Sphingosine-1-Phosphate
While sphingosine-1-phosphate was first identified as an intermediate in sphingoid base catabolism in the laboratory of W. Stoffel in 1970, it was rediscovered but as "a novel lipid, involved in cellular proliferation" by Sarah Spiegel and colleagues in 1991. Now, it is recognized as a crucial element in both intra- and inter-cellular signalling, especially in animal cells, to influence innumerable metabolic pathways, and hundreds of publications appear every year on the topic. Its biochemistry cannot be described in isolation but must be considered together with those of the metabolically related sphingolipids ceramides, sphingoid bases and ceramide-1-phosphate, which have their own web pages here, as part of the sphingolipid rheostat. Catabolism of sphingosine-1-phosphate is ultimately the mechanism for removal of all sphingolipids from cells.
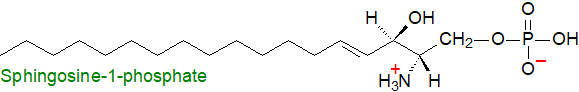
1. Occurrence and Biosynthesis in Animals
Sphingosine-1-phosphate, often abbreviated to S1P, is a zwitterionic lysophospholipid and cellular metabolite derived from sphingosine obtained by hydrolysis of other sphingolipids and is synthesised de novo or as part of the sphingomyelin cycle or sphingolipid rheostat (see below) in animal cells. Although it is a minor lipid in quantitative terms, it is vital for health in that it affects the heart, vascular development, immune cells, inflammation and cancer. Like most sphingolipids, it is resistant to oxidative stress. Unlike the lysoglycerophospholipids, with which it has some structural kinship, it exists mainly as a single molecular species in most animals, but in humans and mice, platelets contain small amounts of dihydrosphingosine-1-phosphate and other sphingoid base-1-phosphates also, and there is some evidence that these may have some different and even opposing properties. Analogues have been found in insects, yeasts and plants.
In most animal cells, sphingosine-1-phosphate occurs at concentrations in the low nanomolar range because of a rapid turnover, but in plasma, it can reach concentrations of 200 nM in humans to 700 nM in mice. A high proportion (~60%) is found in intimate association with the lipoproteins, mainly the high-density lipoproteins (HDL) and in particular the HDL3 subfamily, where it is bound to apolipoprotein M (apo M), a 25-kDa member of the lipocalin protein superfamily, which has a lipophilic binding pocket within the lipocalin structure; much of the remainder (~30%) is bound to albumin. When not bound to proteins, sphingosine-1-phosphate behaves in aqueous media as a soluble amphiphile with a critical micellar concentration of ~12 μM. Apo M has been termed a sphingosine-1-phosphate 'chaperone' that controls the levels of the lipid in plasma, although the highest concentrations are found in red blood cells. In contrast to most other sphingolipids, sphingosine-1-phosphate per se does not participate in formation of cholesterol-sphingolipid enriched regions (rafts) in membranes, although many of the proteins for its biosynthesis and signalling pathways are located in these membrane microdomains.
The primary precursor is sphingomyelin, which is hydrolysed by sphingomyelinases to produce ceramides, and these are in turn acted upon by ceramidases (5 family members) to release sphingosine. While this occurs in a constitutive manner in the endosomal/lysosomal compartment, those ceramidases that take part in the production of sphingosine-1-phosphate are present in other membranes in the vicinity of relevant effectors. Finally, sphingosine is phosphorylated by ATP-dependent sphingosine kinases, which catalyse the transfer of phosphate from a molecule of ATP onto the C1 hydroxyl group of sphingosine to form sphingosine-1-phosphate.
 |
| Figure 1. Biosynthesis of sphingosine-1-phosphate. |
There are in fact two sphingosine kinases, designated Types 1 and 2 (SPHK1 and SPHK2, or SK1/2), which are part of a super-family of enzymes that includes ceramide kinase and diacylglycerol kinase. They are distributed ubiquitously in tissues but are highly abundant in erythrocytes and epithelial cells. Although the enzymes differ substantially in size, they have a high degree of polypeptide sequence similarity, but with different developmental expression and tissue and subcellular distributions, suggesting that each has distinct and non-overlapping physiological functions. SPHK1 is predominantly cytosolic, although present to a lesser extent in the endoplasmic reticulum and Golgi, and it is considered to be pro-survival, probably by inhibiting ceramide biosynthesis. Upon stimulation by growth factors, hormones and cytokines, it translocates to raft microdomains in the plasma membrane where it interacts with phosphatidylserine and phosphatidic acid and with proteins of the calmodulin family. A significant fraction is released to the extracellular space. Thus, SPHK1 controls the levels of sphingosine-1-phosphate in the cytosol and plasma membrane, and it is regulated by several factors including transcription, phosphorylation/dephosphorylation, protein-protein interactions (e.g., with flotillins), membrane accessibility and degradation.
SPHK2 is found mainly in the nucleus where it has the potential to regulate gene expression, but upon activation, it can translocate to the cytosol, mitochondria, internal membranes and plasma membrane, depending on cell type, and it has a broader specificity with the ability to phosphorylate phytosphingosine and dihydrosphingosine (and Fingolimod - see below) as well as sphingosine. As it is not secreted, it may be more important in the regulation of gene expression and of apoptosis. SPHK2 is the main form of the enzyme in platelets and the only form in the central nervous system.
