Phosphatidylethanolamine and Related Lipids
Phosphatidylethanolamine or 1,2-diacyl-sn-glycero-3-phosphoethanolamine (once given the trivial name 'cephalin') is usually the second most abundant phospholipid in animals and plants after phosphatidylcholine, and it is often the main lipid component of microbial membranes. It can amount to 20% of liver phospholipids and as much as 45% of those of brain; higher proportions are found in mitochondria than in other organelles. As such, it is obviously a key building block of membrane bilayers, where it is present exclusively in the inner leaflet of the plasma membrane in animal cells. It is a neutral or zwitterionic phospholipid (at least in the pH range 2 to 7)

1. Phosphatidylethanolamine – Structure and Occurrence
In animal tissues, phosphatidylethanolamine tends to exist in diacyl, alkylacyl and alkenylacyl forms, and data for the compositions of these various forms from phosphatidylcholine from bovine heart muscle as an example are listed in our web page on ether lipids; a diacyl form is illustrated (of one molecular species). As much as 70% of the phosphatidylethanolamine in some cell types (e.g., inflammatory cells, neurons and tumour cells) can have an ether linkage in position sn-1, but in liver, the plasmalogen form of phosphatidylethanolamine, i.e., with an O‑alk-1′-enyl linkage, accounts for only 0.8% of total phospholipids. Generally, there is a much higher proportion of phosphatidylethanolamine with ether linkages than of phosphatidylcholine. If biosynthesis of the plasmalogen form is inhibited by physiological conditions, it is replaced by the diacyl form so that the overall content of the phospholipid remains constant.
In general, animal phosphatidylethanolamine tends to contain higher proportions of arachidonic and docosahexaenoic acids than the other zwitterionic phospholipid, phosphatidylcholine. These polyunsaturated components are concentrated in position sn-2 with saturated fatty acids most abundant in position sn-1, as illustrated for rat liver and chicken egg in Table 1. In most other animals, it would be expected that the structure of the phosphatidylethanolamine in the same tissues would exhibit comparable features.
Table 1. Positional distribution of fatty acids in phosphatidylethanolamine in animal tissues. |
|||||||
| Position | Fatty acid | ||||||
|---|---|---|---|---|---|---|---|
| 14:0 | 16:0 | 18:0 | 18:1 | 18:2 | 20:4 | 22:6 | |
| Rat liver [1] | |||||||
| sn-1 | 25 | 65 | 8 | ||||
| sn-2 | 2 | 11 | 8 | 8 | 10 | 46 | 13 |
| Chicken egg [2] | |||||||
| sn-1 | 32 | 59 | 7 | 1 | |||
| sn-2 | 1 | 1 | 25 | 22 | 29 | 12 | |
| 1, Wood, R. and Harlow, R.D., Arch. Biochem. Biophys., 131, 495-501 (1969);
DOI. 2, Holub, B.J. and Kuksis, A. Lipids, 4, 466-472 (1969); DOI. | |||||||
The O-alkyl and O-alkenyl groups at the sn-1 position of the analogous ether lipids generally have 16:0, 18:0 or 18:1 chains, whereas arachidonic and docosahexaenoic acids are the most abundant components at the sn-2 position. Please note that is not possible to reproduce recent molecular species data in tabular form here.
The positional distributions of fatty acids in phosphatidylethanolamine from the leaves of the model plant Arabidopsis thaliana are listed in Table 2. Here again, saturated fatty acids are concentrated in position sn-1, and there is a preponderance of di- and triunsaturated components in position sn-2. The pattern is somewhat different for the yeast Lipomyces lipoferus, where the compositions of the two positions are comparable, while in the gram-negative bacterium Escherichia coli saturated fatty acids are again concentrated in position sn-1, while monoenoic and cyclopropane fatty acids are located in position sn-2.
Table 2. Composition of fatty acids (mol %) in positions sn-1 and sn-2 in the phosphatidylethanolamine from A. thaliana (leaves), L. lipoferus and E. coli. |
|||||||
| Position | Fatty acid | ||||||
|---|---|---|---|---|---|---|---|
| 16:0 | 16:1 | 18:0 | 18:1 | 18:2 | 18:3 | Other | |
| A. thaliana [1] | |||||||
| sn-1 | 58 | trace | 4 | 5 | 15 | 18 | |
| sn-2 | trace | trace | trace | 2 | 60 | 38 | |
| L. lipoferus [2] | |||||||
| sn-1 | 29 | 18 | 4 | 28 | 13 | 6 | |
| sn-2 | 23 | 15 | 3 | 34 | 17 | 6 | |
| E. coli [3] | |||||||
| sn-1 | 75.2 | 2.9 | 3.2 | 16.1 | - | - | - |
| sn-2 | 6.9 | 44.1 | - | 38.4 | - | - | 10.6* |
| 1, Browse, J., Warwick, N., Somerville, C.R. and Slack, C.R. Biochem.
J., 235, 25-31 (1986); DOI. 2, Haley, J.E. and Jack, R.C. Lipids, 9, 679-681 (1974); DOI. 3, Van Golde, L.M.G. and van Deenen, L.L.M. Chem. Phys. Lipids, 1, 157-164 (1967); DOI. * C17 cyclopropanoid fatty acid |
|||||||
2. Phosphatidylethanolamine – Biosynthesis
The two main pathways employed by mammalian cells for the biosynthesis of phosphatidylethanolamine are the CDP-ethanolamine pathway, i.e., one of the general routes to phospholipid biosynthesis de novo in plants and animals, and the phosphatidylserine decarboxylase pathway, which occur in two organelles, the endoplasmic reticulum and mitochondria (and at contact points between the two). Ethanolamine is obtained by decarboxylation of serine in plants, but in animals, most must come from dietary sources and requires facilitated transport into cells. A small amount of ethanolamine phosphate comes from catabolism of sphingosine-1-phosphate, and this is essential for the survival of the protozoon Trypanosoma brucei. Diacyl and ether forms of phosphatidylethanolamine are produced by different mechanisms, and the latter are discussed briefly below and in greater detail in a separate webpage.
