Simple N-Acylamides and Lipoamino Acids
Many simple fatty amides occur naturally in animals, plants and microorganisms, and these contain numerous amine and fatty acid moieties with the latter often saturated or monoenoic in nature (851 in the microbiome at the last count!). Of these, anandamide or N‑arachidonoylethanolamine is noteworthy as an endocannabinoid, and it is more appropriately discussed under that heading, together with oleamide and N-acyldopamines, which interact with the same receptors. On the other hand, it can be difficult at times to draw the line between what is or is not an endocannabinoid in a strict sense, since some agonists or antagonists for the endocannabinoid receptors (CB1 and CB2) also interact with those for other N‑acylethanolamides, while the endocannabinoids can interact with receptors for other N-acyl-amides.
In addition to these, many simple fatty acyl-amino acid conjugates (lipoamino acids) are present in animal, plant and bacterial tissues and have properties that are now being revealed and are discussed below. Fatty acid conjugates with short peptides, such as glutathione, are described here, but proteolipids in which fatty acids are linked covalently to proteins have their own web page. Similarly, bacterial proteolipids and lipopeptides are very different in structure and function so constitute a substantial topic in their own right and are discussed elsewhere in this web site, as are some of the taurolipids and the cysteinyl leukotrienes, which could be considered to be lipoamino acids. Fatty amides are produced synthetically in industry in large amounts (> 300,000 tons per annum) for use as ingredients of detergents, lubricants, inks and many other products, but here we are concerned with natural lipids only.
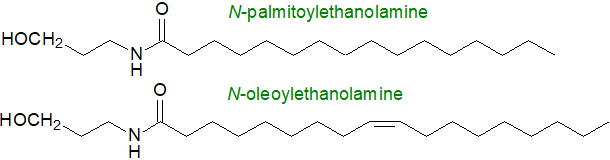
1. Long-Chain N-Acylethanolamides in Animals
Long-chain N-acylethanolamides are ubiquitous trace constituents of human and other animal cells, tissues and body fluids and have significant pharmacological properties. In rat plasma, the concentrations of N-palmitoyl-, oleoyl- and arachidonoyl-ethanolamines were found to be 2.5, 2.0 and 0.2 ng/ml, respectively, in one study, and somewhat higher concentrations have been reported in brain and other tissues. Similar lipids have been found in fish, molluscs, slime moulds and certain bacteria. The fatty acylethanolamides other than anandamide and N‑docosahexaenoylethanolamine are the subject of this section, and indeed they are the most abundant components of this lipid class often with distinctive biological properties of their own. Although most do not interact with the primary cannabinoid receptors, they may potentiate endocannabinoids by minimizing their degradation by competing for the hydrolytic enzymes. As described below, they are derived biosynthetically from precursor N-acyl-phosphatidylethanolamines, which are not lipid mediators (see below).
Occurrence and function: N-Palmitoylethanolamine takes part in a signalling system resembling that of the endocannabinoids but utilizes receptors other than the CB1/CB2 receptors characteristic of endocannabinoids. This lipid was first identified in egg yolk more than 50 years ago, and its anti-inflammatory properties were recognized immediately, but little more was done until there was a resurgence of interest in recent years, during which it has been shown additionally to have analgesic and neuroprotective actions. It is produced on demand in most mammalian tissues, although most research has been concerned with its occurrence in the central nervous system where it may act to counter neuroinflammation. In particular, it has been shown to have neuroprotective properties in mast cell-mediated models of stroke, spinal cord injury and traumatic brain injury, and in other studies, there is preliminary evidence that the progression of neurodegenerative diseases, such as Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, multiple sclerosis and cognitive decline is slowed down. It is beneficial towards several gastrointestinal diseases, including irritable bowel syndrome and inflammatory bowel diseases. It is undergoing clinical trials for the relief of chronic pain, and inhibition of the catabolic enzyme N‑acylethanolamine acid amidase (see below) has therapeutic potential in this regard. N‑Palmitoylethanolamine is available commercially as a nutraceutical in some countries.