The different locations of the two enzymes enables them to regulate distinct but crucial aspects of cell metabolism. Both enzymes act to control the sphingosine-1-phosphate gradient between cells and in the circulation and so influence innumerable metabolic pathways, but they can act in opposition. Thus, sphingosine-1-phosphate from SPHK1 contributes to cell growth and proliferation, whereas that from SPHK2 can lead to the inhibition of cell growth and induce apoptosis. Sphingosine-1-phosphate synthesised on the inner leaflet of the plasma membrane can cross this via the action of at least two transporter proteins, spinster homologue 2 (SPNS2) and major facilitator superfamily transporter 2b (Mfsd2b), from the ATP-binding cassette (ABC) family, or it can be transported in plasma to more distant tissues to interact with receptors on the surface of the same cell or on nearby cells. SPNS2 is present in endothelial cells, while Mfsd2b is more abundant in the plasma membranes of red blood cells and platelets, and in spleen and bone marrow. Sphingosine-1-phosphate can also be transported from the extracellular compartment back into cells by the cystic fibrosis trans-membrane conductance receptor.
In animals, the reverse reaction to synthesis occurs by the action of sphingosine phosphatases as part of the 'sphingolipid rheostat' (see below), and the enzymes act in concert to control the cellular concentrations of the metabolite, which are always low.
Sphingosine-1-phosphate in the circulation: It now seems certain that much of the sphingosine-1-phosphate in blood is synthesised in erythrocytes, platelets, mast cells and monocytes. In platelets, sphingosine-1-phosphate is produced mainly by the action of SPHK2 on free sphingosine absorbed from plasma or derived from ceramide generated in the plasma membrane from sphingomyelin. Resting platelets store sphingosine-1-phosphate in α-granules in the inner leaflet of the plasma membrane and secrete it upon stimulation by thrombin, a product of the coagulation process, during injury and inflammation by degranulation and binding to its receptor. Incidentally, the mechanism of secretion from human platelets in this circumstance requires generation of thromboxane and is mediated via the thromboxane receptor, an interesting link between sphingolipid and eicosanoid metabolism. Platelets can thus provide sphingosine-1-phosphate rapidly as required as in the case of an injury with consequent platelet activation, when MFSD2B in the plasma membrane exports it from the cell before much is bound to albumin. On the other hand, platelets are able to influence immune cells over a longer term in inflammation-driven vascular diseases.
Erythrocytes produce sphingosine-1-phosphate using SPHK1 and export it constitutively into blood in an ATP-dependent manner again by means of MFSD2B and utilizing a proton gradient to facilitate its release. This occurs partly in free form but mainly by extraction by the protective chaperone apo M with incorporation into high-density lipoproteins (HDL) to provide more than 60% of the sphingosine-1-phosphate in plasma, with the remainder bound to albumin and VLDL. Erythrocytes can take up preformed sphingosine-1-phosphate from plasma, and they may act mainly as a storage organ as they lack the relevant degradative enzymes. They thus constitute a buffering system to maintain appropriate levels in the circulation.
A proportion of the sphingosine-1-phosphate in plasma comes from lymphatic endothelial cells, from which there is a transport mechanism from the interior of the cells with the aid of the transporter SPNS2; inhibition of the synthesis of this protein influences inflammatory and autoimmune diseases. Both proteins are present in other cell types where they can import as well as export this lipid, although a high proportion is hydrolysed at the plasma membrane to sphingosine, which can be reutilized within the cell for synthesis of sphingosine-1-phosphate and other sphingolipids. Levels of sphingosine-1-phosphate in lymph fluid are four to five-fold lower than plasma, while those in interstitial fluid are roughly 1000-fold lower. Some sphingosine-1-phosphate may be produced within plasma by the hydrolysis of sphingosylphosphorylcholine by the enzyme autotaxin, better known for the production of lysophosphatidic acid from lysophosphatidylcholine.
A high proportion of the sphingosine-1-phosphate in blood is solubilized by an interaction with albumin, although this does not have a specific binding site, as well as with the HDL. There is a high turnover of the lipid in the circulation, and it has been determined that the liver is the primary site of clearance as well as being a major site for its synthesis. At the surface of hepatocytes, it is hydrolysed by lipid phosphate phosphatase 3 (LPP3) to sphingosine, which is taken up into the cells and converted back to sphingosine-1-phosphate by SPHK2; ultimately, it is catabolized as described below. It has been suggested that this may be a general mechanism whereby sphingosine-1-phosphate gradients are shaped.
2. Membrane Receptors and Functions
Like its precursors, sphingosine-1-phosphate is a potent messenger molecule that perhaps uniquely operates both intra- and inter-cellularly, i.e., it is both an autocrine and a paracrine agent. Although it is often compared to lysophosphatidic acid with which it has some structural similarities, the latter only operates extracellularly. Sphingosine-1-phosphate transported out of cells binds selectively to G protein-coupled receptors (GPCRs), which trigger a cascade of downstream signalling pathways, but alternatively, it can act directly on intracellular targets to mediate or coordinate cellular metabolism. It has very different functions from other sphingolipid metabolites such as ceramides, ceramide-1-phosphate and sphingosine, and the balance between them has sometimes been termed the 'sphingolipid rheostat', although the readiness with which they can be interconverted can make it difficult to determine the true effects or relative contributions of each. Within the cell (as a paracrine agent or first messenger) in contrast to ceramide and sphingosine, sphingosine-1-phosphate is pro-survival in that it promotes cellular division (mitosis) as opposed to cell death (apoptosis), which it inhibits in fact. There are differences between cell types under various conditions and the description of the sphingolipid rheostat may be an over-simplification as a balancing mechanism, though one that is necessary for health. A general hypothesis is that this mechanism evolved early in the development of life to regulate cell survival under environmental stress.
