more efficient than RvE2 in vivo (femtomole concentrations) at reducing inflammation and was more stable, while addition of a methyl
group to carbon 15 of lipoxin A4 to produce 15R/S-methyl-LXA4 prevents metabolism by dehydrogenation while
retaining the activity of native LXA4; it has shown efficacy and safety in early clinical trials of patients
with infantile eczema.
As SPMs tend to be relatively unstable and are not available in sufficient quantities, it is unlikely that they will be used
directly in a clinical setting.
However, they are being used as templates for novel designer drugs or mimetics to treat inflammatory diseases, and hopefully these will reduce
the requirement for antibiotics if they can be delivered in humanized nano particles.
A synthetic RvE2 analogue, ortho-benzo-resolvin E2, replacing C9 to C15 with a benzene ring, was found to be
more efficient than RvE2 in vivo (femtomole concentrations) at reducing inflammation and was more stable, while addition of a methyl
group to carbon 15 of lipoxin A4 to produce 15R/S-methyl-LXA4 prevents metabolism by dehydrogenation while
retaining the activity of native LXA4; it has shown efficacy and safety in early clinical trials of patients
with infantile eczema.
From a nutritional or health standpoint, it has been reported that dietary supplements of the precursor omega-3 fatty acids, taken together with aspirin, can stimulate the synthesis of SPMs with the potential to ameliorate the symptoms of many inflammatory disorders by regulating the time course of resolution via the production of resolvins and protectins. However, there do not appear to be any definitive clinical trials to confirm this. Of course, omega-3 fatty acids may have benefits in many normal physiological processes, including ensuring maternal/foetal health.
8. Analysis
Because of the low levels at which SPMs occur naturally in tissues and their chemical reactivity, analysis is a considerable challenge with few standards available for comparison, calibration or validation purposes. Modern tandem mass spectrometric methods linked to liquid chromatography on reversed-phase columns are essential, and even these are pushing the boundaries of sensitivity at natural levels. Such difficulties are at the root of the present debate on how SPMs operate in tissues in vivo. Ideally, chiral chromatography procedures should be used as part of any analytical protocol to ensure that those forms of SPMs with the correct R/S stereochemistry are present rather than random and racemic autoxidation products, including those formed adventitiously during faulty handling and storage of samples. ELISA methods are not sufficiently specific and reliable.
Recommended Reading
- Bazan, N.G. Overview of how N32 and N34 elovanoids sustain sight by protecting retinal pigment epithelial cells and photoreceptors. J. Lipid Res., 62, 10058 (2021); DOI.
- Clària, J., Flores-Costa, R., Duran-Güell, M. and López-Vicario, C. Proresolving lipid mediators and liver disease. Biochim. Biophys. Acta, Lipids, 1866, 159023 (2021); DOI.
- Dalli, J. (Editor) The physiology and pharmacology of specialized pro-resolving mediators. Molecular Aspects of Medicine, Volume 58, Pages 1-130 (2017) - special journal issue.
- Dyall, S.C., Balas, L., Bazan, N.G., Brenna, J.T., Chiang, N., Souza, F.D., Dalli, J., Durand, T., Galano, J.M., Lein, P.J., Serhan, C.N. and Taha, A.Y. Polyunsaturated fatty acids and fatty acid-derived lipid mediators: Recent advances in the understanding of their biosynthesis, structures, and functions. Prog. Lipid Res., 86, 101165 (2022); DOI.
- Fredman, G. and Serhan, C.N. Specialized pro-resolving mediators in vascular inflammation and atherosclerotic cardiovascular disease. Nature Rev. Cardiol., 21, 808-823 (2024); DOI.
- Guichardant, M., Véricel, E. and Lagarde, M. Biological relevance of double lipoxygenase products of polyunsaturated fatty acids, especially within blood vessels and brain. Biochimie, 159, 55-58 (2019); DOI.
- Hansen, T.V. and Serhan, C.N. Protectins: Their biosynthesis, metabolism and structure-functions. Biochem. Pharm., 206, 115330 (2022); DOI.
- Ji, R.R. Specialized pro-resolving mediators as resolution pharmacology for the control of pain and itch. Annu. Rev. Pharm. Toxic., 63, 273-293 (2023); DOI.
- Jordan, P.M. and Werz, O. Specialized pro-resolving mediators: biosynthesis and biological role in bacterial infections. FEBS J., 289, 4212-4227 (2022); DOI.
- Julliard, W.A., Myo, Y.P.A., Perelas, A., Jackson, P.D., Thatcher, T.H. and Sime, P.J. Specialized pro-resolving mediators as modulators of immune responses. Seminars Immunol., 59, 101605 (2022); DOI.
- Kampschulte, N., Kirchhoff, R., Löwen, A. and Schebb, N.H. Deducing formation routes of oxylipins by quantitative multiple heart-cutting achiral-chiral 2D-LC-MS. J. Lipid Res., 65, 100694 (2024); DOI.
- Panigrahy, D., Gilligan, M.M., Serhan, C.N. and Kashfi, K. Resolution of inflammation: An organizing principle in biology and medicine. Pharm. Therapeut., 227, 107879 (2021); DOI.
- Ponce, J. and 10 others. Role of specialized pro-resolving mediators in reducing neuroinflammation in neurodegenerative disorders. Front. Aging Neurosci., 14, 780811 (2022); DOI.
- Quinlivan, K.M., Howard, I.V., Southan, F., Bayer, R.L., Torres, K.L., Serhan, C.N. and Panigrahy, D. Exploring the unique role of specialized pro-resolving mediators in cancer therapeutics. Prostaglandins Other Lipid Mediators, 178, 106944 (2025); DOI.
- Reinertsen, A.F., Vik, A. and Hansen, T.V. Biology and total synthesis of n-3 docosapentaenoic acid-derived specialized pro-resolving mediators. Molecules, 29, 2833 (2024); DOI.
- Serhan, C.N. and Levy, B.D. Proresolving lipid mediators in the respiratory system. Annu. Rev. Physiol., 87, 491-512 (2025); DOI.
- Serhan, C.N., Libreros, S. and Nshimiyimana, R. E-series resolvin metabolome, biosynthesis and critical role of stereochemistry of specialized pro-resolving mediators (SPMs) in inflammation-resolution: Preparing SPMs for long COVID-19, human clinical trials, and targeted precision nutrition. Seminars Immunol., 59, 101597 (2022); DOI.
- Sun, S.J., Yang, D., Lv, J., Xia, H.F., Mao, Z.Y., Chen, X.D. and Gao, Y.F. Pharmacological effects of specialized pro-resolving mediators in sepsis-induced organ dysfunction: a narrative review. Front. Immun., 15, 1444740 (2024); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: October 2025 | ||
© The LipidWeb is open access and fair use is encouraged but not text and data mining, AI training, and similar technologies.
