Endocannabinoids:
Anandamide, 2‑Arachidonoylglycerol and Related Lipids
 Long-chain N-acylethanolamides have important metabolic and pharmacological
properties and are ubiquitous minor constituents of animal and human cells, tissues and body fluids.
After its discovery in 1992, N‑arachidonoylethanolamine or anandamide attracted special interest
when it was found to have a similar neurological impact to that of some phytocannabinoids, i.e., the bioactive compounds in marijuana
or cannabis (from Cannabis sativa), mainly Δ9‑tetrahydrocannabinol and cannabidiol ('ananda' means inner bliss and
tranquillity in Sanskrit).
They interact with the same G protein-coupled receptors to produce comparable physiological results in spite of having no apparent structural
relationship.
Long-chain N-acylethanolamides have important metabolic and pharmacological
properties and are ubiquitous minor constituents of animal and human cells, tissues and body fluids.
After its discovery in 1992, N‑arachidonoylethanolamine or anandamide attracted special interest
when it was found to have a similar neurological impact to that of some phytocannabinoids, i.e., the bioactive compounds in marijuana
or cannabis (from Cannabis sativa), mainly Δ9‑tetrahydrocannabinol and cannabidiol ('ananda' means inner bliss and
tranquillity in Sanskrit).
They interact with the same G protein-coupled receptors to produce comparable physiological results in spite of having no apparent structural
relationship.
Three years later, it was recognized that 2‑arachidonoylglycerol has very similar functions to anandamide and that it interacts with the same receptors, although it is produced by a very different biosynthetic route and is much more abundant in tissues. Together, anandamide and 2‑arachidonoylglycerol have been termed endogenous cannabinoids or 'endocannabinoids', as their receptors were first identified through their interactions with phytocannabinoids, shortly before the discovery of the endogenous ligands. These lipids are synthesised from membrane lipids on demand in response to increased intracellular Ca2+ concentration, membrane depolarization and/or receptor stimulation. The signalling process is rapidly attenuated by their catabolism, i.e., there is tight regulation of the endocannabinoid system.
Some other simple amides, including oleamide and arachidonoyldopamine, are now known to interact with the same receptors as anandamide and 2‑arachidonoylglycerol so can be classified as endocannabinoids. On the other hand, while some authors place palmitoylethanolamine and other acyl ethanolamides in this category, there is no evidence for an interaction with these receptors, so they are discussed in the web page dealing with other simple amides, some of which share the synthetic and degradative pathways of anandamide and thus may affect it indirectly ('entourage effects'). Arguably, the definition of an endocannabinoid could be widened, as they are now known to affect a greater range of G protein-coupled receptors than was initially thought, and for example, ethanolamides of some polyunsaturated fatty acids of the omega-3 family are borderline candidates for endocannabinoid status. This wider family and their various regulators have been termed the “endocannabinoidome”, while oxygenated metabolites have been designated the “oxyendocannabinoidome”. For reasons of practical convenience, I have tried to adhere to a relatively strict definition of endocannabinoids in this web page, i.e., those lipids that interact with the primary cannabinoid/endocannabinoid receptors (CB1 and CB2 - see below).
Cellular concentrations of endocannabinoids depend on a balance between synthesis and degradation by multiple regulated enzymes, and they exist in dynamic equilibria with many other lipid-derived mediators, including eicosanoids and other oxylipins, as part of a versatile system for fine-tuning different physiological and metabolic processes, including inflammation, in a tissue- or cell-specific manner. Indeed, many of these general lipid signalling pathways have aspects that are conserved evolutionarily in most eukaryotes, including primitive organisms such as hydra, black truffles and Tetrahymena.
1. N-Arachidonoylethanolamine (Anandamide)
Anandamide is synthesised in all animals, including primitive nematodes and parasitic helminths and is produced upon demand from phospholipid precursors in cell membranes in almost all cells and tissues in response to a rise in intra-cellular calcium levels. In the more reliable recent analyses, basal levels in tissues are estimated to lie in the picomolar to low nanomolar range.
Biosynthesis: Although direct N-acylation of ethanolamine is possible, the main mechanism for the biosynthesis of anandamide and related amides requires as a first step the conversion of phosphatidylethanolamine (PE) to N-acyl-phosphatidylethanolamine, a lipid that is normally present in animal tissues at very low levels other than during injury. Unusual transacylase reactions rather than hydrolysis and re-synthesis via CoA esters are involved, and only 1-O-acyl groups of phospholipids can serve as N-acyl donors, mainly by a reaction that is energy-independent with a direct transfer of the fatty acids of position sn-1 of phosphatidylcholine (PC) to N‑acylate phosphatidylethanolamine by N-acyltransferases (Ca2+-dependent or -independent), with concomitant formation of lyso-phosphatidylcholine. The first of these N-acyltransferases is now known to be the ε isoform of cytosolic phospholipase A2 (cPLA2ε (PLA2G4E), Ca2+-dependent), although other isoforms are known including one specific for the plasmalogen form of phosphatidylethanolamine. A second related mechanism is an intramolecular transfer of the fatty acid in position sn‑1 of phosphatidylethanolamine by phospholipase/acyltransferase 1-5 (PLAAT1-5, Ca2+‑independent) to form a hypothetical intermediate, 2‑O‑acyl-sn-glycero-3-phospho-(N‑acyl)-ethanolamine (or N-acyl-lysoPE), which is then reacylated in position 1 by an analogous transfer reaction.
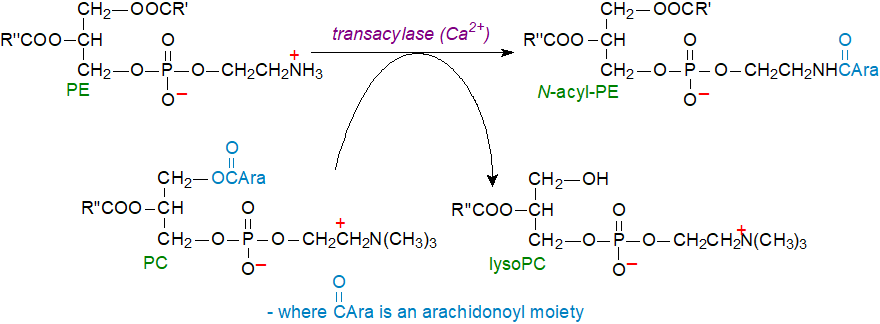 |
| Figure 1. Biosynthesis of the precursor in anandamide biosynthesis, N-acyl-phosphatidylethanolamine. |
The high specificity of this reaction might be considered surprising in view of the fact that the arachidonic acid levels in position 1 of phospholipids are usually very low (typically <0.3%) other than in testis. In neurons, there is evidence that 1,2-diarachidonoyl-phosphatidylcholine produced by acylation of 2‑arachidonoyl-lysophosphatidylcholine is the required intermediate. This is the key to the specificity of the process since subsequent reactions are independent of the N-acyl substituent.