The effects on inflammation and inflammatory pain are mediated through interactions with a number of high-affinity targets, which include thyroid hormone receptor beta, a G protein coupled receptor designated GPR55, peroxisome proliferator-activated receptor-α (PPARα), Toll-like receptors and the transient receptor potential vanilloid 1TPRV1, although other mechanisms have been postulated such as synergism with further acyl amides as part of a complex web of direct and indirect interactions. In macrophages, administration of palmitoylethanolamine promotes a shift from a proinflammatory phenotype to a proresolving profile that supports resolution of inflammation and may be relevant to many disease states, including cancer. While it does not bind to the cannabinoid receptors, it can increase the concentration of anandamide to enhance its activity. Palmitoylethanolamine is reported to negatively regulate some anaphylactic responses, such as mast cell degranulation, and in this instance, it may operate through an endocannabinoid-like system that involves CB2 and GPR55 with 2‑arachidonylglycerol, although this has been questioned. Like oleoylethanolamine, it displays an affinity for GPR119, although the former is a more potent agonist in vivo. Inhibition of its hydrolysis has therapeutic potential (see below).
N-Stearoylethanolamine is an immunomodulator, and it induces apoptosis of glioma cells. It down-regulates the expression of liver stearoyl-CoA desaturase-1 mRNA, it is anorexic, and it shares some of the anti-inflammatory properties of palmitoylethanolamine. Unlike the latter, it does not activate the nuclear receptor PPARα, although it does have an affinity for GPR119. It has anti-cancer properties and may have some cardioprotective effects but appears to be much less studied that other acylethanolamines, so far at least.
N-Oleoylethanolamine is an endogenous regulator of food intake, and it may have some potential as an anti-obesity drug. It acts as a local satiety signal rather than as a blood-borne hormone, and food intake is inhibited in rats following intraperitoneal injection and even after oral administration. Under normal physiological conditions, oleic acid from dietary fat is transported into enterocytes in the small intestine by a fatty acid translocase CD36, and some is converted to oleoylethanolamine via the intermediate N‑acylphosphatidylethanolamine (see below) and acts as a sensor for ingestion of fat by an interaction with the receptor GPR119 to release gut hormones. N-Oleoylethanolamine increases synthesis and secretion of triacylglycerols in enterocytes and apolipoprotein B secretion in chylomicrons.
The influences on metabolism are mediated in the intestinal brush border of enterocytes (and in hepatocytes) by binding with high affinity to PPARα, while TPRV1 may also be involved. This stimulates the vagal nerve via the fatty acid translocase/CD36 and sends neuronal signals to the brainstem, leading to increased lipolysis and β-oxidation of fats. Plasma oleate is not a precursor, and the fatty acid translocase CD36 is necessary for its synthesis. On the other hand, sustained ingestion of a high-fat diet abolishes the anorexic signal of oleoylethanolamine, and failure of the biosynthesis of this lipid mediator in enterocytes might be responsible for reduced satiety and weight gain. Oleoylethanolamine has anti-inflammatory and antioxidant properties also, but it does not interact with receptors CB1/2 so it is not considered to be an endocannabinoid by the strict definition. On the other hand, while oleoyl- and palmitoylethanolamines do not bind to cannabinoid receptors, they can enhance the activity of anandamide by inhibiting its hydrolysis by fatty acid amide hydrolase ('entourage effects').
 Anandamide and N-oleoylethanolamine are selectively decreased and then increased in rat intestine during food deprivation
and subsequent re-feeding through remodelling of the original acyl donor phospholipids, although they have opposing effects upon lipogenesis.
In adipose tissue and liver, oleoylethanolamine reduces the triacylglycerol content by stimulating lipolysis and elevating the circulating levels
of unesterified fatty acids and glycerol.
It is reported to mitigate cardiometabolic disruption secondary to obesity induced by high-fat diets in mice.
In liver, it leads to a decrease in triacylglycerol synthesis and the expression of the relevant enzymes, while
it may stimulate thermogenesis in brown adipose tissue.
Clinical trials of dietary supplements of oleoylethanolamine have shown benefits in terms of several parameters in relation
to the metabolic syndrome in the short term at least.
Anandamide and N-oleoylethanolamine are selectively decreased and then increased in rat intestine during food deprivation
and subsequent re-feeding through remodelling of the original acyl donor phospholipids, although they have opposing effects upon lipogenesis.