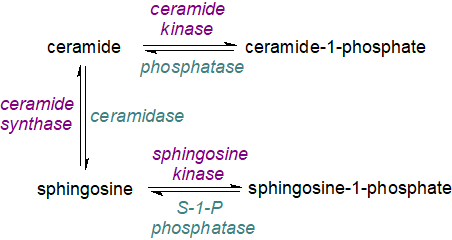 |
| Figure 2. Sphingolipid rheostat. |
Sphingosine-1-phosphate operates extra-cellularly as an autocrine agent or second messenger through acting as a ligand for receptors, in this instance five G protein-coupled receptors on cell surfaces that are designated S1PR1 to S1PR5. Both sphingosine-1-phosphate and its dihydro analogue bind to them with a high affinity, although the C16 or C20 homologues may not interact in the same way with metabolic consequences. These receptors are all differentially expressed and are coupled to one or more further G proteins (Gs, Gi/o, G12/13 and Gq/11), resulting in multiple different downstream messaging targets. In mammals, S1PR1 and S1PR2 are found in all tissues, often located within raft microdomains or caveolae, with S1PR1 (coupled to Gi) usually the most highly expressed. S1PR3 is expressed highly in the heart, lung, kidney and spleen and is located in the plasma membrane, while S1PR4 is restricted to blood cells and lymphoid tissues. S1PR5 is relatively abundant in brain, skin and natural killer (NK) cells, and it is one of the main regulators of the egress of natural killer cells from both bone marrow and spleen into the blood.
When activated, each receptor triggers distinctive signalling pathways and cellular responses, some of which can be antagonistic. The ligand-receptor interactions are required for the growth of new blood vessels, vascular maturation, cardiac development and immunity, organ morphogenesis, directed cell movement and for the regulation of corticosteroid hormone biosynthesis and function. Sphingosine-1-phosphate acts directly (receptor-independent) on other intracellular targets that include tumor necrosis factor-α-receptor-associated factor 2 (TRAF-2), histone deacetylase and prohibitin 2 to mediate multiple signalling cascades.
Many of the effects are mediated ultimately by the regulation of intracellular calcium fluxes by various mechanisms. As one example, sphingosine-1-phosphate binds to its receptors that couple to calcium signalling through the activation of phospholipase C and thence drive the hydrolysis of phosphatidylinositol 4,5‑bisphosphate to inositol triphosphate and diacylglycerols to act upon the inositol triphosphate receptor to release calcium from intracellular stores. Sphingosine-1-phosphate may bind to phosphatidylinositol monophosphates, targeting them to intracellular membranes.
The cellular location of sphingosine-1-phosphate production can dictate how it operates, even to the extent of producing
opposing effects, although the two biosynthetic enzymes may compensate for each other if one is inhibited, but only in part.
Thus, there is evidence that cytosolic sphingosine-1-phosphate formed by the action of SPHK1 is pro-survival in that it induces cell
proliferation and inhibits synthesis of ceramide de novo, while the SPHK2 isoform located in the endoplasmic reticulum promotes
ceramide synthesis through the sphingosine salvage pathway. Loss of both enzymes is embryonically fatal.
 Most functions of
sphingosine-1-phosphate have been attributed to that generated by the action of SPHK1, with much less known of the role of SPHK2.
In response to external stimuli, phosphorylated SPHK2 translocates into the nucleus
and produces sphingosine-1-phosphate, which inhibits DNA synthesis to influence subsequent cellular events.
This also increases the acetylation of lysine residues on histones to regulate gene transcription, mainly of pro-inflammatory genes,
and it is a factor in in the regulation of the immune and inflammatory responses of cytokines.
SPHK2 present in mitochondria is necessary for correct assembly of the cytochrome oxidase complex, and reductions of SPHK2 levels result in
defective mitochondrial respiration and can lead eventually to metabolic diseases.
Most functions of
sphingosine-1-phosphate have been attributed to that generated by the action of SPHK1, with much less known of the role of SPHK2.
In response to external stimuli, phosphorylated SPHK2 translocates into the nucleus
and produces sphingosine-1-phosphate, which inhibits DNA synthesis to influence subsequent cellular events.
This also increases the acetylation of lysine residues on histones to regulate gene transcription, mainly of pro-inflammatory genes,
and it is a factor in in the regulation of the immune and inflammatory responses of cytokines.
SPHK2 present in mitochondria is necessary for correct assembly of the cytochrome oxidase complex, and reductions of SPHK2 levels result in
defective mitochondrial respiration and can lead eventually to metabolic diseases.