The initial steps in diacyl-phosphatidylethanolamine biosynthesis de novo occur in the cytosol with first the phosphorylation of ethanolamine by two ethanolamine kinases to produce ethanolamine phosphate; the reverse reaction can occur by means of the enzyme ethanolamine phosphate phospholyase and this may be regulatory in some tissues. In the second and rate-limiting step, the reaction of ethanolamine phosphate with cytidine triphosphate (CTP) to form cytidine diphosphoethanolamine is catalysed by CTP:phosphoethanolamine cytidylyltransferase. In the seeds of higher plants, a deficiency of this last enzyme has profound effects upon the viability and maturation of embryos.
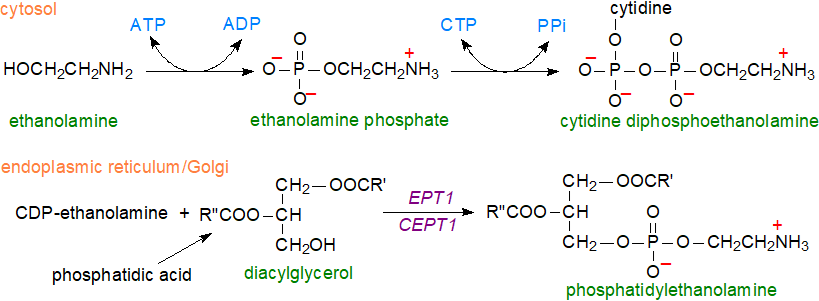 |
| Figure 1. Biosynthesis of phosphatidylethanolamine de novo in cells. |
In the final step, a membrane-bound enzyme CDP-ethanolamine:diacylglycerol ethanolamine-phosphotransferase catalyses the reaction of cytidine diphosphoethanolamine with diacylglycerol to form phosphatidylethanolamine. There are two such enzymes, ethanolamine phosphotransferase 1 (EPT1) in the Golgi and a choline/ethanolamine-phosphotransferase (CEPT1) in the endoplasmic reticulum, but EPT1 is more active in the biosynthesis of the plasmalogen form, 1‑alkenyl-2-acyl-glycerophosphoethanolamine, and especially those molecular species containing polyunsaturated fatty acids, while CEPT1 produces species with shorter-chain fatty acids. The diacylglycerol precursor is formed from phosphatidic acid by the action of the enzyme phosphatidic acid phosphohydrolase (see our web pages on triacylglycerols and phosphatidylcholine). Phosphatidylethanolamine is transported from the endoplasmic reticulum to the plasma membrane by a bridge-like tether, the Vps13-like protein BLTP2, which regulates phosphatidylethanolamine levels to maintain plasma membrane fluidity. An enzyme that degrades ethanolamine phosphate, i.e. ethanolamine phosphate phospholyase, is part of the regulatory mechanism for hepatic phosphatidylethanolamine synthesis and plasma lipoprotein metabolism in mice.
The second major pathway for biosynthesis of phosphatidylethanolamine is decarboxylation of phosphatidylserine in mitochondria (as discussed further in our web pages on phosphatidylserine), and conservation of this pathway from bacteria to humans suggests that it has been preserved to optimize mitochondrial performance.
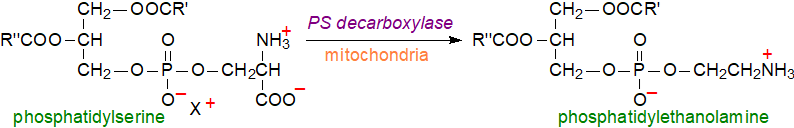 |
| Figure 2. Phosphatidylethanolamine synthesis from phosphatidylserine in mitochondria. |
The phosphatidylserine decarboxylase is located on the external aspect of the mitochondrial inner membrane, and most of the phosphatidylethanolamine in mitochondria is obtained in this way. While various isoforms of the phosphatidylserine decarboxylase exist in prokaryotes, yeasts and mammals, the main forms designated 'PSD1' are found only in mitochondria and are related structurally. An additional isoform designated 'PSD2' is located in the endosomal membranes of yeasts, and the phosphatidylethanolamine formed from this is the preferred substrate for phosphatidylcholine biosynthesis. It is evident that cellular concentrations of phosphatidylethanolamine and phosphatidylserine are interdependent and tightly regulated.
As mitochondria are not connected with the rest of the cell's membrane network by classical vesicular routes, they must receive and export small molecules through non-vesicular transport at zones of close proximity with other organelles at membrane contact sites such as a specific domain of the endoplasmic reticulum termed the mitochondria-associated membrane (MAM). Transport of phosphatidylserine is then enabled by tethers that bridge two membranes and by lipid transfer and recruitment proteins by which the lipid must traverse two aqueous compartments, the cytosol and the mitochondrial intermembrane space, to reach the inner mitochondrial membrane.
This pathway is important in mammalian cells and yeasts, although the relative contributions of the two main pathways for phosphatidylethanolamine synthesis in mammalian cells vary with cell type. In cells in culture, more than 80% of the phosphatidylethanolamine is reported to be produced by phosphatidylserine decarboxylase, but in hamster heart and rat hepatocytes, only ~5% comes from this route, and most is from the CDP-ethanolamine pathway. In yeasts, 70% of the phosphatidylethanolamine is generated by PSD1. The spatially segregated pools within the cell are functionally distinct, but both are essential to life. In embryonic mice, disruption of the phosphatidylserine decarboxylase gene causes misshapen mitochondria and has lethal consequences, although phosphatidylethanolamine synthesis continues for a time in other cellular regions. Similarly, elimination of the endoplasmic reticulum route is embryonically lethal, although in mature animals, a deficiency in synthesis is countered by reductions in degradation and molecular species remodelling by the Land’s cycle. Phosphatidylthreonine and phosphatidylhomoserine can be decarboxylated in the same way to form phosphatidylpropanolamine.