In adipose tissue and liver, oleoylethanolamine reduces the triacylglycerol content by stimulating lipolysis and elevating the circulating levels
of unesterified fatty acids and glycerol.
It is reported to mitigate cardiometabolic disruption secondary to obesity induced by high-fat diets in mice.
In liver, it leads to a decrease in triacylglycerol synthesis and the expression of the relevant enzymes, while
it may stimulate thermogenesis in brown adipose tissue.
Clinical trials of dietary supplements of oleoylethanolamine have shown benefits in terms of several parameters in relation
to the metabolic syndrome in the short term at least.
Recent research suggests that N-oleoylethanolamine is a mediator that fuels the growth of cells in chronic lymphocytic leukaemia and may be a factor in drug resistance and the wasting in the disease. By acting as a PPARα agonist, it enhances memory consolidation through noradrenergic activation of certain regions of the brain, and it may have an influence on sleep patterns and stress. It is reported to be beneficial towards non-alcoholic fatty liver disease (NAFLD), the most prevalent chronic liver disease, as a PPARα agonist. Preclinical studies have shown that oleoylethanolamine is neuroprotective in alcohol abuse. As a selective ligand for hypoxia-inducible factors (HIFs), i.e., α/β heterodimeric transcription factors that modulate cellular responses to the low oxygen condition, oleoylethanolamine may be a component of a regulatory mechanism. In the nematode Caenorhabditis elegans, considered a model for primitive animals, oleoylethanolamine binds to the lysosomal lipid chaperone LBP-8, which promotes longevity by its interaction with nuclear hormone receptors and thence transcription of target genes. Males of this nematode family produce complex N-acylethanolamides, often with a cyclopropyl fatty acyl group of bacterial origin, but linked to β-sophorosylphosphate, and these act as sex attractants for the females.
Surprisingly, the isomeric N-cis-vaccenoylethanolamine (i.e., of 11-18:1 rather than 9-18:1) was shown to be the most abundant 18:1 fatty acylethanolamide in rat plasma in unbound form and the second most abundant in human plasma, although the reason for this is as yet unknown; there was much more N‑oleoylethanolamine in the circulation as the N-acyl-phosphatidylethanolamine precursor. As there has been a potential for confusion with N‑oleoylethanolamine in certain studies, some re-appraisal of the latter may be necessary.
N‑Linoleoylethanolamine seems to have been little studied, but recent finding suggest that it has a role in metabolic regulation, immune modulation, and neuroprotection; like the oleoyl form, it inhibits intake of dietary fatty acids by signalling through PPARα and GPR119. A lipoxygenase metabolite, 13-HOHE-ethanolamine has been detected in human skin and saliva. α-Linolenoylethanolamine may have a function in brain, while N-acylethanolamides in human reproductive fluids may help to regulate many physiological and pathological processes in the reproductive system.
There is increasing evidence that an optimum balance between the various ethanolamides is necessary for the correct operation of innumerable biological systems, with an imbalance leading to pathological conditions. In some stress situations, increased levels of saturated and mono-unsaturated ethanolamides are produced, and in others, there is selective stimulation of anandamide synthesis. Saturated and monoenoic N-acylethanolamides may serve as intracellular messengers by activating kinases and interacting with the signalling pathways mediated by ceramide with which they have some structural similarities, effects that may be characteristic of particular tissues.
Biosynthesis: N-Oleoyl- and N-palmitoylethanolamine are produced in tissues from the precursor N-acyl-phosphatidylethanolamines by a comparable mechanism to that of anandamide (there are more details on that web page), i.e., by an initial transfer of the fatty acyl group from positions sn‑1 of phosphatidylcholine to phosphatidylethanolamine to form N-acyl phosphatidylethanolamine, from which the N‑acylethanolamide is released mainly by the action of a phospholipase D selective for this lipid, although other hydrolases may be utilized. Amides as products of phospholipid metabolism are thus in a state of dynamic equilibrium as part of the normal system of redistribution of molecular species in phospholipids.