The second step in the biosynthesis of anandamide and related amides is hydrolysis of the N‑acyl-phosphatidylethanolamine by an N‑acyl phosphatidylethanolamine-specific phospholipase D-like hydrolase (NAPE-PLD), which acts in the same way as phospholipase D, but differs from all others of this type in its amino acid sequence. It has a hydrophobic cavity that provides an entryway for N‑acyl-phosphatidylethanolamine into the catalytic site, where uniquely bile acids are bound with high affinity to enhance dimer assembly and enable catalysis. Phosphatidic acid (PA) is formed as a by-product and also is a signalling mediator.
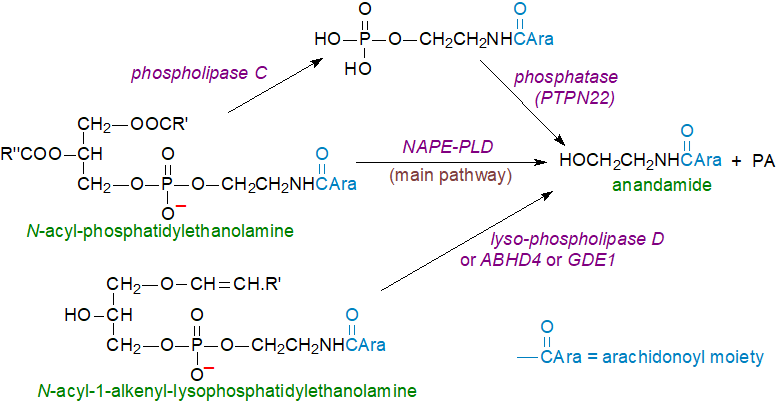 |
| Figure 2. Final step in the biosynthesis of anandamide. |
A second pathway has been described that does not use NAPE-PLD but rather either single or double O-deacylation of N‑acyl- or N‑alkenyl-phosphatidylethanolamine catalysed by a phospholipase, sPLA2 or by ABHD4 - a member of the 'serine hydrolase' family, prior to the action of lysophospholipase D isoforms 1/3 (GDPD1,3) or a metal-dependent glycerophosphodiester phosphodiesterase 1 (GDE1) on the resulting lysoglycerophospho- or glycerophospho-N-arachidonoylethanolamines. Indeed, it has been suggested that this may be the major route to anandamide from plasmalogens in brain, where the intermediate alkenyl-glycerophospho-N-acylethanolamines have been detected in a mouse model. In rodent brain, the endogenous precursor of anandamide is mainly the plasmalogen form of N‑arachidonoyl-phosphatidylethanolamine (N‑acylplasmenylethanolamine) and contains alkenyl groups (16:0, 18:0, 18:1) in position sn-1 and mono- (18:1) and polyunsaturated (20:4, 22:4, 22:6) acyl groups in position sn-2 of the glycerol backbone.
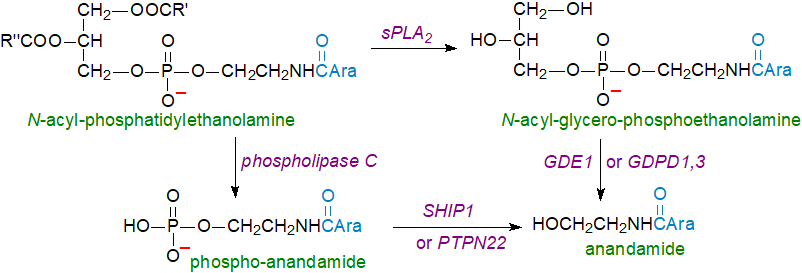 |
| Figure 3. Alternative routes for biosynthesis of anandamide. |
Yet another pathway is known in which N-acyl-phosphatidylethanolamine is again the main precursor but is acted upon by phospholipase C to release phospho-anandamide, which is then de-phosphorylated to anandamide by tyrosine-protein phosphatase non-receptor type 22 (PTPN22) or by phosphatidylinositol 3,4,5-trisphosphate 5-phosphatase 1 (SHIP1). This may be the main route when anandamide is required rapidly in response to bacterial endotoxins. Further biosynthetic pathways to anandamide are known to exist, depending upon tissue or cell type, and the relative contributions of these and the manner of their regulation pose complex questions. Evidently, a high degree of redundancy exists for the biosynthesis of anandamide and its precursors, and these may intersect those leading to the formation of the other endocannabinoids, confirming the vital requirements for these lipid mediators in animals.
Function: Anandamide and other endocannabinoids share many physical and functional properties. They are highly lipophilic and tend to remain in membranes, which they may cross by passive diffusion, though facilitated transport is possible, to encounter membrane-bound enzymes and receptors. Alternatively, they can diffuse into the cytoplasm, where they are transported, possibly by microvesicles or exosomes, or by fatty acid binding proteins, the fatty acid amide hydrolase (see below), or other lipid carriers to the cannabinoid receptors. Cytoplasmic lipid droplets may act as a reservoir that is accessible upon demand, and they are a site for metabolism. In plasma, anandamide binds reversibly to serum albumin and is presumably carried to other tissues in this form, although this has still to be established definitively, and transport by extracellular vesicles is another possibility. Biosynthesis, activation of receptors and degradation of endocannabinoids are highly regulated and are initiated by an increase in intracellular Ca2+.
 Although anandamide and other endocannabinoids have no structural relationships
to the constituent molecules of cannabis such as the phytocannabinoid Δ9‑tetrahydrocannabinol, they exert similar
effects at nanomolar to sub-micromolar concentrations mainly through interactions with the same cannabinoid
receptors, designated ‘CB1’ (or CB1 or CB1R) and ‘CB2’ (or CB2 or CB2R).
These are membrane-bound G proteins that contain seven transmembrane spanning domains and are generally coupled to heterotrimeric
G proteins of the Gαi and Gαo classes.
They are members of a large and diverse family of receptors, which convert extracellular stimuli into intracellular signals
and are present only in the chordate branch of the animal kingdom.
CB1 is found in the central nervous system and in some other organs, including the heart, uterus, testis, skin and small intestine,
while the CB2 receptor is found in the skin, spleen and other cells associated with immunochemical responses, and in activated glia cells
in brain.
Although their physiological properties are similar, the CB2 receptor has only 44% homology with CB1 (but 68% in the membrane-spanning region).
They are located mainly in the plasma membrane, although there are suggestions that there may be a limited presence in other organelles.
Although anandamide and other endocannabinoids have no structural relationships
to the constituent molecules of cannabis such as the phytocannabinoid Δ9‑tetrahydrocannabinol, they exert similar
effects at nanomolar to sub-micromolar concentrations mainly through interactions with the same cannabinoid
receptors, designated ‘CB1’ (or CB1 or CB1R) and ‘CB2’ (or CB2 or CB2R).
These are membrane-bound G proteins that contain seven transmembrane spanning domains and are generally coupled to heterotrimeric
G proteins of the Gαi and Gαo classes.
They are members of a large and diverse family of receptors, which convert extracellular stimuli into intracellular signals
and are present only in the chordate branch of the animal kingdom.
CB1 is found in the central nervous system and in some other organs, including the heart, uterus, testis, skin and small intestine,
while the CB2 receptor is found in the skin, spleen and other cells associated with immunochemical responses, and in activated glia cells
in brain.
Although their physiological properties are similar, the CB2 receptor has only 44% homology with CB1 (but 68% in the membrane-spanning region).