The various biosynthetic mechanisms produce different pools of phosphatidylethanolamine, which are often in different cellular compartments and have distinctive compositions. Studies with mammalian cell types in vitro suggest that the CDP-ethanolamine pathway produces molecules with mono- or di-unsaturated fatty acids on the sn-2 position preferentially, while the phosphatidylserine decarboxylation reaction generates mainly those with polyunsaturated fatty acids on the sn-2 position. In addition, two proteins designated TLCD1/2 promote the incorporation of monounsaturated fatty acids into phosphatidylethanolamine.
In prokaryotic cells, such as E. coli, in which phosphatidylethanolamine is the most abundant membrane phospholipid, all of it is derived from phosphatidylserine decarboxylation. In this instance, the enzyme undergoes auto-cleavage for activation and utilizes a pyruvoyl moiety to form a Schiff base intermediate with phosphatidylserine to facilitate decarboxylation.
Three further minor biosynthetic pathways are known. Phosphatidylethanolamine can be formed by re-acylation of lysophosphatidylethanolamine in the mitochondria-associated membrane where the phosphatidylserine synthase II is located or by an enzymatic exchange reaction of ethanolamine with phosphatidylserine, while the bacterial plant pathogen Xanthomonas campestris is able to synthesise phosphatidylethanolamine by condensation of cytidine diphosphate diacylglycerol with ethanolamine.
Ether lipids: It should be noted that all of these pathways for the biosynthesis of diacyl-phosphatidylethanolamine are very different and are separated spatially from that producing alkylacyl- and alkenylacyl-phosphatidylethanolamines and described in a separate web page, suggesting that there may be differences in their cellular roles. In the protozoon T. brucei, it has been demonstrated that the diacyl and ether pools of phosphatidylethanolamine are used for separate purposes and cannot substitute for each other.
Remodelling of molecular species: As with other phospholipids, the final fatty acid composition in animal tissues is attained by a process of remodelling known as the Lands' cycle (see the web page on phosphatidylcholine for a more detailed discussion). The first step is hydrolysis by a phospholipase A2 to lysophosphatidylethanolamine, followed by reacylation by means of various acyl-CoA:lysophospholipid acyltransferases. The enzymes LPLAT1, 2 and 3, which are involved in phosphatidylcholine biosynthesis, remodel phosphatidylethanolamine also; LPLAT1 utilizes lysophosphatidylethanolamine mainly and is specific for oleoyl-CoA. Some of these isoforms are confined to particular tissues, and there are independent acyltransferases in macrophages and other cells/tissues that transfer such fatty acids as arachidonic from phosphatidylcholine to ethanolamine-containing phospholipids. A further enzyme TLCD1 is an evolutionarily conserved lysophosphatidylethanolamine acyltransferase that takes part in this process. In lysosomes, lysophosphatidylethanolamine and lysophosphatidylcholine formed during degradation are exported into the cytosol by a transporter Spns1 for recycling.
3. Phosphatidylethanolamine – Biological Function
Physical properties: Although phosphatidylethanolamine has sometimes been equated with phosphatidylcholine in biological systems, there are significant differences in the chemistry and physics of these lipids, and they are utilized in different ways in membranes and biochemical processes. Both are crucial components of membrane bilayers and the ratio of the two may be a relevant factor, although phosphatidylethanolamine has a smaller head group, which gives the lipid a cone shape. On its own, it does not form bilayers but inverted hexagonal phases. With other lipids in a bilayer, it exerts a lateral pressure that modulates membrane curvature and stabilizes membrane proteins in their optimum conformations. It can hydrogen bond intermolecularly to adjacent lipids and to proteins through its polar head group, and the phosphate can be stabilized at the binding site by interactions with lysine and arginine side chains, or with hydroxyls from tyrosine side chains; there can be strong interactions too with the acyl chains of the phospholipid.
In contrast to phosphatidylcholine, phosphatidylethanolamine is concentrated with phosphatidylserine in the inner leaflet of the plasma membrane. On the other hand, it is present in both the inner and outer membranes of mitochondria, and in the former, it is present at much higher levels than in other organelles and facilitates membrane fusion and protein movement across membranes in addition to many other essential mitochondrial functions, including oxidative phosphorylation via the electron transport chain. It is a component of membrane contact sites between the endoplasmic reticulum and mitochondria. In eukaryotic cells in general, but especially those of insects, studies suggest that like cholesterol it increases the rigidity of the bilayer to maintain membrane fluidity, while in bacterial membranes, a primary role may be simply to dilute the high negative charge density of the anionic phospholipids.
Protein Interactions: Membrane proteins amount to 30% of the genome, and they are involved in transport, energy production, biosynthesis, signalling and communication. Within a membrane, most integral proteins consist of hydrophobic α-helical trans-membrane domains that zigzag across it and are connected by hydrophilic loops. Of those parts of the proteins outwith the bilayer, positively charged residues are much more abundant on the cytoplasmic side of membrane proteins as compared to the trans side (the positive-inside rule). Phosphatidylethanolamine may inhibit location of negative amino acids on the cytoplasmic side, and it has an appropriate charge density to balance that of the membrane surface and the protein, supporting the positive-inside rule. However, it can permit the presence of negatively charged residues on the cytosolic surface in some circumstances in support of protein function. When transported to the outer leaflet, it can bind to annexin V where it may inhibit blood coagulation by competing for phosphatidylserine binding.
Phosphatidylethanolamine is vital for mitochondrial efficiency, as demonstrated by defects in the operation of oxidative phosphorylation in the absence of phosphatidylserine decarboxylase PSD1 and by the role of the lipid in the regulation of mitochondrial dynamics in general but especially in the biogenesis of proteins in the outer mitochondrial membrane. Synthesis of phosphatidylethanolamine in the inner membrane of mitochondria is critical for the cytochrome bc1 complex (III), where there is a conserved binding site for the lipid in a specific subunit.