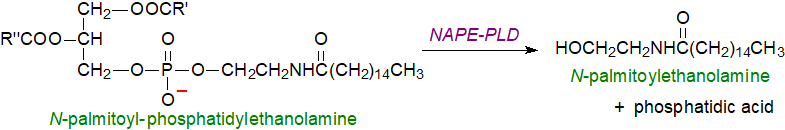 |
| Figure 1. N-Palmitoylethanolamine biosynthesis from N-acyl-phosphatidylethanolamine (main pathway). |
Catabolism: As with anandamide, other N-acylethanolamides are hydrolysed though more slowly by the fatty acid amide hydrolase (FAAH-1) and by the lysosomal acid ceramidase. A lysosomal enzyme that is highly specific for N‑palmitoylethanolamine has been characterized (N‑acylethanolamine-hydrolysing acid amidase (NAAA)), and inhibition of this is seen as a promising pharmaceutical target with the potential to control chronic pain and inflammation. In mice, it has been demonstrated that inhibition of this enzyme is beneficial towards triple negative breast cancer-associated inflammation and progression.
2. Long-Chain N-Acylserotonins in Animals
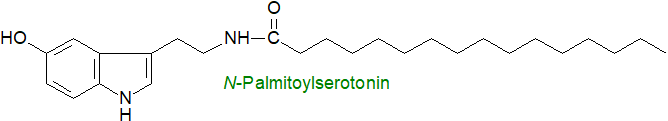
Serotonin or 5-hydroxytryptamine per se is a neurotransmitter derived from tryptophan and is synthesised mainly in the enteric nervous system of the gastrointestinal tract (90%); it is present in platelets and the central nervous system of animals where it is popularly known as a contributor to feelings of well-being. N‑Acylserotonins were first found in coffee beans (next section), but others with 16:0, 18:0, 18:1, 20:4, 20:5 and 22:6 acyl moieties have been detected in intestinal tissue (jejunum and ileum) from the rat, pig and humans where they may have regulatory functions. N‑Docosahexaenoylserotonin in human intestinal tissue is a potent anti-inflammatory mediator that may be relevant to intestinal inflammatory conditions such as Crohn's disease and ulcerative colitis. N‑Arachidonoylserotonin has been found in brain where it is reported to be an analgesic; it is an inhibitor of fatty acid amide hydrolase and binds to the vanilloid TRPV1 receptor. Other N-acyl-serotonins have been found in the brains of mice, and N‑palmitoylserotonin is reported to have anti-allergy properties and to improve memory in animal models. N-Acetylserotonin is an intermediate in melatonin biosynthesis and has neuroprotective, antioxidant and anti-apoptotic properties.
An arylalkylamine N-acyltransferase has been characterized from Drosophila melanogaster that catalyses the formation of long-chain N‑acylserotonins from CoA esters and serotonin, but little appears to be known of their biosynthesis in higher animals.
3. N-Acylamides in Plants
N-Acylethanolamides are minor but ubiquitous components of plant tissues, and they are especially abundant in desiccated seeds. Oleoylethanolamine is present naturally at low levels in such food products as oatmeal, nuts and cocoa powder (up to 2 μg/g), and in general, the fatty acids are representative of those in plants with zero to three double bonds and 12 to 18 carbon atoms. In this instance, the precursor N‑acylphosphatidylethanolamine is synthesised by a different mechanism from that in animals, i.e., by direct acylation of phosphatidylethanolamine by an N‑acyl phosphatidylethanolamine synthase. N‑acylethanolamides are released from this by the action of two of the isoforms of phospholipase D in response to stress situations. Another aspect of the biosynthetic mechanisms for most plant amides that differs from animals is that the amine moiety is often derived from an amino acid.
As in animals, such compounds are required in plants for many different purposes, which include toxicity to insect predators, bacteria and fungi, while promoting plant growth; they have a conserved role in cell signalling, although research is still at a relatively early stage. They may block essential processes that are part of the transition from seed germination to seedling growth in some circumstances, although they can be beneficial in others. N‑Linoleoylethanolamine disrupts root development in seedlings, although a 9‑lipoxygenase metabolite of this (9‑hydro(pero)xy-linoleoylethanolamine) may be a more important metabolite that works synergistically with abscisic acid to modulate the transition from embryo to seedling. N‑Linolenoylethanolamine inhibits chloroplast development, N‑lauroylethanolamine hinders seedling growth and flower senescence, and N‑myristoylethanolamine serves in plant defence against pathogen attack and inhibits stomatal closure.