They are located mainly in the plasma membrane, although there are suggestions that there may be a limited presence in other organelles.
Within a given tissue, expression of a receptor can vary between different locations, and isoforms of the two receptors in humans and rodents have been identified that differ in their tissue distributions. It has become evident that endocannabinoid receptors exert a wide variety of different cellular effects by interactions with G protein sub-types, and that this is dependent on such factors as the origin of the cell, its subcellular location and physiological state. In turn, variation in the location of CB1-interacting proteins may have a role in the cell-specific modulation of endocannabinoid signalling. Anandamide is a high-affinity, partial agonist of CB1 but is relatively inert at CB2, while 2‑arachidonoylglycerol acts as a full agonist at both CB1 and CB2 with moderate-to-low affinity (see below). CB1 is the main receptor mediating the neurological consequences of cannabis use.
 Anandamide is known to
bind to several receptors other than CB1 and CB2, including the peroxisome proliferator-activated receptors (PPARα, β and γ)
to trigger metabolic, anti-inflammatory and neuroprotective responses.
By binding to PPARβ or PPARγ in brain, it may regulate memory and learning.
Endocannabinoids are now known to act as ligands for such receptors as the transient receptor potential vanilloid 1 (TRPV1
- a non-selective cation channel ubiquitously expressed in all tissues), GPR55, GPR119 and others, possibly including toll-like receptor 4 (TLR4).
Most of these are extracellular binding sites, but TRPV1 and related receptors are intracellular.
Anandamide is known to
bind to several receptors other than CB1 and CB2, including the peroxisome proliferator-activated receptors (PPARα, β and γ)
to trigger metabolic, anti-inflammatory and neuroprotective responses.
By binding to PPARβ or PPARγ in brain, it may regulate memory and learning.
Endocannabinoids are now known to act as ligands for such receptors as the transient receptor potential vanilloid 1 (TRPV1
- a non-selective cation channel ubiquitously expressed in all tissues), GPR55, GPR119 and others, possibly including toll-like receptor 4 (TLR4).
Most of these are extracellular binding sites, but TRPV1 and related receptors are intracellular.
Further levels of complexity in regulation comes from the interactions with endogenous modulators, and these include lipoxin A4, which increases the affinity for CB1, pregnenolone which inhibits CB1 signalling, and the peptide Pepcan-12, which acts as a negative allosteric modulator of CB1 but a positive allosteric modulator of CB2. Some anandamide properties may be mediated through the production of nitric oxide, which is a versatile signalling mediator and is ubiquitous in tissues, and there are suggestions that endocannabinoids may act in concert with other lipid signalling molecules such as sphingosine-1-phosphate.
As in many other membrane associated processes, lipid rafts and caveolae serve as platforms for regulation of the endocannabinoid system and especially in the modulation of binding and signalling of the CB1 receptor. Cholesterol in these membrane domains may have a role in mediating the interaction between anandamide and the receptor.
In brain, some types of neurons contain very high levels of CB1 receptor protein on synaptic terminals, whereas others have much lower levels with least in some regions of the hypothalamus and astroglial cells. The endocannabinoid system is a factor in the regulation of neuron production during embryonal development, and the CB1 receptor participates in the modulation of neuronal and glial proliferation, while CB2 assists the proliferation of glial cells. As with the phytocannabinoids, the endocannabinoids produce neurobehavioral symptoms and may have signalling roles in the central nervous system in the perception of pain, anxiety and fear, in the regulation of body temperature, and in the control of appetite. The main mechanism is via retrograde signalling in response to activation of post-synaptic neurons, when endocannabinoids are synthesised and released to stimulate pre-synaptic CB1 receptors and suppress the release of neurotransmitters. They are reported to be beneficial towards some neurological disorders, including Alzheimer's disease, Parkinson's disease, schizophrenia and some major depressive disorders, but not to all. Increased synthesis occurs during exercise, and this may contribute to healthy ageing. As there is evidence that glucocorticoids recruit endocannabinoid signalling, this may mediate many of the neurobiological and physiological actions of glucocorticoids.
Anandamide has anti-inflammatory and anti-cancer properties both in vivo and in vitro in animal models, and it can induce apoptosis in many cell types. It affects the cardiovascular system by inducing profound decreases in blood pressure and heart rate, and it is an anabolic regulator of metabolism in that it increases the intake of food, promotes the storage of lipid, decreases the expenditure of energy, and is a regulator of body temperature, locomotion, feeding and anxiety. There are suggestions that modulation of anandamide levels in the gut has potential for treatment of inflammatory bowel disease and colon cancer; it promotes cellular senescence in a human glioblastoma cell line. Anandamide is present in the reproductive fluids of both males and females and may be required for reproduction in relation to both the production of spermatozoa and female fertility and pregnancy. Macrophages generate anandamide in response to the presence of bacterial endotoxin, and it plays a part in the pathology of septic shock and cirrhosis of the liver. In this manner, anandamide is anti-inflammatory both in the peripheral and central nervous system. Anandamide (and 2‑arachidonoylglycerol and perhaps other long-chain ethanolamides) are able to modulate several antioxidant enzymes and control redox homeostasis through targeting the cannabinoid and other receptors.
 O-Arachidonoylethanolamine, i.e., with an ester instead of an amide linkage between ethanolamine and arachidonic acid
and termed virodhamine, has been isolated from brain tissues.
It acts as a full agonist for the CB2 receptor and is a partial agonist for the CB1 receptor.
While it has yet to be determined how it is synthesised, stored or degraded, inter-conversion with anandamide can occur.
N‑Acylethanolamides derived from other members of the n-6 family of polyunsaturated fatty acids (docosatetraenoic and
docosapentaenoic acids as well as arachidonic) interact with both CB1 and CB2 receptors, as well as TRPV1 channels, so these should be
considered true endocannabinoids (and ‘endovanilloids’).
In mice genetically modified to lack the Δ6 desaturase, the ethanolamide of cis-5,cis-11,cis-14-eicosatrienoic acid
and the 2-acylglycerol analogue are formed and are ligands of CB1.
O-Arachidonoylethanolamine, i.e., with an ester instead of an amide linkage between ethanolamine and arachidonic acid
and termed virodhamine, has been isolated from brain tissues.
It acts as a full agonist for the CB2 receptor and is a partial agonist for the CB1 receptor.
While it has yet to be determined how it is synthesised, stored or degraded, inter-conversion with anandamide can occur.
N‑Acylethanolamides derived from other members of the n-6 family of polyunsaturated fatty acids (docosatetraenoic and
docosapentaenoic acids as well as arachidonic) interact with both CB1 and CB2 receptors, as well as TRPV1 channels, so these should be
considered true endocannabinoids (and ‘endovanilloids’).
In mice genetically modified to lack the Δ6 desaturase, the ethanolamide of cis-5,cis-11,cis-14-eicosatrienoic acid
and the 2-acylglycerol analogue are formed and are ligands of CB1.