When produced in the nucleus by SPHK2, sphingosine-1-phosphate delays senescence of cells, probably by binding to the telomerase reverse transcriptase (hTERT) to promote telomere maintenance. When operating extra-cellularly, it can enhance apoptosis, as apoptotic cells send cell–cell adhesion–dependent signals via sphingosine-1-phosphate and S1PR2 to neighbouring cells to induce a contractile ring, which moves the apoptotic cell to the apical space and stimulates Rho proteins. By promoting the formation of ceramide, it induces autophagy, i.e., the controlled turnover of damaged organelles, proteins and invading microorganisms within cells while providing nutrients to maintain vital cellular processes.
The concentration of sphingosine-1-phosphate in cells or the gradient in its concentration within or between tissues is regulated closely by a combination of synthesis de novo and catabolism (see below). The high levels in blood and especially that bound to apolipoprotein M help to maintain vascular integrity, as apo M protects the lipid from phosphatases and facilitates the interaction with receptors, and it is of importance in down-stream signalling while contributing to shaping the anti-inflammatory properties of HDL. While sphingosine-1-phosphate bound to albumin interacts with receptors S1PR1 to S1PR3, there are some differences in its effects. A relatively high concentration of sphingosine-1-phosphate is maintained in blood and lymph in contrast to low levels in intracellular or interstitial fluids, and this gradient in concentration is utilized to facilitate the exit of lymphocytes from lymphoid organs and direct them to sites of inflammation.
The vascular endothelium is the barrier that lines blood vessels and assists in cardiovascular homeostasis and blood flow and in controlling the passage of leukocytes into and out of the bloodstream. Plasma sphingosine-1-phosphate limits disruption of vascular endothelial monolayers, i.e., it protects the permeability barrier against potentially disruptive molecules such as histamine or platelet-activating factor. Damage to such endothelial barriers as the blood-brain barrier and placenta can result from a deficiency in sphingosine-1-phosphate, and this is a factor in many inflammatory disease states including sepsis. In contrast, ceramide and lysophosphatidic acid increase vascular permeability.
Small dense HDL3 subfractions display potent vasorelaxing properties, presumably because of their content of sphingosine-1-phosphate. It now known that sphingosine-1-phosphate is responsible for further clinical benefits of HDL by stimulating the production of the potent anti-atherogenic and anti-inflammatory signalling molecule nitric oxide by this tissue.
Receptor-mediated sensing of elevated sphingosine-1-phosphate (and lysophosphatidic acid) levels in the blood and lymph serves as a general mechanism in the regulation of the proliferation, survival, differentiation and migration of many types of stem cells, but crucially in the development of the vascular and nervous systems. Evidently, this lipid is an essential factor in embryogenesis, for example in the formation, positioning and working of the primitive heart tube. It also regulates keratinocyte differentiation and epidermal homeostasis. By activation of S1PR1 or S1PR3 (but not S1PR2), sphingosine-1-phosphate is beneficial towards glucose homeostasis and obesity. Together with vitamin D, it controls the migratory behaviour of circulating osteoclast precursors and thus mediates a critical point in bone homeostasis; systemically elevated blood levels may be an independent risk factor for osteoporosis-related fractures.
3. Sphingosine-1-Phosphate and Disease
Malfunctions of sphingosine-1-phosphate signalling have been implicated in many pathological conditions in humans, including cardiovascular disease, cancer, neurological diseases, multiple sclerosis, metabolic disease, inflammatory bowel disease and viral infections such as influenza. High levels of circulating sphingosine-1-phosphate and ceramides have been correlated with pregnancy disorders. Signalling by sphingosine-1-phosphate is often opposed to that by ceramides.
Inflammation and cardiovascular disease: As a blood-born lipid modulator, sphingosine-1-phosphate may be critical during platelet aggregation and thrombosis. The relatively high concentration of this lipid in plasma high-density lipoprotein (HDL) may have beneficial implications for atherogenesis via a signalling mechanism that facilitates HDL-derived cholesterol transport. It has been reported that decreased serum concentrations of sphingosine-1-phosphate is a better marker of peripheral artery disease and carotid stenosis than is HDL cholesterol (though less easily measured). The plasma concentration of sphingosine-1-phosphate, especially that bound to HDL, is decreased in patients with diabetes with negative effects upon cardiomyopathy, a risk factor for heart failure. As this lipid activates the receptor S1PR1 when bound to apo M, their combined interactions may be of relevance to atherosclerosis. On the other hand, sphingosine-1-phosphate induces vascular calcification (hardening of the arteries), an independent risk factor for cardiovascular disease. There is accumulating evidence that sphingosine-1-phosphate and its receptors regulate heart rate, blood flow in the coronary artery and blood pressure. By secreting apo M, the liver modulates the plasma levels of sphingosine-1-phosphate, and dysregulation of its metabolism may contribute to the development of liver diseases.
 In contrast,
sphingosine-1-phosphate is pro-inflammatory in that in response to certain cytokines and bacterial lipopolysaccharides,
it induces up-regulation of the enzyme cyclooxygenase-2 (COX-2) and thence production of the prostaglandin PGE2.
While SPHK1 upregulates the expression of pro-inflammatory mediators in rheumatoid arthritis, SPKH2 is reported to be anti-inflammatory
and controls platelet activation to limit or prevent arterial thrombosis after vascular injury.
Increased levels of sphingosine-1-phosphate are induced during chronic inflammation that decrease the level of anticoagulants
while inducing thrombin release to introduce blood clotting complications in some disease states including renal disease.