Further metabolites of N‑lauroylethanolamine have been characterized in which glucose is attached to the free hydroxyl of the ethanolamine moiety together with mono- or dimalonylglucosides; these may represent a mechanism for modulating the activities of N‑acylethanolamides in plants. As in animals, a fatty acid amide hydrolase (FAAH) that degrades N‑acylethanolamides in vivo is present in Arabidopsis and is a regulatory factor.
Serotonin amides with saturated and monoenoic acyl groups are components of the wax layer of coffee beans, and when consumed in coffee, they are antinociceptive and neuroprotective, although they can cause stomach irritation. They have been characterized with 22:0 to 26:0 fatty acid components from the seed oil of the baobab tree (Adansonia digitata) and of the citrus family (together with N-methyl forms) where they may be antioxidants.
While these simple derivatives have attracted most recent interest because of their similarity to animal
lipids, more than 500 different N-acylamides have been identified from many families of plants and some fungi
with as many as 200 different fatty acids and innumerable amine moieties including propyl, isopropyl, butyl and often isobutyl moieties.
The fatty acid moieties are distinctive and fall into two groups, those with double bonds only and those with double and triple bonds.
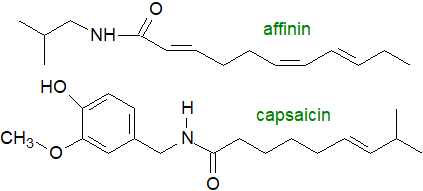 As an example, affinin (spilanthol), an insecticidal amide and an analgesic and anti-inflamatory agent, has a
10‑carbon fatty acid attached to isobutylamine, i.e., it is N‑isobutyl-2E,6Z,8E-decatrienamide.
Many plant amides have pharmaceutical properties for consumers that are now being explored, i.e., with anti-inflammatory, antitumour,
antidiabetic, analgesic, neuroprotective, antioxidant, and antibacterial properties, although many have been long known in folk medicine.
As an example, affinin (spilanthol), an insecticidal amide and an analgesic and anti-inflamatory agent, has a
10‑carbon fatty acid attached to isobutylamine, i.e., it is N‑isobutyl-2E,6Z,8E-decatrienamide.
Many plant amides have pharmaceutical properties for consumers that are now being explored, i.e., with anti-inflammatory, antitumour,
antidiabetic, analgesic, neuroprotective, antioxidant, and antibacterial properties, although many have been long known in folk medicine.
Capsaicinoids, the pungent components of chili peppers, are N‑acylated phenolic amides in which the fatty acid is nonanoic or decanoic acid, sometimes with an isomethyl group and sometimes a trans-double bond in position 6 or 7. The pungency is mainly determined by the benzene ring and is modified by the acyl chain. Capsaicin per se is the vanillyl derivative, (6E)-N-(4-hydroxy-3-methoxybenzyl)-8-methyl-6-nonenamide, and it affects humans by causing gastrointestinal discomfort through binding to the vanilloid receptor subtype 1 (TRPV1), although it can exert protective effects on some liver diseases and cancers; toxicity problems inhibit its value for pharmaceutical use. Macamides (benzylamides of long-chain fatty acids) and macaenes (unusual unsaturated long-chain fatty acid derivatives) are characteristic components of the maca plant (Lepidium meyenii), reputed to be a nutraceutical. It was from Peru originally but is now grown commercially in China and Tibet.
N-Acylureas are produced by fungi, often in the form of symmetrical molecular species with palmitoleic, oleic or linoleic acids as the fatty acid constituents. They interact with the human immune system, possibly alerting the host to the presence of a fungal infection.
4. Simple Lipoamino Acids from Animal Cells
N-acetyl derivatives of amino acids are minor but ubiquitous components of animal tissues, and they may be formed simply as a means of excreting or detoxifying excesses of particular amino acids. Innumerable long-chain fatty acid-amino acid conjugates have been identified, with more than 50 different in rat brain, some of which are lipid mediators (although few have yet been studied). They are sometimes termed 'elmiric acids'. N-Acyl-taurines are discussed in our web page on lipid sulfates.