Catabolism: There are facilitated transport systems for anandamide from the plasma membrane to other tissues, although the detailed mechanisms are poorly understood. Once it enters a cell, it can influence metabolism, and some can be converted to oxylipin metabolites (see below), but any excess is rapidly degraded to free arachidonic acid and ethanolamine. In vivo, the concentrations of these amides in many animal species are controlled by a single hydrolytic enzyme present in most tissues other than skeletal muscle and heart, i.e., a fatty acid amide hydrolase (FAAH-1), which is an integral membrane protein found primarily in the perinuclear membranes. It belongs to a large family of enzymes ('serine hydrolases') that share a highly conserved 130 amino acid motif termed the ‘amidase signature’ sequence and is conserved in the primary structure.
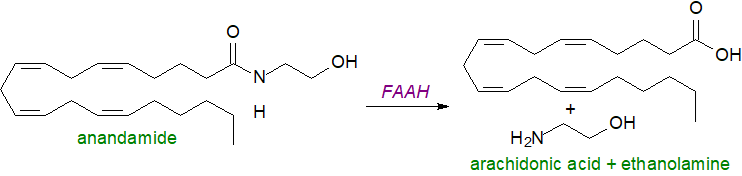 |
| Figure 4. Hydrolysis of anandamide. |
A second enzyme of this type (FAAH-2) has been found in humans and other primates but is absent in mice and rats and is more selective towards N‑oleoylethanolamine than to anandamide. The two enzymes are found in different tissues with the second present mainly in heart and ovary, where perhaps surprisingly it is located on the surface of cytoplasmic lipid droplets. Although N‑palmitoylethanolamine is the preferred substrate, there is an N‑acylethanolamine-hydrolysing acid amidase (NAAA) in lysosomes, macrophages and other immune cells with similar specificity, suggesting that it may regulate fatty acid ethanolamide levels at sites of inflammation.
There is currently great interest in the potential use of anandamide and other amides for therapeutic purposes, such as the alleviation of inflammation, asthma and some forms of chronic pain, and as anti-tumour drugs. Because of their role in terminating anandamide signalling, amide hydrolases are the subject of intensive study and are targets for potential drug therapies, and there is evidence that the growth of certain tumour cells is curtailed by inhibiting hydrolases and increasing the concentration of anandamide. Administration of such inhibitors is beneficial towards inflammatory pain, depression and anxiety with the potential to reduce the need for non-steroidal anti-inflammatory drugs (or cannabis-derived drugs) with their well-known side effects. It should not be forgotten that any arachidonic acid released can affect tissue metabolism.
Ethanolamides of n-3 fatty acids: N-acylethanolamides derived from the n-3 family of polyunsaturated fatty acids (eicosapentaenoic (EPA), docosapentaenoic (DPA) and docosahexaenoic (DHA) acids) have been detected in many animal tissues, but especially brain, and they are presumed to be synthesised and catabolized by the same mechanism as for anandamide. They may have a weak affinity for CB2 receptors, and the C22 derivatives are ligands for TRPV1 channels but not the CB1 receptor. N‑Docosahexaenoylethanolamine or 'synaptamide' is present in brain tissue in amounts comparable to anandamide, but it binds only weakly to the CB2 receptor. Rather, it binds strongly to the G protein-coupled receptor GPR110 (ADGRF1) to trigger cAMP production and signalling with low nM potency to induce neurogenesis, neuritogenesis and synaptogenesis in developing neurons (hence the trivial name), a further mechanism by which DHA promotes brain development and function. It has been suggested that the preferential stimulation of CB2 receptors by N‑acylethanolamides of the n‑3 family of polyunsaturated fatty acids contributes in part to the broad anti-inflammatory profile of the latter. As synaptamide is present in human breast milk, it is possible that it has a role in the development of the new-born infant.
EPA-ethanolamine is barely detectable in animal tissues under normal conditions, although it has been found in human milk and is apparently beneficial in psoriatic skin, while tetracosahexaenoylethanolamine has been detected in rat brain and may be neuroprotective. Saturated and monoenoic N‑acylethanolamides influence many metabolic processes in tissues but are not endocannabinoids by the strict definition, and they are discussed on a separate web page with other fatty amides and lipoamino acids.
Oxygenated metabolites of anandamide
It has been demonstrated that anandamide can be converted by cellular systems in vitro to ethanolamides of the prostaglandins PGE2, PGD2, PGF2α, PGJ2 and PGI2 (but not thromboxanes) with cyclooxygenase-2 (COX-2) rather than COX‑1 as a key enzyme. These metabolites have been termed 'prostamides'. The first significant product is PGG2-ethanolamide and this is acted upon by the same prostaglandin synthases that operate to produce the unesterified prostanoids (for PGH, PGD, PGE, PGF and PGI), though a quite distinct prostamide/PGF synthase has now been discovered that is abundant in brain and spinal cord. The step-wise enzymatic process is the same as that for prostaglandins per se, and only the initial substrate differs. For cyclo-oxygenation to occur, there is an essential requirement for the hydroxyl group of anandamide. Analogous reactions occur with the other endocannabinoid 2‑arachidonoylglycerol (see below).
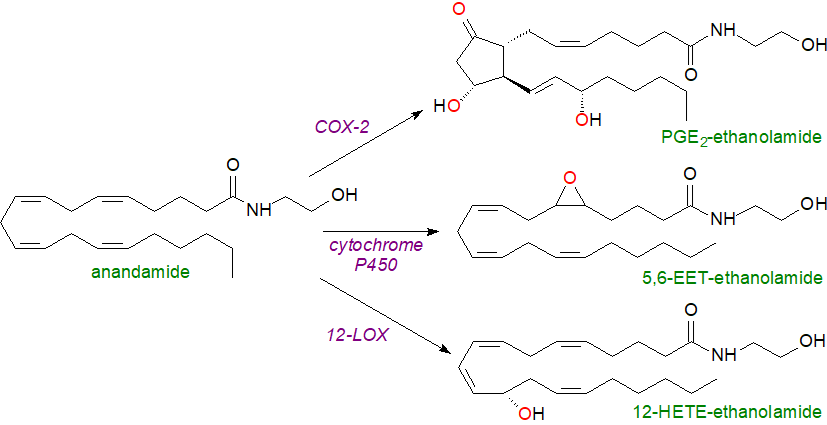 |
| Figure 5. Formation of oxygenated metabolites of anandamide. |
Anandamide is a substrate for 12- and 15-lipoxygenases (but not 5-LOX) to produce ethanolamides of hydroxy- and epoxyeicosatrienoic acids (HETE and EET). The full range of HETE-ethanolamide products is formed, but in a tissue and species-specific manner. It is noteworthy that human 15‑lipoxygenase-1 is able to convert anandamide to cysteine-containing metabolites ('eoxamides'), analogous to the eoxins, more rapidly than in the corresponding reaction with arachidonic acid per se. Cytochrome P450 enzymes produce the four epoxy metabolites (EET) and their dihydroxy derivatives that might be expected together with 20-hydroxy-anandamide. Mouse analogues of these enzymes have the same specificities. In contrast to anandamide, prostamides are not efficiently hydrolysed by FAAH1/2 or NAAA, although they can be catabolized in a comparable manner to the prostanoids per se.