On the other hand, studies with enzyme inhibitors suggest that SPKH2 can be pro-inflammatory, and it takes part
in the production of inflammatory cytokines in the skin when triggered by mechanical stress or bacterial invasion.
In contrast,
sphingosine-1-phosphate is pro-inflammatory in that in response to certain cytokines and bacterial lipopolysaccharides,
it induces up-regulation of the enzyme cyclooxygenase-2 (COX-2) and thence production of the prostaglandin PGE2.
While SPHK1 upregulates the expression of pro-inflammatory mediators in rheumatoid arthritis, SPKH2 is reported to be anti-inflammatory
and controls platelet activation to limit or prevent arterial thrombosis after vascular injury.
Increased levels of sphingosine-1-phosphate are induced during chronic inflammation that decrease the level of anticoagulants
while inducing thrombin release to introduce blood clotting complications in some disease states including renal disease.
On the other hand, studies with enzyme inhibitors suggest that SPKH2 can be pro-inflammatory, and it takes part
in the production of inflammatory cytokines in the skin when triggered by mechanical stress or bacterial invasion.
Cancer: Like lysophosphatidic acid, sphingosine-1-phosphate is a marker for certain types of cancer, and there is increasing evidence that its role in cell division or proliferation has an influence on the development of cancers. It promotes the expression and release of interleukin 8 (IL-8), which is a potent angiogenic factor, which aids the growth and spread of cancers by enhancing the availability of nutrients and oxygen. In contrast to ceramide, it stimulates the growth, survival (delay of senescence) and migration of tumour cells, and it is abundant in malignant tissue, such as breast, colon and brain cancers. In a glioblastoma, the sphingosine-1-phosphate concentration was found to be nine-fold higher than in normal brain tissue, and there was a corresponding reduction in ceramide levels. A therapeutic aim is to ensure that these two sphingolipids are balanced to optimize cell survival and proliferation, so that the pro-apoptotic signalling of ceramides outweighs sphingosine-1-phosphate signalling in glioma cells to enhance tumour cell death.
There is increased expression of SPHK1 in many different cancers, and this enzyme has been linked with inflammation and the subsequent development of cancer, while SPHK2 is involved in B-cell acute lymphoblastic leukaemia and many other cancers, including those of the prostate, breast, ovaries, pancreas, lung and kidney. Sphingosine-1-phosphate produced inside the cell by SPHK1, but not by SPHK2, is exported to the extracellular space with the aid of ABC transporters, and high levels are found in the interstitial fluid that bathes cancer cells in the tumour microenvironment. After secretion from cancer cells, it can be passed to non-cancer cells to promote the spread of the disease.
Both sphingosine-1-phosphate and ceramide-1-phosphate are potent chemo-attractants for a variety of cell types to influence the trafficking of normal and malignant cells, thus promoting metastasis of cancer; the transporter SPNS2 is important to this process. This is currently a topic that is attracting great interest amongst medical researchers, who see a potential for therapeutic intervention in sphingosine-1-phosphate metabolism by selectively inhibiting biosynthesis by one or other of the sphingosine kinases or by targeting the receptors (e.g., by fingolimod, see below). On the other hand, this lipid may be beneficial towards wound healing by stimulating the proliferation of new cells that close the wound.
Immune system: Sphingosine-1-phosphate in the immune system signals newly made B and T lymphocytes to migrate from the bone marrow and thymus, respectively, to secondary lymphoid tissues including the spleen and lymph nodes where they may encounter foreign antigens. If they are not activated by an antigen, sphingosine-1-phosphate directs their circulation via other lymphoid tissues back into lymph and then into blood. Diminished circulating levels of the lipid are seen in patients with sepsis, and it is a factor in autoimmune diseases such as rheumatoid arthritis and related diseases in joints and bone, while the receptors for sphingosine-1-phosphate and relevant enzymes are viewed as promising therapeutic targets for other autoimmune diseases such as psoriasis, polymyositis and lupus. Sphingosine-1-phosphate induces differentiation of keratinocytes in skin, but the role of this lipid in psoriasis vulgaris is complex; increased levels are found in plasma of psoriatic patients, but there are several reports that it can be protective, and modulators of S1PR1 ameliorate the disease.
Sphingosine-1-phosphate and enzymes for its metabolism are protective in some virus infections by regulating their replication and/or controlling the innate defence of the host. Sphingosine-1-phosphate lyase inhibits replication of the influenza virus by promoting antiviral interferon, and inhibitors of SPHK2 have shown promise against the disease. It has been demonstrated that both SPHK1 and SPHK2 can regulate the replication or pathogenicity of many other viruses, including those affecting respiratory and entero diseases, hepatitis and herpes, but some viruses can divert sphingosine-1-phosphate metabolism to their own benefit.
Neurological diseases: There is much more sphingosine-1-phosphate in the brain than in any other organ, and this lipid is crucial to the regulation of diverse processes that include neural development, differentiation, migration and survival, while in synaptic transmission, it modulates the release of neurotransmitters. It protects myelin-forming oligodendrocytes after central nervous system injury and attenuates demyelination in vivo. On the other hand, in neurodegenerative processes, there are some reports that it is neuroprotective and others that it is neurotoxic, and while it may be protective during autophagy in neurons, it is pro-inflammatory in glial cells. It has been implicated in hypersensitivity to spontaneous and thermal pain, although the results appear to be controversial, but it does seem to have been established that sphingosine-1-phosphate and its receptor S1PR3 are critical regulators of acute mechanical pain mediated by fast-conducting mechanonociceptors, which close potassium channels and thence modulate the excitability of neurons.