N-Acylglycine derivatives of short-chain fatty acids (C2 to C12) have long been recognized as minor constituents of urine and blood, and their compositions in the former may have some relationship to metabolic dysfunction as they accumulate in specific disorders of branched-chain amino acid metabolism and fatty acid β-oxidation. They are formed in the liver and kidney by a mitochondrial acyltransferase, glycine N‑acylase, which catalyses the transfer of acyl groups from coenzyme A to glycine. This may be a detoxification mechanism for removal of excess acyl-coenzyme A esters in part, although acylglycines can serve as intercellular messengers via cell surface receptors. Acyl-glycines produced by gut bacteria (see below) may influence the metabolism of the host.
N-Arachidonoylglycine is present in bovine and rat brain as well as other tissues at low levels, and it is synthesised by at least three pathways in mammalian cells. In one, there is sequential oxidation of anandamide by alcohol dehydrogenase and aldehyde dehydrogenase, while in a second arachidonoyl-CoA and glycine combine in a reaction catalysed by glycine N-acyltransferase-like 3 (other glycine N‑acyltransferases are responsible for the formation of oleoylglycine and for short-chain fatty acylglycines). In a third pathway, cytochrome C catalyses the formation of arachidonoylglycine from arachidonoyl-CoA and glycine in the presence of hydrogen peroxide.
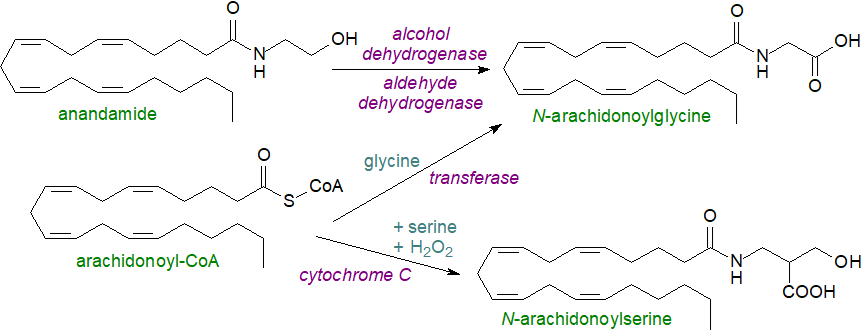 |
| Figure 2. Biosynthesis of N-arachidonoylglycine and N-arachidonoylserine. |
N-Arachidonylglycine has been shown to suppress inflammatory pain. It does not bind to the CB1/2 receptors for endocannabinoids, but it is a ligand for other G protein-coupled receptors such as GPR55 (as is lysophosphatidylinositol), GPR18 and GPR92, and it may have a role in regulating tissue levels of anandamide by inhibiting the fatty acid amide hydrolase. In this respect, the dividing line with endocannabinoids is tenuous. Among the reported physiological effects of N‑arachidonylglycine are calcium mobilization, control of apoptosis, anti-inflammatory actions, alleviation of neuropathic pain, and regulation of intraocular pressure. It is a substrate for cyclooxygenase-2 (COX-2), producing glycine conjugates of prostaglandins, and it may divert the biosynthetic pathway from the pro-inflammatory PGE2 towards the anti-inflammatory J-series prostaglandins. Similarly, N-docosahexaenoyl-glycine and its epoxide metabolites are reported to be multifunctional lipid mediators.
N-Palmitoylglycine is produced in most tissues, but especially skin and spinal cord, and it has a role in sensory neuronal signalling by acting as a modulator of calcium influx and nitric oxide production. There are reports of an involvement in gynaecological disorders, and its concentration is elevated in breast cancer. Activation of cation channels such as GPR132 mediate its signalling properties.
N‑Oleoylglycine was first detected in mouse neuroblastoma cells, and it is now known to regulate body temperature by inducing hypothermia and to influence locomotion; it is produced by the cytochrome C route. There is a report that it promotes adipogenesis via stimulation of the CB1 receptor, implying that it is an endocannabinoid with the potential to increase insulin sensitivity and suppress obesity and diabetes, and it has powerful antinicotine addiction properties (as has N‑oleoylalanine). It is a precursor for oleamide by oxidative cleavage in a reaction catalysed by peptidylglycine α‑amidating monooxygenase (other acylglycines may react in the same way).