The prostamides do not bind strongly to any of the canonical cannabinoid or prostanoid receptors, but a distinct prostamide F2α receptor complex has been identified. On the other hand, 12S‑HETE-anandamide and the related cytochrome P450 metabolites bind to both CB receptors with an affinity that is like that of anandamide per se or even greater in some instances, and they appear to act as cannabimimetics. For example, 5,6‑EET‑ethanolamine has a 300-fold selectivity for stimulation of the CB2 receptor over CB1 and a 1000 times greater affinity for CB2 than does anandamide, and it is possible that the oxygenated metabolites have their own as yet unidentified receptors. HETE-ethanolamides inhibit the fatty acid amide hydrolase, which catabolizes endocannabinoids.
The physiological significance of these novel lipids is now being explored. PGE2-ethanolamine is stable in human plasma and mobilizes calcium in cell preparations in vitro at picomolar concentrations. PGF2α-EA analogues are efficacious in the experimental treatment of glaucoma, while PGF2α-EA and PGE2-EA have variable effects in inflammatory conditions, although the former may be a factor in the perception of pain. PGD2-EA, 15‑deoxy-Δ12,14-PGJ2-EA and Δ12‑PGJ2/PGJ2-EA are produced and induce apoptosis in various cancer cell types such as those that over-express COX-2. In particular, PGD2-EA and its metabolite 15-deoxy-Δ12,14-PGJ2-EA are reported to act in this manner by inhibiting the antioxidants glutathione and thioredoxin, thus causing oxidative stress and thence apoptosis. PGE2-EA induces apoptosis in rat decidual cells, but PGF2α-EA acts as a regulator of the preadipocyte pool in adipose tissue.
Synaptamide can undergo a lipoxygenase reaction to form the 17-hydroperoxy-derivative, which can be reduced to 17S‑hydroxy-docosahexaenoylethanolamine, or it can be a precursor for further oxygenated metabolites. Similarly, cytochrome P450 (CYP450) products of synaptamide have been described, i.e., 19,20‑epoxy-docosapentaenoylethanolamine and related n‑3 fatty acid derivatives, which are potent lipid mediators. COX‑2 generates 13- and 16‑hydroxy-docosahexaenoylethanolamines with immunomodulating properties in macrophages, while eicosapentaenoylethanolamine is converted into PGE3-ethanolamine and 11-, 14- and 18-hydroxy-eicosapentaenoylethanolamines.
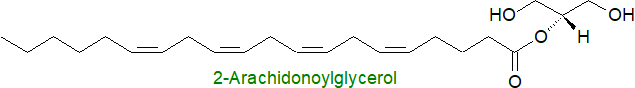
2. 2-Arachidonoylglycerol
2-Arachidonoylglycerol, a distinctive species of monoacylglycerol, was the second endocannabinoid to be discovered. It was first detected in canine gut and was demonstrated to be as an endogenous ligand for the cannabinoid receptors in 1995. Subsequently, it was shown to be abundant in the central nervous system with the concentrations in brain reportedly ~170 to 1000 times higher than that of anandamide, although it has been argued that much of the reported differences can be accounted for by a more rapid increase of 2‑arachidonoylglycerol post-mortem. Neurons release extracellular microvesicles containing this lipid when stimulated, but not anandamide. As a neutral lipophilic molecule, it is possible that 2‑arachidonoylglycerol can diffuse freely through membranes, although it does bind to the cytosolic carrier protein fatty acid binding protein 5 (FABP5); it is a little more soluble in aqueous media than anandamide.
As with anandamide, 2-arachidonoylglycerol is synthesised upon demand mainly from phospholipid precursors in cell membranes, probably in raft microdomains, in response to a rise in intra-cellular calcium levels. The main biosynthetic route for signalling purposes starts with generation of sn‑1,2‑diacylglycerols from phosphatidylinositol (or phosphatidylinositol 4,5-bisphosphate) in neurons by the action of phospholipase Cβ; these are hydrolysed by sn-1-specific diacylglycerol lipases (DAGLs) to yield 2‑arachidonoylglycerol. DAGLα is expressed throughout the brain, while DAGLβ has a more limited occurrence in brain and is more abundant in immune cells, although it can be highly active in certain cancers. Lysophosphatidylinositol is a direct precursor in a non-canonical pathway in humans via the action of glycerophosphodiesterase 3, a lysophospholipase C, which suppresses the normal receptor for lysophosphatidylinositol, i.e., GPR55, and so acts as a switch between GPR55 and CB2 signalling. Glutathione is a key regulator of this synthesis in the brain.
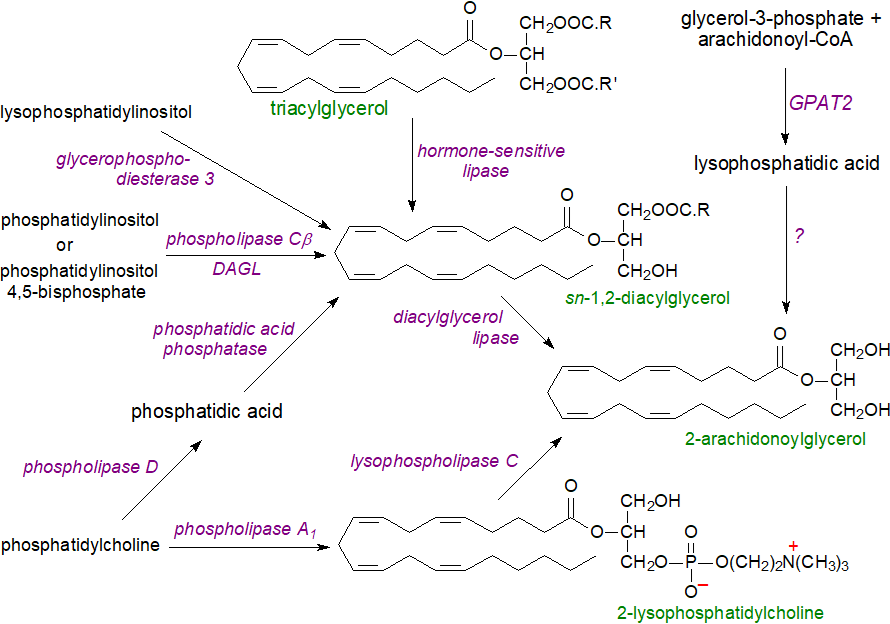 |
| Figure 6. Biosynthesis of 2-arachidonoylglycerol. |
Alternatively, the diacylglycerol intermediate can be produced by the sequential action of phospholipase D and phosphatidic acid phosphatase on phosphatidylcholine. The latter can serve as precursor via the action of a phospholipase A1 to generate 2‑arachidonoyl-lysophosphatidylcholine, which is in turn acted upon by a lysophospholipase C to produce 2‑arachidonoylglycerol. Triacylglycerols containing arachidonic acid in position sn‑2 can serve as precursors of the diacylglycerol intermediate after hydrolysis by hormone-sensitive lipase, carboxyl esterase or other lipases, but the relative contributions of these various pathways are not yet known. It is also possible that 2‑arachidonoylglycerol can be synthesised de novo from glycerol-3-phosphate and arachidonoyl-CoA and lysophosphatidic acid (possibly via phosphatidic acid), suggesting a link to signalling by these metabolites.