 Phosphatidylethanolamine
binds non-covalently to a superfamily of cytosolic proteins with multiple functions termed 'PE-binding proteins'.
While four members have been identified in mammals (PEBP1 to 4), more than 400 members of the family that are conserved during evolution
are known from bacteria to higher eukaryotes, and are required for such tasks as lipid binding, neuronal development, serine protease inhibition
and the regulation of several signalling pathways.
Of these, PEBP4 is of particular interest as it is highly expressed in many different cancers and can increase their resistance to therapy,
while the transport protein Vps13-like protein BLTP2 has comparable effects in breast cancer.
In animal tissues, phosphatidylethanolamine is important in the sarcolemmal membranes of the heart during ischemia, and
it participates in secretion of the nascent very-low-density lipoproteins (VLDL) from liver and in membrane fusion and fission.
It is required by the Ca2+-ATPase in that one molecule of phosphatidylethanolamine
is bound in a cavity between two transmembrane helices and acts as a wedge to keep them apart.
This is displaced when Ca2+ is bound to the enzyme.
Many other proteins bind non-covalently to phosphatidylethanolamine in a comparable way, including rhodopsin and aquaporins.
PE-binding proteins in plants regulate including shoot growth, flowering, inflorescence architecture, seed development and nodulation.
Phosphatidylethanolamine
binds non-covalently to a superfamily of cytosolic proteins with multiple functions termed 'PE-binding proteins'.
While four members have been identified in mammals (PEBP1 to 4), more than 400 members of the family that are conserved during evolution
are known from bacteria to higher eukaryotes, and are required for such tasks as lipid binding, neuronal development, serine protease inhibition
and the regulation of several signalling pathways.
Of these, PEBP4 is of particular interest as it is highly expressed in many different cancers and can increase their resistance to therapy,
while the transport protein Vps13-like protein BLTP2 has comparable effects in breast cancer.
In animal tissues, phosphatidylethanolamine is important in the sarcolemmal membranes of the heart during ischemia, and
it participates in secretion of the nascent very-low-density lipoproteins (VLDL) from liver and in membrane fusion and fission.
It is required by the Ca2+-ATPase in that one molecule of phosphatidylethanolamine
is bound in a cavity between two transmembrane helices and acts as a wedge to keep them apart.
This is displaced when Ca2+ is bound to the enzyme.
Many other proteins bind non-covalently to phosphatidylethanolamine in a comparable way, including rhodopsin and aquaporins.
PE-binding proteins in plants regulate including shoot growth, flowering, inflorescence architecture, seed development and nodulation.
The content of phosphatidylethanolamine in newly secreted VLDL particles and in apo B-containing particles isolated from the lumen of the Golgi is much higher than that in circulating VLDLs, suggesting that this lipid participates in VLDL assembly and/or secretion, although it is rapidly and efficiently removed from VLDL in the circulation. With lipid droplets in cells, phosphatidylethanolamine promotes coalescence of smaller droplets into larger ones; its synthesis is increased in brown adipocytes (and in liver) during cold exposure.
Although the mechanism has yet to fully elucidated, effects on protein conformation may be behind a finding that phosphatidylethanolamine is the primary factor in brain required for the propagation and infectivity of mammalian prions. Host defence peptides are antimicrobial agents produced by both prokaryotic and eukaryotic organisms, and many of these have a high affinity for phosphatidylethanolamine as a lipid receptor to modulate their activities. The peptide antibiotics cinnamycin and duramycins from Streptomyces have a hydrophobic pocket that fits around phosphatidylethanolamine such that the binding is stabilized by ionic interaction between the ethanolamine group of the lipid and the carboxylate moiety of the peptide to form a complex that inhibits Gram-positive organisms such as Bacillus species.
Autophagy and ferroptosis: Ubiquitin family members conjugated to phosphatidylethanolamine are present in yeast and mammalian cells, and ubiquitinated-PE accumulates at endosomes and the vacuole (or lysosomes). The most studied example is a covalent conjugate with a protein designated 'Atg8', formed by the action of cysteine protease ATG4 of the caspase family, that influences the process of autophagy (controlled degradation of cellular components) in yeast by promoting the formation of membrane vesicles containing the components to be degraded; phosphatidylinositol 3-phosphate is essential to the process. Oxidatively modified phosphatidylethanolamine is a factor in ferroptosis, a form of apoptosis in which disturbances to iron metabolism lead to an accumulation of hydroperoxides.
Bacteria: Much of the evidence for the unique properties of phosphatidylethanolamine comes from studies of the biochemistry of the bacterium E. coli, where this lipid is a major component of the membranes. Gram-negative bacteria have two membrane bilayers in the cell wall (see our web page on lipid A), and as much 90% of the phospholipid in the inner leaflet of the outer membrane is phosphatidylethanolamine, together with a high proportion in the cytoplasmic leaflet of the inner membrane. Phosphatidylethanolamine is required to support transport of lactose across membranes by the enzyme lactose permease, and other transport systems may require or be stimulated by it. There is evidence that it acts as a 'chaperone' during the assembly of this and other membrane proteins to guide the folding path for the proteins and to aid in the transition from the cytoplasmic to the membrane environment, and in its absence, the transport membranes may not have the correct tertiary structure and so will not work properly. Whether the lipid is required once the protein is correctly assembled is not fully understood in all cases, but it may be needed to orient enzymes correctly in the inner membrane. In contrast, it can inhibit folding of some multi-helical proteins. Some studies suggest that life in this organism can be maintained without phosphatidylethanolamine, but that crucial processes are inhibited.