Receptor-mediated signalling by both and lysophosphatidic acid is certainly critical for the pathogenicity of diverse neurological diseases, including such as neuropathic pain, systemic sclerosis, spinal cord injury, multiple sclerosis, traumatic brain injury and neuropsychiatric disorders, although it is not certain that changes in concentration of these lipids in some of these disorders are a cause or an effect of neuronal malfunction. In contrast, sphingosine-1-phosphate is reported to act as an inhibitor of neuronal apoptosis and to be protective against cellular damage induced by oxidative stress in the brain. It may inhibit the aggregation of aberrant amyloid-β peptide and the formation of neurofibrillary tangles with the resultant neuro-inflammation, which results in neuronal death in Alzheimer’s disease.
Others: Dysregulation of signalling by sphingosine-1-phosphate has been associated with obesity, insulin resistance and type II diabetes by a mechanism that involves induction of lipolysis of triacylglycerols in adipose tissue through direct activation of protein kinase C zeta. It is a regulatory factor in airway inflammation and bronchoconstriction in asthmatic individuals as high concentrations in the airways cause bronchoconstriction and provoke airway hyperresponsiveness to exacerbate asthma. By inducing the migration of immune cells into the lungs to cause or enhance inflammation, it contributes to the development of respiratory diseases, such as chronic obstructive pulmonary disease (COPD), pulmonary malignancies and infectious lung diseases, e.g., COVID-19 and influenza. Concentrations of sphingosine-1-phosphate are significantly correlated with the severity and prognosis of these diseases.
 Fingolimod: Drugs that antagonize sphingosine-1-phosphate and its receptors are being tested
clinically as immuno-suppressants to prevent rejection of kidney grafts, to reduce inflammatory and allergic responses in neurological diseases,
and as anti-cancer agents.
A molecule derived synthetically from considerations of the structures of sphingoid bases
and termed 'fingolimod (or FTY720)' has been approved by the Food and Drug Administration (FDA) in the USA
as an oral treatment for relapsing and remitting multiple sclerosis, a condition caused by an autoimmune attack on the myelin sheaths of nerves.
Within affected tissues, fingolimod is phosphorylated by SPHK2 and the resulting fingolimod-phosphate is released from cells
as an agonist for sphingosine-1-phosphate receptors to cause immunosuppression by inducing a marked decrease in circulating lymphocytes
where the S1PR1 receptor is expressed on the cell surface.
Fingolimod: Drugs that antagonize sphingosine-1-phosphate and its receptors are being tested
clinically as immuno-suppressants to prevent rejection of kidney grafts, to reduce inflammatory and allergic responses in neurological diseases,
and as anti-cancer agents.
A molecule derived synthetically from considerations of the structures of sphingoid bases
and termed 'fingolimod (or FTY720)' has been approved by the Food and Drug Administration (FDA) in the USA
as an oral treatment for relapsing and remitting multiple sclerosis, a condition caused by an autoimmune attack on the myelin sheaths of nerves.
Within affected tissues, fingolimod is phosphorylated by SPHK2 and the resulting fingolimod-phosphate is released from cells
as an agonist for sphingosine-1-phosphate receptors to cause immunosuppression by inducing a marked decrease in circulating lymphocytes
where the S1PR1 receptor is expressed on the cell surface.
Fingolimod is an anti-cancer agent by preventing the formation of sphingosine-1-phosphate and inhibiting its metabolic effects. In mouse breast cancer models, FTY720 treatment reduces tumour growth and metastasis while improving the response to conventional therapies and reducing neuropathic pain, and it is under consideration as a simultaneous adjuvant treatment of triple negative breast cancer. In consequence, this drug has reached the phase II stage in clinical trials for amyotrophic lateral sclerosis, acute stroke and schizophrenia, and the phase I stage for Rett syndrome, colorectal and breast cancer, and glioblastoma.
Pre-clinical studies suggest that fingolimod is neuroprotective against Alzheimer's, Parkinson's and Huntington's diseases. While sphingosine-1-phosphate prevents photoreceptor and ganglion cell degeneration, it can promote inflammation in age-related macular degeneration, glaucoma and pro-fibrotic disorders, and fingolimod preserves neuronal viability and retinal function. Second generation analogues of fingolimod ('siponimod', 'ozanimod' and 'ponesimod') with greater specificity for the S1PR1 receptor are undergoing advanced clinical testing both as primary therapies and as neuroprotective adjuvants to existing treatments. Siponimod has been approved by the FDA for the treatment of secondary progressive multiple sclerosis and ozanimod for moderate to severe ulcerative colitis.
Dihydrosphingosine(sphinganine)-1-phosphate may produce different results in various disease states from the sphingosine analogue, and they include cardiovascular disease, cancer, and lung, liver and kidney diseases.