Function: The defining properties of the endogenous cannabinoids and their receptors are discussed above in relation to anandamide, but there is evidence to suggest that 2‑arachidonoylglycerol is a more efficient natural ligand for both the CB1 and the CB2 receptors. While anandamide may only act as a partial agonist at these receptors, 2-arachidonoylglycerol is a full agonist. The two molecules may interact to regulate some processes, but the latter is distinctive in many ways and is a messenger molecule that regulates the transmission of signals across synapses in the brain, facilitated by an interaction with fatty acid-binding protein 5 (FABP5). At excitatory synapses, the 2‑arachidonoylglycerol 'signalosome' consists of a supra-molecular complex in a single functional unit containing three crucial components: phospholipase Cβ, an activator protein designated mGluR5 and DAGLα. Activation of this complex with synthesis of 2‑arachidonoylglycerol results in 'post-synaptic depression' through the synapse, i.e., a sustained suppression of excitatory signalling, by preventing the excessive stimulation of glutamate receptors. In damaged neurons, it enhances the clearance of myelin debris by microglia and the differentiation of oligodendrocyte progenitor cells to enable a complete re‑myelination. It can also act as a precursor of other bioactive lipids.
Indeed, many of the metabolic processes mediated by 2-arachidonoylglycerol and the CB1 receptor are expressed through brain metabolism, and evidence suggests that it is responsible for the regulation of food intake and energy metabolism as increased levels were found during fasting in limbic forebrain and hypothalamus. It is important in modulating anxiety and depressive behaviours and in drug addiction, and studies with a rat model suggest that augmenting 2‑arachidonoylglycerol in brain may ameliorate the symptoms of autism. By providing the main pool of arachidonic acid for the generation of inflammatory eicosanoids, it is required, if indirectly, in the regulation of numerous neuro-inflammatory processes in the brain and this may be especially important in neonates. Anandamide may act in the same way but at a much lower level.
2-Arachidonoylglycerol is a mediator of inflammatory reactions and immune responses in many other tissues, and it can reduce pain. In immune cells such as platelets and macrophages, this lipid is produced after injury and is thought to be beneficial by decreasing pro-inflammatory mediators. As well as ameliorating some neurodegenerative diseases, there is evidence that as a result of pro-inflammatory stimuli it suppresses the elevation of the expression of COX-2, a key enzyme in prostaglandin biosynthesis. It may be a factor in systemic lupus erythematosus. Via its regulation of systemic energy metabolism, 2‑arachidonoylglycerol may be relevant to cardiovascular disease and to human reproduction. In the intestines, peripheral CB1 antagonists and CB2 agonists have shown efficacy in treating inflammation, reducing hepatic steatosis, and controlling the symptoms of irritable bowel syndrome.
Recent research has demonstrated that it has a role in the regulation of the proliferation and invasion of certain types of cancer cells with concentrations both in the primary tumour and in plasma increasing appreciably during cancer development and metastasis. It has been suggested that this increase may be due to activated immune cells, which trigger a phenotypic switch from aggressive to tumour-tolerant cells via the CB2 receptor, but an alternative explanation from experiments with human colon cancer cells in vitro is that it inhibits cyclooxygenase-2 (COX-2) expression induced by prostaglandin E2. The result is that 2‑arachidonoylglycerol is an anticancer agent by inhibiting the proliferation, migration and/or invasion of cancer cells, and tumour angiogenesis. In contrast, DAGLβ is the main enzyme that synthesises 2‑arachidonoylglycerol in intrahepatic cholangiocarcinoma, and it promotes the aggressive phenotype of this form of the disease. The expression of the CB1 and CB2 receptors increases markedly in some types of cancer in relation to healthy tissue but not in others. Therefore, increasing the concentrations of endocannabinoids by direct administration or by inhibiting their catabolism is seen as a potential therapeutic strategy in some if not all cancers. Although some consider that they present ethical problems, phytocannabinoids such as Δ9‑tetrahydrocannabinol interfere with the endocannabinoid pathways and are being tested to alleviate pain in cancer.
 The 2-glycerol ether analogue (termed ‘noladin ether’) acts similarly in vitro, and it has been reported to
occur naturally in pig brain but at very low levels.
It is more stable metabolically than 2‑arachidonoylglycerol, and it is an agonist of both CB1 and CB2 receptors and a partial agonist of
TRPV1 and PPARα; it serves a neuroprotective role among other functions.
2-Glycerol esters of EPA and DHA have been detected in embryonic stem cells of mice and have been shown to be anti-inflammatory in vitro.
The 2-glycerol ether analogue (termed ‘noladin ether’) acts similarly in vitro, and it has been reported to
occur naturally in pig brain but at very low levels.
It is more stable metabolically than 2‑arachidonoylglycerol, and it is an agonist of both CB1 and CB2 receptors and a partial agonist of
TRPV1 and PPARα; it serves a neuroprotective role among other functions.
2-Glycerol esters of EPA and DHA have been detected in embryonic stem cells of mice and have been shown to be anti-inflammatory in vitro.
An endocannabinoid system utilizing 2-arachidonoylglycerol and anandamide has been identified in the primitive organism Hydra vulgaris (Cnidaria) and in molluscs, annelids and some crustaceans. In contrast, the fruit fly Drosophila melanogaster, widely used as a model species of insect, is a vegetarian and derives much of its unsaturated fatty acids from its diet; this does not include arachidonic acid, which can be lethal in some developmental stages. It has now been determined that this species utilizes 2-linoleoyl-glycerol as a signalling molecule in an endocannabinoid-like way, suggesting that this may a signalling system from which endocannabinoids developed during evolution.
Catabolism/deactivation: When isomerized to the more thermodynamically stable 1/3-sn-glycerol isomers, arachidonoylglycerol interacts much less efficiently with the CB1 receptor and loses much of its potency; this probably occurs rapidly and spontaneously in vivo. In plasma and within cells, 2‑arachidonoylglycerol has a short half-life as do the prostanoid metabolites (see below), and it is rapidly hydrolysed to arachidonic acid and glycerol mainly by the cytosolic monoacylglycerol lipase present in most cell types but also by two α/β‑hydrolases such as the fatty acid amide hydrolase and the ubiquitously expressed serine hydrolase α/β-hydrolase domain 6 (ABHD6), with some tissue differences, and the last may be of most importance in regulating signalling. It is worthy of note that most monoacylglycerol lipases target the 1/3‑isomers preferentially and are less effective against 2-isomers.
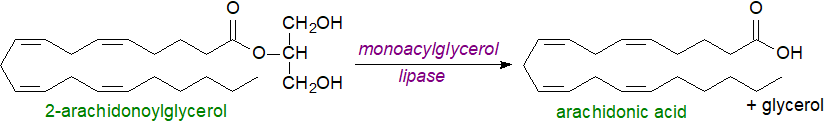 |
| Figure 7. Catabolism-hydrolysis of 2-arachidonoylglycerol. |
In brain and probably in other tissues, some of the arachidonic acid released in this way is re-cycled into phospholipids and some is a significant precursor for pro-inflammatory prostaglandins and other eicosanoids with implications for the regulation of many signalling pathways. Accordingly, inhibitors of the monoacylglycerol lipase may have pharmaceutical potential against several disorders including cancer, and neurodegenerative and inflammatory diseases, with fewer side-effects than many alternatives. They can promote hypothermia, hypomotility and analgesia. It is hoped that inhibitors of ABHD6 may prove efficacious against diseases such as epilepsy. Phosphorylation of 2-arachidonoylglycerol by an acylglycerol kinase generates another signalling molecule, lysophosphatidic acid.