Phosphatidylethanolamines containing polyunsaturated fatty acids support resolution of inflammation by modulating the macrophage lipidome and attenuating the responses to stimulation by lipopolysaccharides. Francisella tularensis bacteria, the cause of tularemia, suppresses host inflammation and the immune response when infecting mouse cells, because of its content of distinctive phosphatidylethanolamine containing 10:0 and 24:0 fatty acids. As the synthetic lipid produces the same effects in vitro in human cells infected with dengue fever virus, it is hoped that this lipid will prove to be a potent anti-inflammatory therapeutic agent. Akkermansia muciniphila, a bacterial species from human gut, produces phosphatidylethanolamine with two different branched chain components (anteiso-15:0 and iso-15:0) that has remarkable specificity for immune signalling in its host via a toll-like receptor TLR2-TLR1 heterodimer.
Precursor of other lipids: Phosphatidylethanolamine is a precursor for the synthesis of N-acyl-phosphatidylethanolamine (see below) and thence of anandamide (N‑arachidonoylethanolamine), and the plasmalogen form is the precursor for the 1-alkenyl phosphatidylcholine. In liver, it is the substrate for the hepatic enzyme phosphatidylethanolamine N-methyltransferase, which provides about a third of the phosphatidylcholine. It is the donor of ethanolamine phosphate during the synthesis of the glycosylphosphatidylinositol anchors that attach many signalling proteins to the surface of the plasma membrane, and in bacteria, it is used in the same way for the biosynthesis of lipid A and other lipopolysaccharides.
Other miscellaneous functions: Phosphatidylethanolamine is the precursor of an ethanolamine phosphoglycerol moiety bound to two conserved glutamate residues in the eukaryotic translation elongation protein elongation factor 1A, which mediates the recruitment of aminoacyl-tRNA to ribosomes during protein synthesis. This unique modification is required for many purposes, such as cell growth and proliferation, including cytoskeleton organization, and signal transduction, while in plants, it is of importance for resistance to attack by pathogens.
Changes in the concentrations of phosphatidylethanolamine or of its fatty acid or molecular species compositions in plasma and other tissues have been reported in various disease states, where they may serve as biomarkers. These include diseases of the liver, cardiovascular system, kidney and brain, as well as cancer, sepsis and diabetes, and increased concentrations of phosphatidylethanolamine and its lyso form may be useful for predicting the progression of mild cognitive impairment. In addition, dysregulated metabolism of phosphatidylethanolamine is a factor in such lung diseases as asthma, pulmonary fibrosis and chronic obstructive pulmonary disease.
4. Lysophosphatidylethanolamine
 Lysophosphatidylethanolamine (LPE), with one mole of fatty acid per mole of lipid, is found in trace
amounts only in animal tissues, other than plasma (10 to 50µM, or ~1% of total serum phospholipids), and it is formed mainly by hydrolysis of
phosphatidylethanolamine by the enzyme phospholipase A2 as part of the de‑acylation/re-acylation cycle that controls its overall
molecular composition as discussed above.
There are reports of the participation of LPE in cellular functions, such as differentiation and migration of certain neuronal and bone cells
but also of various cancer cells.
Some of these effects may depend on the nature of the acyl group, and for example, oleoyl-LPE in brain stimulates neurite outgrowth and protects
against glutamate toxicity, while greatly reduced levels of 22:6-LPE have been observed in post-mortem brains from patients with Alzheimer's
disease in comparison to controls.
Administration of lysophosphatidylethanolamine to aging rats improves diastolic dysfunction by alleviating mitochondrial injury.
An alkenyl form (18:1) of LPE has been identified as a signalling molecule that responds to inflammatory stimuli by transcriptional regulation
of the nuclear hormone receptor RORγt (retinoic-related orphan receptor gamma).
Some further biological effects have been reported in animal tissues in vitro, and it may be an agonist for the G-protein-coupled
receptor for lysophosphatidic acid LPAR1.
Lysophosphatidylethanolamine (LPE), with one mole of fatty acid per mole of lipid, is found in trace
amounts only in animal tissues, other than plasma (10 to 50µM, or ~1% of total serum phospholipids), and it is formed mainly by hydrolysis of
phosphatidylethanolamine by the enzyme phospholipase A2 as part of the de‑acylation/re-acylation cycle that controls its overall
molecular composition as discussed above.
There are reports of the participation of LPE in cellular functions, such as differentiation and migration of certain neuronal and bone cells
but also of various cancer cells.
Some of these effects may depend on the nature of the acyl group, and for example, oleoyl-LPE in brain stimulates neurite outgrowth and protects
against glutamate toxicity, while greatly reduced levels of 22:6-LPE have been observed in post-mortem brains from patients with Alzheimer's
disease in comparison to controls.
Administration of lysophosphatidylethanolamine to aging rats improves diastolic dysfunction by alleviating mitochondrial injury.
An alkenyl form (18:1) of LPE has been identified as a signalling molecule that responds to inflammatory stimuli by transcriptional regulation
of the nuclear hormone receptor RORγt (retinoic-related orphan receptor gamma).
Some further biological effects have been reported in animal tissues in vitro, and it may be an agonist for the G-protein-coupled
receptor for lysophosphatidic acid LPAR1.
In plants, lysophosphatidylethanolamine is an inhibitor of phospholipase D, a key enzyme in the degradation of membrane phospholipids during the early stages of plant senescence, where it retards the senescence of leaves, flowers and post-harvest fruits. Indeed, it has horticultural applications when applied externally, e.g., to stimulate ripening and extend the shelf-life of fruit, delay senescence and increase the vase life of cut flowers.
In bacteria, lysophosphatidylethanolamine displays chaperone-like properties to promote the folding of citrate synthase and other enzymes. With other lysophospholipids, it is produced in the envelope membranes by many different endogenous and exogenous factors and must be transported back into the bacterial cell by flippases for reconversion to the diacyl forms by the action of a peripheral enzyme, acyl-ACP synthetase/LPL acyltransferase. Lysophosphatidylethanolamines produced by certain bacteria act synergistically with sulfonolipid rosette-inducing factors (RIFs) to induce choanoflagellates to move from a unicellular to a multicellular state.