4. Sphingoid Base-1-Phosphates in Plants, Fungi and Bacteria
In yeasts and plants, sphinganine, sphingosine, phytosphingosine and other long-chain bases are phosphorylated by kinases, three such identified in the model plant Arabidopsis thaliana, to form the appropriate 1‑phosphates. As sphingosine-1-phosphate per se is rarely present in detectable amounts, it has been suggested that the term ‘long-chain (sphingoid) base-1-phosphates’ should be used in discussing the metabolism of these lipids in plants. On the other hand, sphingosine-1-phosphate can accumulate in leaves of Arabidopsis, when these are stressed by application of the fungal toxin fumonisin B1. Phytosphingosine-1-phosphate is a mediator of abscisic acid-mediated stomatal closure in Arabidopsis, but sphingosine-1-phosphate and sphingadienine-1-phosphate act in this manner in Commelina communis and rice, respectively. Less is known of these lipids in comparison to animal tissues and no receptors have yet been found, but there is evidence that they take part in such diverse processes as defence mechanisms, pathogenesis, calcium mobilization, membrane stability, and the response to drought or heat stress. As in animals, they inhibit the process of apoptosis.
Sphingosine-1-phosphate and dihydrosphingosine-1-phosphate have been detected in pathogenic fungi (Cryptococcus species), and in yeasts, phytosphingosine-1-phosphate has a role in the regulation of genes required for mitochondrial respiration.
The oral anaerobic bacterium Porphyromonas gingivalis, which synthesises a variety of sphingolipids, utilises DhSphK1, a protein that shows high similarity to a eukaryotic sphingosine kinase, to synthesise dihydrosphingosine-1-phosphate, which may then be a signalling molecule in its human host.
5. Catabolism of Sphingosine-1-Phosphate and Sphingoid Bases
The balance between catabolic enzymes and sphingosine kinases and synthases is tightly regulated, and while the half-life of sphingosine-1-phosphate is very short (ranging from 1 to 15 min) as it is rapidly cleared from the circulation by degradative enzymes, it can be quickly synthesised to maintain appropriate plasma concentrations. Long-chain bases can be regenerated from sphingosine-1-phosphate by the action of certain phosphatases (SPP1 and SPP2), located in the endoplasmic reticulum, and of three lipid phosphate phosphatases (LPP1 to 3). In mammalian cells, there is an unusual pathway for the salvage of sphingosine that requires its phosphorylation by SPHK2 (but not SPHK1) and then de-phosphorylation by a specific phosphatase for acylation by ceramide synthase to regenerate ceramide. Extracellular sphingosine-1-phosphate is degraded at the plasma membrane by LPP3, an enzyme required for the catabolism of lysophosphatidic acid, to release sphingosine, which can then be taken up by cells for synthesis of complex sphingolipids or resynthesis of intracellular sphingosine-1-phosphate.
 |
| Figure 3. Catabolism of sphingosine-1-phosphate. |
In animals and plants, production of sphingosine-1-phosphate is a necessary step in the catabolism of long-chain bases. Within cells, sphingosine-1-phosphate is cleaved irreversibly in the endoplasmic reticulum by the enzyme sphingosine-1-phosphate lyase, which like the serine palmitoyltransferase for synthesis of sphingoid bases requires pyridoxal 5’-phosphate as a cofactor. This enzyme catalyses the retro-aldol-like cleavage of sphingosine-1-phosphate to yield trans-2-hexadecenal and ethanolamine phosphate. In humans, it will only interact with the naturally occurring D-erythro-isomer of a long-chain base, and it will not react with sphinganine-1-phosphate, although the analogous enzyme from rat liver extracts is much less regiospecific and can cleave a variety of different sphingoid bases including sphingosine-1-phosphate, sphinganine-1-phosphate, phytosphingosine-1-phosphate and sphingosine-1-phosphonate. As this catabolic reaction is irreversible, it is ultimately the mechanism for removal of all sphingolipids from cells.
The reaction with sphingosine-1-phosphate lyase, which is found in many different organs and especially lymphoid tissues but not in erythrocytes, reduces the cellular levels of sphingosine and ceramide. Because it regulates the intracellular and circulating levels of sphingosine-1-phosphate, it is seen as a potential target for pharmacological intervention, though no inhibitor of the enzyme has yet been found with sufficient specificity or that can cross the blood-brain barrier.
trans-2-Hexadecenal produced in the reaction can enter the β-oxidation pathway, or it can be reduced to the long-chain alcohol or converted via four reactions into palmitoyl-coA for incorporation into glycerolipids. In the last of these, the trans-2-enoyl-CoA reductase, responsible for the conversion of trans-2-hexadecenoyl-CoA to palmitoyl-CoA, is a dual function enzyme with a role in the production of very long-chain fatty acids. As an electrophilic α,β-unsaturated aldehyde, trans-2-hexadecenal has the potential to interact with proteins via the Michael reaction, and it is known to induce cytoskeletal reorganization and apoptosis by a signalling mechanism, Failure to oxidize this to palmitic acid is the cause of the rare genetic condition Sjögren-Larsson syndrome. The ethanolamine phosphate that is the other product of the reaction can be utilized for biosynthesis of phosphatidylethanolamine, and this is essential in the protozoan parasite Trypanosoma brucei, where the sphingosine-1-phosphate lyase enzyme is in mitochondria not the endoplasmic reticulum. This reaction is a further link between sphingolipid metabolism and that of the glycerophospholipids.