Oxygenated metabolites of 2-arachidonoylglycerol
 Although the reaction is not as efficient as with arachidonic acid per se, 2‑arachidonoylglycerol
is a precursor of 2‑glycerol-linked prostanoids through an
interaction with the enzyme COX-2, but not COX-1, followed by further downstream processing as with anandamide (discussed above).
Thus, prostaglandin H2‑glycerol ester (2-PGH2-glycerol) is formed first, and this is converted sequentially by other
prostaglandin synthases to esterified forms of PGD2, PGE2, PGF2α and PGI2
(thromboxane analogues are only just detectable) by additional enzymes.
This may be a mechanism for terminating signalling by 2-arachidonoylglycerol in the central nervous system.
On the other hand, macrophages stimulated with the lipopolysaccharide Kdo2-lipid A (KLA) produce prostaglandins mainly within the first 12 hours,
followed later by robust synthesis of 2-glycerol forms.
Although the reaction is not as efficient as with arachidonic acid per se, 2‑arachidonoylglycerol
is a precursor of 2‑glycerol-linked prostanoids through an
interaction with the enzyme COX-2, but not COX-1, followed by further downstream processing as with anandamide (discussed above).
Thus, prostaglandin H2‑glycerol ester (2-PGH2-glycerol) is formed first, and this is converted sequentially by other
prostaglandin synthases to esterified forms of PGD2, PGE2, PGF2α and PGI2
(thromboxane analogues are only just detectable) by additional enzymes.
This may be a mechanism for terminating signalling by 2-arachidonoylglycerol in the central nervous system.
On the other hand, macrophages stimulated with the lipopolysaccharide Kdo2-lipid A (KLA) produce prostaglandins mainly within the first 12 hours,
followed later by robust synthesis of 2-glycerol forms.
Other oxygenated metabolites are produced by the action of lipoxygenases (12‑LOX and 15‑LOX) and a cytochrome P450 enzyme, the last of which produces two metabolites of 2‑arachidonoylglycerol, i.e., 2‑(11,12-epoxyeicosatrienoyl)-glycerol and 2‑(14,15-epoxyeicosatrienoyl)-glycerol, in various animal tissues. These metabolites constitute a new class of lipid mediator, i.e., further members of the endocannabinoid family, although only PGD2- and PGE2-glycerol esters have been confirmed as having physiological activity to date. For example, PGE2-glycerol is a multifunctional signalling molecule that influences pain, immunomodulation and synaptic plasticity. Although they do not interact with the prostaglandin receptors, glycerol-linked prostanoids may interact weakly with CB1 and CB2 and elicit responses in cultured cells, but this has been difficult to establish definitively because they are hydrolysed so readily. It is considered possible that these compounds have as yet unknown receptors of their own. Although they are present in tissues at much lower levels than the free acid forms, some of the glycerol-linked prostanoids have a greater influence in signalling with some being pro-inflammatory and others anti-inflammatory. It has yet to be established whether they are formed in vivo.
2-Epoxyeicosatrienoyl glycerols (and EPA and DHA analogues) bind to both CB1 and CB2 receptors with high affinity. While 2‑(15‑HETE)-glycerol is an agonist for the PPARα receptor with moderate affinity for the CB2 but not for CB1 receptor. It might be expected that these esterified oxylipins would be rendered inert by acyl migration like 2‑arachidonoylglycerol per se, although on release from their link to glycerol, they may signal directly. The monoacylglycerol lipase, a lysophospholipase A2 (LYPLA2) and carboxylesterases are the main enzymes responsible for hydrolysis of these oxygenated metabolites.
3. N-Arachidonoyldopamine and Other Dopamine Conjugates
N-Arachidonoyldopamine has been detected as an endogenous component of mammalian nervous tissue, especially the brain, with high concentrations in the hippocampus, cerebellum and striatum, and it has a distinctive physiological impact. For example, it is reported to be protective against Parkinson’s disease. It binds to the CB1 receptor for endocannabinoids and is cannabimimetic in that it affects the sensation of pain and relieves inflammation while influencing renal and cardiovascular metabolism, but it does not interact with dopamine receptors. As it interacts with the same receptor (vanilloid type 1) as capsaicin, the pungent ingredient of chili peppers, with which it has some structural similarity, it has been termed a ‘vanilloid’ or ‘endovanilloid’. N‑Arachidonoyldopamine induces COX-2 production and biosynthesis of prostaglandin PGD2, while inhibiting synthesis of PGE2.

Biosynthesis in mammals probably occurs by conjugation of dopamine with arachidonic acid, catalysed by a fatty acid amide hydrolase (not via the CoA ester), although there are suggestions that some might be derived from arachidonoyltyrosine. In D. melanogaster, N‑acyldopamides may be formed from CoA esters by the action of an arylalkylamine N-acyltransferase.
The N-oleoyl analogue has been found in brain and has characteristic physiological properties of its own, although it interacts with the same receptors as N‑arachidonoyldopamine; it induces COX-2 production but inhibits 5-lipoxygenase. By acting as an agonist for TRPV1, it induces interleukin 10 (IL-10) in the central nervous system with benefits towards inflammation and sepsis. While the N‑palmitoyl and N-stearoyl derivatives of dopamine do not interact with these receptors to a significant extent, they act together with N‑arachidonoyldopamine and anandamide to enhance calcium mobilization. Among the other activities so far detected, docosahexaenoyl(DHA)-dopamine assists in the transport of dopamine to the brain and is both an antioxidant and an anti-inflammatory agent; eicosapentaenoyl(EPA)-dopamine is likewise anti-inflammatory. DHA‑dopamine is of interest because of potential physiological properties, which are linked to brain synapses and retinal neuroprotection. Oxygenated metabolites are produced from N‑arachidonoyldopamine and DHA by the action of cytochrome P450 enzymes, and they may be involved in the pathogenesis of Parkinson’s disease. N‑Acetyldopamine is present in many animal tissues, but N‑hexanoyldopamine is highly cytotoxic, and N‑octanoyldopamine inhibits T-cell proliferation.
4. Oleamide
9Z-Octadecenamide or 'oleamide' is a primary fatty acid amide. It was first isolated from the cerebrospinal fluid of sleep-deprived cats and was identified as the signalling molecule responsible for causing sleep as it induced physiological sleep when injected directly into the brain of rats (linoleamide is reported to do the same). Although it bears very little structural relationship to other endocannabinoids, it is an agonist for the CB1 receptor and for PPARγ in vitro.
Two routes have been proposed for the biosynthesis of oleamide of which that generally favoured starts with synthesis of N‑oleoylglycine followed by oxidation by peptidylglycine α-amidating monooxygenase.