5. N-Acyl Phosphatidylethanolamine
 N-Acyl phosphatidylethanolamine with the free amino group linked to a further fatty acid has been detected in rather small
amounts in several animal tissues (~0.01%), including the brain, nervous tissues and epidermis, when the N-acyl chain is often palmitic
or stearic acid (human plasma: N16:0-PE (40%), N18:1-PE (23.3%), N18:0-PE (19%), N18:2-PE (16.6%) and N20:4-PE (1.4%)).
In animals, it is the precursor of anandamide (see our web pages on
endocannabinoids for a more detailed discussion of N‑acyl phosphatidylethanolamine
synthesis and metabolism) and of other acyl ethanolamides (e.g.,
N‑oleoylethanolamine) in brain, the intestines and other tissues.
In brief, it is formed biosynthetically by the action of a transferase, such as cytosolic phospholipase A2ε
(Ca2+-dependent), which exchanges a fatty acid from the sn-1 position of a phospholipid (probably phosphatidylcholine) to
the primary amine group of phosphatidylethanolamine without a hydrolytic step and with both diacyl- and alkenylacyl-forms of
phosphatidylethanolamine serving as acceptors.
In addition, some transfer can occur from phosphatidylethanolamine per se by an intramolecular reaction.
N-Acyl phosphatidylethanolamine with the free amino group linked to a further fatty acid has been detected in rather small
amounts in several animal tissues (~0.01%), including the brain, nervous tissues and epidermis, when the N-acyl chain is often palmitic
or stearic acid (human plasma: N16:0-PE (40%), N18:1-PE (23.3%), N18:0-PE (19%), N18:2-PE (16.6%) and N20:4-PE (1.4%)).
In animals, it is the precursor of anandamide (see our web pages on
endocannabinoids for a more detailed discussion of N‑acyl phosphatidylethanolamine
synthesis and metabolism) and of other acyl ethanolamides (e.g.,
N‑oleoylethanolamine) in brain, the intestines and other tissues.
In brief, it is formed biosynthetically by the action of a transferase, such as cytosolic phospholipase A2ε
(Ca2+-dependent), which exchanges a fatty acid from the sn-1 position of a phospholipid (probably phosphatidylcholine) to
the primary amine group of phosphatidylethanolamine without a hydrolytic step and with both diacyl- and alkenylacyl-forms of
phosphatidylethanolamine serving as acceptors.
In addition, some transfer can occur from phosphatidylethanolamine per se by an intramolecular reaction.
Under conditions of degenerative stress, N-acyl phosphatidylethanolamine can accumulate in significant amounts as the result of ischemic injury, infarction or cancer, and it may be involved in neuroprotection, anti-inflammation and membrane stabilization; it is a signalling molecule that stimulates lactate efflux by monocarboxylate transporters in membranes. After feeding a high fat diet to rats, it is present in plasma and can cross into the brain where it accumulates in the hypothalamus. It should be noted that some N-acyl phosphatidylethanolamine can be formed artefactually because of faulty extraction procedures during analysis.
In plants, N-acyl phosphatidylethanolamine is a common constituent of cereal grains (e.g., wheat, barley and oats) and of some other seeds (1.9% of the phospholipids of cotton seeds but 10-12% of oats), but in other plant tissues, it is detected most often under conditions of physiological stress when it is hydrolysed with release of N-acyl ethanolamides and phosphatidic acid. In contrast to animals, synthesis is by direct acylation of phosphatidylethanolamine with a free fatty acid catalysed by a membrane-bound transferase in a reverse serine-hydrolase catalytic mechanism, although lyso‑N-acyl-phosphatidylethanolamines have been detected in tomato plants as possible intermediates. Both N-acyl lipid classes have been implicated in such physiological processes as the elongation of main and lateral roots, regulation of seed germination, seedling growth and defence from attacks by pathogens.
N-Acyl phosphatidylethanolamine has been found in many microorganisms, and N-acetyl phosphatidylethanolamine has been detected in a filamentous fungus, Absidia corymbifera, where it comprised 6% of the total membrane lipids and was accompanied by an even more unusual lipid 1,2‑diacyl-sn-glycero-3-phospho(N-ethoxycarbonyl)-ethanolamine. The Gram-positive bacterium Bacillus subtilis contains aminoacyl phosphatidylethanolamines with alanine or lysine attached to the ethanolamine moiety.
6. Mono- and Dimethylphosphatidylethanolamines
Mono- and dimethylphosphatidylethanolamines are formed by sequential methylation of phosphatidylethanolamine by the enzyme phosphatidylethanolamine N‑methyltransferase as intermediates in one of the mechanisms for the biosynthesis of phosphatidylcholine. This is a minor pathway in general in animals, although it is significant in liver when choline is deficient in the diet, but it is the major route in yeasts and bacteria, although these intermediate lipids do not seem to be vital components of yeast membranes.

They are never found at greater than trace levels in animal or plant tissues, and as might be expected, they are more abundant in many bacteria that interact with plants.
7. Non-Enzymatic Modification of Phosphatidylethanolamine by Carbonyl-Amine Reactions
Michael reaction: Phosphatidylethanolamine can react non-enzymatically to form imine and/or Michael adducts with the hydroxy-alkenals and related compounds that are products of hydroperoxidation of unsaturated fatty acids, and include malondialdehyde, acrolein, epoxyalkenals, hydroxyalkenals (e.g., 4‑hydroxy-trans-2-nonenal (HNE)), oxoalkenals and γ‑ketoaldehydes. The chemistry and biochemistry of bioactive aldehydes is discussed in more detail in a separate web page. As an example, the reaction with glycoxal is illustrated.