In brown adipose tissue, a secreted enzyme, peptidase M20 domain containing 1 (PM20D1), is able to catalyse both the condensation of fatty acids and amino acids to generate N-acyl amino acids and the reverse hydrolytic reaction. In mice, N-oleoyl-leucine and N‑oleoylphenylalanine synthesised in this way have been shown to bind to mitochondria to increase the rate of uncoupled respiration in cells, while treatment with N-oleoyl-leucine improved glucose homeostasis, increased energy expenditure and reduced body weight.
N-Acylserines have been detected at trace levels in bovine brain with the palmitoyl and stearoyl forms being most abundant; N‑palmitoylserine is reported to be neuroprotective against traumatic brain injury. A novel lipid that is presumably derived from palmitoylserine, i.e., N-palmitoyl-O-phosphocholineserine, has been found in patients with the genetic disorder Niemann-Pick disease type C1. It was originally termed "lysosphingomyelin 509", and it may be functionally if not structurally related to sphingosylphosphorylcholine (and is illustrated and discussed here..). N‑Oleoylserine is a bone constituent that stimulates osteoblast proliferation through its action on a G protein-coupled receptor.
N-Arachidonoylserine has been found in tissues, and while it does not bind strongly to cannabinoid receptors, it is a potent vasodilatory agent on rat arteries in vitro, and it activates certain calcium channels in neurons. In addition, it was found to suppress the formation of reactive oxygen species and production of nitric oxide (NO) and tumour necrosis factor (TNF) in a murine macrophage cell line. It is presumed to be synthesised by an analogous mechanism to that for anandamide but via N‑arachidonoylphosphatidylserine, although mechanisms analogous to those described above for arachidonoylglycine are possible. At least three other arachidonoyl amino acids, i.e., of γ‑aminobutyric acid, alanine and asparagine, occur naturally and inhibit pain, suggesting that such biomolecules may be integral to pain regulation and perhaps have other functions in mammals. As with oleoylglycine, conversion to primary fatty amides has been demonstrated in vitro, and it is possible that this occurs in vivo.
N‑Linoleoylalanine has anti-inflammatory action that is mediated by a GPR18 response. N‑Acylaspartates with 16:0, 18:2 and 20:4 fatty acid moieties attached to the amino group of aspartic acid that may inhibit Hedgehog signalling but do not interact with endocannabinoid receptors have been isolated from animal tissues. N‑Stearoyltyrosine fed orally to mice was found to ameliorate obesity by inhibiting absorption of dietary fat, promoting lipolysis and reducing lipogenesis, while the N‑palmitoyl analogue has an anti-proliferative action in cancer cells. N-stearoyl tryptophan has been detected in plasma from breast cancer patients. N‑(17‑Hydroxy)-linolenoyl-L-glutamine (volicitin), N-(17-hydroxy)-linoleoyl-L-glutamic acid and related lipoamino acids have been found in insect larvae, and their presence in oral secretions elicits a defence response in plants. Male nematodes produce (2E,4Z,7Z)-N-decatrienoyl-glutamine to accelerate larval development.
N-Arachidonoyl amino acid and vanilloid derivatives are minimally oxidized by COX-2, but they are good substrates for the 12S- and 15S‑lipoxygenases, although it is not yet clear whether this leads to inactivation of these lipids or rather converts them to new lipid mediators. While the fatty acid amide hydrolase (FAAH) will cleave the N-acyltaurines and N-arachidonoylglycine to the corresponding fatty acid and amino acid, the other N-acyl amino acids are not affected, although they are potent inhibitors of the enzyme (their catabolic fate is uncertain).
5. Lipid-Glutathione Adducts in Animals
Glutathione is the tripeptide, γ-L-glutamyl-L-cysteinylglycine, and is most abundant thiol-containing small molecule (3 to 4 mM) in animal cells where it is located mainly in the cytosol. It has a major defensive role in combating oxidative stress by undergoing oxidation to glutathione disulfide while reducing lipid (and other) hydroperoxides to hydroxides, and it reacts with lipid oxidation products to produce lipid-glutathione adducts by the action of glutathione S‑transferases. By conversion to glutathione conjugates in this way, eicosanoids and α,β‑unsaturated aldehydes (e.g., trans,trans-2,4-decadienal) and malondialdehyde can be deactivated or detoxified.