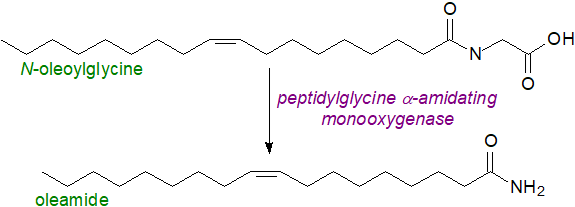 |
| Figure 8. Biosynthesis of oleamide. |
The second rather unusual mechanism suggested makes use of the enzyme cytochrome c with oleoyl-CoA and ammonium ions as the substrates and hydrogen peroxide as an essential cofactor (this reaction is illustrated in the web page dealing with the biosynthesis of simple lipoamino acids, which may follow the same route).
As well as its sleep-inducing properties, oleamide influences neurological behaviour, including regulation of memory processes, decreasing body temperature and locomotive ability, stimulating Ca2+ release, blocking potassium channels, in the perception of pain, and as an anti-cancer agent. As with the N‑acylethanolamides, the concentration of oleamide is controlled by the fatty acid amide hydrolase in vivo, but it is not known how these simple molecules avoid hydrolysis by the innumerable proteases, lipases and amidases present in brain.
Oleamide is produced by some bacteria and plants. Fatty acid primary amides other than 9Z-octadecenamide are present naturally in the cerebrospinal fluid of animals, and linoleamide is known to increase the Ca2+ flux, while 13-docosenamide or erucamide may act physiologically. The latter has also been identified in the bacterium Penicillium chrysogenum and has antimicrobial and cytotoxic activities. Receptors for these metabolites have not yet been identified, so it is not known whether they are endocannabinoids.
5. Other Endocannabinoids
N-Arachidonoylglycine and N-arachidonoylserotonin have many functions in animal tissues through binding to G‑protein-coupled receptors other than the CB1 and CB2 receptors. These are discussed on our web page dealing with lipoamino acids, although the different treatment of these from the endocannabinoids in this web site now looks somewhat arbitrary. There is a report that N‑oleoylglycine promotes adipogenesis as a ligand for the CB1 receptor, implying that it is an endocannabinoid, with the potential to increase insulin sensitivity and suppress obesity and diabetes.
6. Analysis
The main problems in the analysis of endocannabinoids relate to the low levels at which they occur naturally and the ease of isomerization or hydrolysis on storage of samples or extracts, and there is a concern that artefactually high results might be obtained because of the physiological consequences of inappropriate methods for tissue sampling, especially post-mortem. High-performance liquid chromatography allied to tandem mass spectrometry with electrospray ionization is now the preferred analytical methodology.
Recommended Reading
- Biringer, R.G. The rise and fall of anandamide: processes that control synthesis, degradation, and storage. Mol. Cell. Biochem., 476, 2753-2775 (2021); DOI
- Briand-Mésange, F. and others. From classical to alternative pathways of 2-arachidonoylglycerol synthesis: AlterAGs at the crossroad of endocannabinoid and lysophospholipid signaling. Molecules, 29, 3694 (2024); DOI.
- Cao, J.K., Kaplan, J. and Stella, N. ABHD6: its place in endocannabinoid signaling and beyond. Trends Pharm. Sci., 40, 267-277 (2019); DOI.
- Chen, C. Inhibiting degradation of 2-arachidonoylglycerol as a therapeutic strategy for neurodegenerative diseases. Pharmacol. Therapeut., 244, 108394 (2023); DOI.
- de Bus, I., Witkamp, R., Zuilhof, H., Albada, B. and Balvers, M. The role of n-3 PUFA-derived fatty acid derivatives and their oxygenated metabolites in the modulation of inflammation. Prostaglandins Other Lipid Mediators, 144, 106351 (2019); DOI.
- Grabiec, U. and Dehghan, F. N-Arachidonoyl dopamine: a novel endocannabinoid and endovanilloid with widespread physiological and pharmacological activities. Cannabis Cannabinoid Res., 2, 183-196 (2017); DOI.
- Hamzah, K.A., Turner, N., Nichols, D. and Ney, L.J. Advances in targeted liquid chromatography-tandem mass spectrometry methods for endocannabinoid and N-acylethanolamine quantification in biological matrices: A systematic review. Mass Spectrom. Rev., 44, 513-538 (2025); DOI.
- Howlett, A.C. and Abood, M.E. CB1 and CB2 receptor pharmacology. Adv Pharmacol., 80, 169-206 (2017); DOI.
- Le Foll, B. (Editor). Anandamide in Health and Disease. 540 pages (2025); DOI.
- Lee, J.W., Huang, B.X., Kwon, H., Rashid, M.A., Kharebava, G., Desai, A., Patnaik, S., Marugan, J. and Kim, H.Y. Orphan GPR110 (ADGRF1) targeted by N‑docosahexaenoylethanolamine in development of neurons and cognitive function. Nature Commun., 7, 13123 (2016); DOI.
- Maccarrone, M. and others. Goods and bads of the endocannabinoid system as a therapeutic target: lessons learned after 30 years. Pharmacol. Rev., 75, 885-958 (2023); DOI.
- Misao, T., Fukushima, K. and Fujino, H. Novel anti-cancer effect of 2-arachidonoylglycerol via processing body formation in HCA-7 human colon cancer cells. Prostaglandins Other Lipid Mediators, 174, 106861 (2024); DOI.
- Mock, E.D., Gagestein, B. and van der Stelt, M. Anandamide and other N-acylethanolamines: A class of signaling lipids with therapeutic opportunities. Prog. Lipid Res., 89, 101194 (2023); DOI.
- Ruiz-Contreras, H.A., Santamaría, A., Arellano-Mendoza, M.G., Sánchez-Chapul, L., Robles-Bañuelos, B. and Rangel-López, E. Modulatory activity of the endocannabinoid system in the development and proliferation of cells in the CNS. Neurotox. Res., 40, 1690–1706 (2022); DOI.
- Simankowicz, P. and Stepniewska, J. The role of endocannabinoids in physiological processes and disease pathology: a comprehensive review. J. Clin. Med., 14, 2851 (2025); DOI.
- Simard, M., Archambault, A.S., Lavoie, J.P.C., Dumais, E., Di Marzo, V. and Flamand, N. Biosynthesis and metabolism of endocannabinoids and their congeners from the monoacylglycerol and N-acyl-ethanolamine families. Biochem. Pharm., 205, 115261 (2022); DOI.
- Watson, J.E., Kim, J.S. and Das, A. Emerging class of omega-3 fatty acid endocannabinoids and their derivatives. Prostaglandins Other Lipid Mediators, 143, 10633 (2019); DOI.
- Uyama, T., Sasaki, S., Okada-Iwabu, M. and Murakami, M. Recent progress in N-acylethanolamine research: biological functions and metabolism regulated by two distinct N-acyltransferases: cPLA2ε and PLAAT enzymes. Int. J. Mol. Sci., 26, 3359 (2025); DOI.
- and of historical interest -
- Mechoulam, R. A delightful trip along the pathway of cannabinoid and endocannabinoid chemistry and pharmacology. Annu. Rev. Pharm. Toxic., 63, 1-13 (2023); DOI.
- Piomelli, D. The day anandamide almost died, and other endocannabinoid stories. Handbook Behavioral NeuroSci., 33, 3-17 (2025) DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: November 2025 | ||
© The LipidWeb is open access and fair use is encouraged but not text and data mining, AI training, and similar technologies.