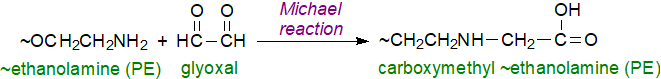 |
| Figure 3. Michael reaction of phosphatidylethanolamine with glycoxal (partial structures illustrated). |
The comparable adduct with HNE can react further to generate toxic pyrroles, but it can also be hydrolysed by N-acyl phosphatidylethanolamine hydrolysing phospholipase D to generate phosphatidic acid and the amine adduct. Related adducts in which the headgroup nitrogen has been enzymatically modified with acyl chains of four carbons or longer also react in this way. This enzyme has an established role in the biosynthesis of N-acyl-ethanolamines, and it seems that it may be important for clearing some products of lipid peroxidation.
 |
| Figure 4. Hydrolysis of the HNE adduct of phosphatidylethanolamine by N-acyl phosphatidylethanolamine hydrolysing phospholipase D (NAPE‑PLD). |
Such Michael products accelerate the peroxidation of membrane lipids, and they are factors in the generation of oxidative stress both in foods and in tissues. In vivo, they are inflammatory mediators and they have been implicated in a several disease states, such as atherogenesis and diabetes, and during aging. Alkyl-modified phosphatidylethanolamines induce a negative membrane curvature in lipid vesicles in vitro, and they have the potential to modify membrane properties of membrane transporters, channels, receptors and enzymes under conditions of oxidative stress.
In the visual cycle, phosphatidylethanolamine reacts with all-trans-retinal in the photoreceptor outer segment membrane of the eye to form N‑retinylidene-phosphatidylethanolamine as part of a transport mechanism and a means of preventing non-specific aldehyde reactions. Normally, the trans-retinal is regenerated for reuse, but the conjugate can sometimes react further to generate a stable bis-retinoid condensation product (illustrated).

This lipid conjugate, together with a 1-alkyl-lysophosphatidylethanolamine analogue and hydrolysis products formed by cleavage of the ethanolamine-phosphate bond by phospholipase D, can accumulate in retinal pigment epithelial cells with age, where it can be involved in the pathogenesis of retinal disorders such as Stargardt disease.
Levuglandins and isolevuglandins (oxylipins and γ-keto aldehydes) are reactive cyclo-oxygenase metabolites of arachidonic and docosahexaenoic acids, which react rapidly with the free amine group of phosphatidylethanolamine (and with proteins) in vivo to form cytotoxic hydroxy-lactam derivatives.
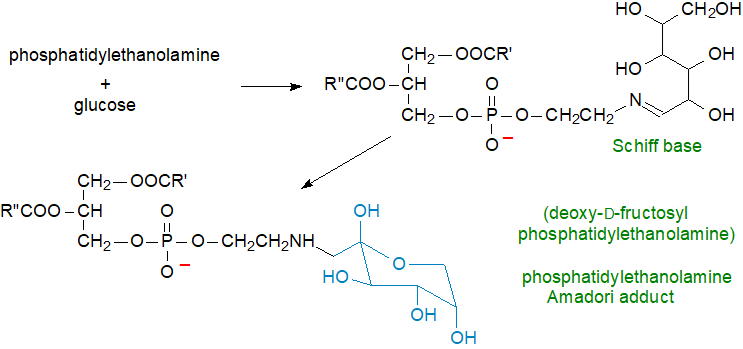
Schiff base formation: Phosphatidylethanolamine reacts with glucose and other sugars to form unstable Schiff bases, which rearrange to produce Amadori products of phosphatidylethanolamine, as illustrated below for glucose with formation of deoxy-D-fructosyl-phosphatidylethanolamine, especially under hyperglycaemic conditions (Maillard reaction). Indeed, there are suggestions that Amadori-phosphatidylethanolamine may be a useful predictive marker for hyperglycaemia in the early stages of diabetes.
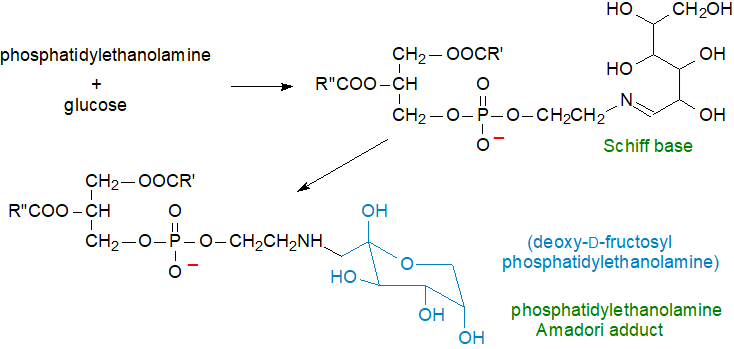 |
| Figure 5. Formation of Amadori adduct of phosphatidylethanolamine by reaction with glucose. |
Once Amadori-phosphatidylethanolamine is formed, it can undergo further reactions, for example with reactive oxygen to form carboxymethyl- and carboxyethyl-adducts, which have the potential to trigger pathological processes, including the neuropathy and retinopathy associated with diabetic complications. Thus, it has been demonstrated that glycated phosphatidylethanolamine and its oxidation products induce the production of pro-inflammatory cytokines and that they have a role in apoptotic cell signalling. Unsurprisingly, such changes to the phospholipid head group change the biophysical properties of membrane bilayers appreciably and can lead ultimately to the disintegration of cell membranes. Phosphatidylserine might be expected to form analogous adducts, but these have proved harder to detect in tissues in vivo.
8. Phosphatidylethanol
 Phosphatidylethanol has little in common with phosphatidylethanolamine other than the obvious structural similarity.
It is formed slowly in cell membranes, notably those of erythrocytes, by a transphosphatidylation reaction from phosphatidylcholine and ingested
ethanol catalysed by the enzyme phospholipase D.
As it has a long half-life in serum relative to alcohol per se, potentially for 2-3 weeks, it is a useful biochemical and forensic
marker for alcohol abuse; chronic alcoholics have much higher levels in the blood than healthy subjects who consume alcohol in moderation.