In plants and yeasts, phytosphingosine with an additional hydroxyl group in position 4 is catabolized in a similar way in the form of the 1‑phosphate by a phytosphingosine-1-phosphate lyase (DPL1) to yield 2-hydroxy-hexadecanal, which is then subject to alpha-oxidation to form pentadecanoic acid (15:0) and thence further odd-chain homologues. There is also a plant phosphatase, phyto-S1P phosphatase (SPPASE).
6. Analysis
Analysis of sphingosine-1-phosphate presents problems because of its high polarity and relatively low hydrophobicity, but methods are now available for quantitative extraction from tissues, and modern electrospray-ionization mass spectrometry techniques for detection and quantification afford high sensitivity and specificity.
Recommended Reading
- Bravo, G.Á., Cedeño, R.R., Casadevall, M.P. and Ramió-Torrentà, L. Sphingosine-1-phosphate (S1P) and S1P signaling pathway modulators, from current insights to future perspectives. Cells, 11, 2058 (2022); DOI.
- D'Aprile, C., Prioni, S., Mauri, L., Prinetti, A. and Grassi, S. Lipid rafts as platforms for sphingosine 1-phosphate metabolism and signalling. Cell. Signal., 80, 109929 (2021); DOI.
- Escarcega, R.D., McCullough, L.D. and Tsvetkov, A.S. The functional role of sphingosine kinase 2. Front. Mol. Biosci., 8, 683767 (2021); DOI.
- Ghaderi, S. and Levkau, B. An erythrocyte-centric view on the MFSD2B sphingosine-1-phosphate transporter. Pharmacol. Therapeut., 249, 108483 (2023); DOI.
- Gomez-Larrauri, A., Larrea-Sebal, A., Martín, C. and Gomez-Muñoz, A. The critical roles of bioactive sphingolipids in inflammation. J. Biol. Chem., 301, 110475 (2025); DOI.
- Green, C.D., Maceyka, M., Cowart, L.A. and Spiegel, S. Sphingolipids in metabolic disease: The good, the bad, and the unknown. Cell Metab., 33, 1293-1306 (2021); DOI.
- Hallisey, V.M. and Schwab, S.R. Get me out of here: Sphingosine 1-phosphate signaling and T cell exit from tissues during an immune response. Immun. Rev., 317, 8-19 (2023); DOI.
- Kleuser, B. and Baumer, W. Sphingosine 1-phosphate as essential signaling molecule in inflammatory skin diseases. Int. J. Mol. Sci., 24, 1456 (2023); DOI.
- Luo, Y.Q., Xue, H.P., Gao, Y., Ji, G. and Wu, T. Sphingosine kinase 2 in cancer: A review of its expression, function, and inhibitor development. Int. J. Biol. Macromol., 306, 141392 (2025); DOI.
- Luttgeharm, K.D., Kimberlin, A.N. and Cahoon, E.B. Plant sphingolipid metabolism and function. In: Lipids in Plant and Algae Development. pp. 249-286 (Edited by Y. Nakamura and Y. Li-Beisson, Springer International Publishing, Switzerland) (2016); DOI.
- Phan, F., Bourron, O., Foufelle, F., Le Stunff, H. and Hajduch, E. Sphingosine-1-phosphate signalling in the heart: exploring emerging perspectives in cardiopathology. FEBS Letts, 598, 2641-2655 (2024); DOI.
- Pulkoski-Gross, M.J. and Obeid, L.M. Molecular mechanisms of regulation of sphingosine kinase 1. Biochim. Biophys. Acta, Lipids, 1863, 1413-1422 (2018); DOI.
- Rufail, M.L., Bassi, R. and Giussani, P. Sphingosine-1-phosphate metabolic pathway in cancer: implications for therapeutic targets. Int. J. Mol. Sci., 26, 1056 (2025); DOI.
- Tolksdorf, C., Moritz, E., Wolf, R., Meyer, U., Marx, S., Bien-Möller, S., Garscha, U., Jedlitschky, G. and Rauch, B.H. Platelet-derived S1P and its relevance for the communication with immune cells in multiple human diseases. Int. J. Mol. Sci., 13, 10278 (2022); DOI.
- Weigel, C., Bellaci, J. and Spiegel, S. Sphingosine-1-phosphate and its receptors in vascular endothelial and lymphatic barrier function. J. Biol. Chem., 299, 104775 (2023); DOI.
- Wieczorek, I. and Strosznajder, R.P. Recent insight into the role of sphingosine-1-phosphate lyase in neurodegeneration. Int. J. Mol. Sci., 24, 6180 (2023); DOI.
- Xiao, S.Q., Peng, K.X., Li, C.X., Long, Y.Y. and Yu, Q. The role of sphingosine-1-phosphate in autophagy and related disorders. Cell Death Disc., 9, 380 (2023); DOI.
- Yadav, R., Obinata, H. and Venkataraman, K. Delineating the intricacies of polymorphisms, structures, functions and therapeutic applications of biological high-density lipoprotein-apolipoprotein M: A review. Int. J. Biol. Macromol., 310, 143187 (2025); DOI.
- and in relation to the history of the topic -
- Spiegel, S. Sphingosine-1-phosphate: From insipid lipid to a key regulator. J. Biol. Chem., 295, 3371-3384 (2020); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: October 2025 | ||
© The LipidWeb is open access and fair use is encouraged but not text and data mining, AI training, and similar technologies.