While this lipid is usually considered to be physiologically inert, there is evidence with Drosophila that it affects lipid-regulated
potassium channels.
There are suggestions that it may be beneficial towards ethanol tolerance but harmful for colon cancer.
Phosphatidylethanol has little in common with phosphatidylethanolamine other than the obvious structural similarity.
It is formed slowly in cell membranes, notably those of erythrocytes, by a transphosphatidylation reaction from phosphatidylcholine and ingested
ethanol catalysed by the enzyme phospholipase D.
As it has a long half-life in serum relative to alcohol per se, potentially for 2-3 weeks, it is a useful biochemical and forensic
marker for alcohol abuse; chronic alcoholics have much higher levels in the blood than healthy subjects who consume alcohol in moderation.
While this lipid is usually considered to be physiologically inert, there is evidence with Drosophila that it affects lipid-regulated
potassium channels.
There are suggestions that it may be beneficial towards ethanol tolerance but harmful for colon cancer.
9. Analysis
Analysis of phosphatidylethanolamine and related lipids is straightforward, as they are readily isolated by thin-layer or high-performance liquid chromatography methods for further analysis, for example by mass spectrometry, but analysis of adducts of phosphatidylethanolamine is much more challenging.
Suggested Reading
- Chakrabarti, A. Phospholipid asymmetry in biological membranes: is the role of phosphatidylethanolamine underappreciated? J. Membrane Biol., 254, 127-132 (2021); DOI.
- Coulon, D., Faure, L., Salmon, M., Wattelet, V. and Bessoule, J.J. Occurrence, biosynthesis and functions of N-acylphosphatidylethanolamines (NAPE): Not just precursors of N-acylethanolamines (NAE). Biochimie, 94, 75-85 (2012); DOI.
- Dawaliby, R., Trubbia, C., Delporte, C., Caroline Noyon, C., Ruysschaert, J.-M., Van Antwerpen, P. and Govaerts, C. Phosphatidylethanolamine is a key regulator of membrane fluidity in eukaryotic cells. J. Biol. Chem., 291, 3658-3667 (2016); DOI.
- Di Bartolomeo, F., Wagner, A. and Daum, G. Cell biology, physiology and enzymology of phosphatidylserine decarboxylase. Biochim. Biophys. Acta, Lipids, 1862, 25-38 (2017); DOI.
- Dowhan, W. and Bogdanov, M. Eugene P. Kennedy's legacy: defining bacterial phospholipid pathways and function. Front. Mol. Biosci., 8, 666203 (2021); DOI.
- Kang, J.H., Toita, R., Kawano, T., Murata, M. and Kano, A. Phospholipids and their metabolites as diagnostic biomarkers of human diseases. Prog. Lipid Res., 99, 101340 (2025); DOI.
- Phoenix, D.A., Harris, F., Mura, M. and Dennison, S.R. The increasing role of phosphatidylethanolamine as a lipid receptor in the action of host defence peptides. Prog. Lipid Res., 59, 26-37 (2015); DOI.
- Plouzennec, S., de la Barca, J.M.C. and Chevrollier, A. The role of phospholipids in mitochondrial dynamics and associated diseases. Front. Biosci.-Landmark, 30, 27634 (2025); DOI.
- Pohl, E.E. and Jovanovic, O. The role of phosphatidylethanolamine adducts in modification of the activity of membrane proteins under oxidative stress. Molecules, 24, 4545 (2019); DOI.
- Ridgeway, N.D. and McLeod, R.S. (Editors) Biochemistry of Lipids, Lipoproteins and Membranes. 6th Edition. (Elsevier, Amsterdam) (2016) - several chapters - see Science Direct - there is now a 7th edition.
- Rohricht, M., Paschke, K., Sack, P.M., Weinmann, W., Thomasius, R. and Wurst, F.M. Phosphatidylethanol reliably and objectively quantifies alcohol consumption in adolescents and young adults. Alcoholism, Clin. Exp. Res., 44, 2177-2186 (2020); DOI.
- Sakamaki, J., Ode, K.L., Kurikawa, Y., Ueda, H.R., Yamamoto, H. and Mizushima, N. Ubiquitination of phosphatidylethanolamine in organellar membranes. Mol. Cell, 82, 3677-3692.e (2022); DOI.
- St Germain, M., Iraji, R. and Bakovic, M. Phosphatidylethanolamine homeostasis under conditions of impaired CDP-ethanolamine pathway or phosphatidylserine decarboxylation. Front. Nutr., 9, 1094273 (2023); DOI.
- Teng, Y.Y., Xue, H.P., Deng, X.L., Luo, Y.Q. and Wu, T. The role of phosphatidylethanolamine-binding protein (PEBP) family in various diseases: Mechanisms and therapeutic potential. Progr Biophys. Mol. Biol., 196, 102-113 (2025); DOI.
- Van der Veen, J.N., Kennelly, J.P., Wan, S., Vance, J.E., Vance, D.E. and Jacobs, R.L. The critical role of phosphatidylcholine and phosphatidylethanolamine metabolism in health and disease. Biochim. Biophys. Acta, Biomembranes, 1859, 1558-1572 (2017); DOI.
- Wellner, N., Diep, T.A., Janfelt, C. and Hansen, H.S. N-Acylation of phosphatidylethanolamine and its biological functions in mammals. Biochim. Biophys. Acta, Lipids, 1831, 652-662 (2013); DOI.
- Xu, T., Molday, L.L. and Molday, R.S. Retinal-phospholipid Schiff-base conjugates and their interaction with ABCA4, the ABC transporter associated with Stargardt disease. J. Biol. Chem., 299, 104614 (2023); DOI.
- and of interest from a historical perspective-
- Vance, J.E. Historical perspective: phosphatidylserine and phosphatidylethanolamine from the 1800s to the present. J. Lipid Res., 59, 923-944 (2018); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: November 2025 | ||
© The LipidWeb is open access and fair use is encouraged but not text and data mining, AI training, and similar technologies.