In contrast, the eicosanoid 5-hydroperoxyeicosapentaenoic acid (5-HPETE) is converted to a glutathione adduct as an intermediate in the biosynthesis of the cysteinyl leukotrienes, and related resolvin/maresin adducts are synthesised in the same manner, as described elsewhere on this website.
6. Simple Lipoamino Acids and N-Acylamides from Bacteria
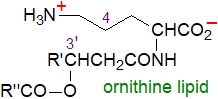 Ornithine lipids:
A variety of lipoamino acids have been isolated from bacteria, of which the best known is probably the zwitterionic
N-acyl-ornithine derivative illustrated, which is widely distributed
among prokaryotes (perhaps 50% of all species), but especially Gram-negative bacteria and other eubacteria
where it is located mainly in the outer membrane, although some is present in the cytoplasmic membrane.
These lipids contain a non-hydroxy fatty acid with an estolide linkage to the hydroxyl group of a 3‑hydroxy acid
(often but not always C16 or C18) and thence via an amide bond to the α‑amino group of ornithine.
It may be relevant that such fatty acid linkages are present in the bacterial endotoxin lipid A.
Lyso-ornithine lipids occur in some bacteria, i.e., lacking the O-acyl fatty acid.
Although they are normally minor components, ornithine lipids can assume major proportions when phosphate is limiting or in response to stress.
Under normal conditions, they may help bacteria to avoid immune surveillance by host animals, but during phosphate deprivation, they activate
Toll-like receptor 4 (TLR4) and trigger inflammatory reactions.
Ornithine lipids:
A variety of lipoamino acids have been isolated from bacteria, of which the best known is probably the zwitterionic
N-acyl-ornithine derivative illustrated, which is widely distributed
among prokaryotes (perhaps 50% of all species), but especially Gram-negative bacteria and other eubacteria
where it is located mainly in the outer membrane, although some is present in the cytoplasmic membrane.
These lipids contain a non-hydroxy fatty acid with an estolide linkage to the hydroxyl group of a 3‑hydroxy acid
(often but not always C16 or C18) and thence via an amide bond to the α‑amino group of ornithine.
It may be relevant that such fatty acid linkages are present in the bacterial endotoxin lipid A.
Lyso-ornithine lipids occur in some bacteria, i.e., lacking the O-acyl fatty acid.
Although they are normally minor components, ornithine lipids can assume major proportions when phosphate is limiting or in response to stress.
Under normal conditions, they may help bacteria to avoid immune surveillance by host animals, but during phosphate deprivation, they activate
Toll-like receptor 4 (TLR4) and trigger inflammatory reactions.
In most bacteria, biosynthesis of ornithine lipids occurs in two steps via sequential acyl-ACP-dependent acylation of ornithine by two different acyltransferases. The first N-acyltransferase (OlsB) transfers a 3-hydroxy fatty acyl residue from acyl carrier protein to the α-amino group of ornithine forming a lyso-ornithine lipid, which is then acylated by an O-acyltransferase (OlsA) to produce the final ornithine lipid. At least one species contains a bifunctional acyltransferase in which the N‑terminal domain is responsible for the O‑acyltransferase reaction, whereas the C-terminal domain carries out the N‑acyltransferase reaction. In some bacteria, either the ester- or amide-linked fatty acid has a hydroxyl group in position 2.
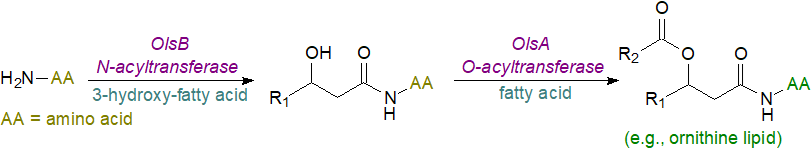 |
| Figure 3. Biosynthesis of bacterial ornithine and related lipoamino acids. |
Analogous lipids in which the ornithine moiety is hydroxylated in position 4 by a hydrolase are known with both modifications inserted post synthesis of the basic lipid. Indeed, many bacteria contain comparable lipids derived from 4-hydroxy-ornithine in which the 4‑hydroxyl group is also esterified by a long-chain fatty acid. In the nodule bacterium Mesorhizobium loti, the ornithine moiety is surprisingly mainly of the D‑configuration. Mono-, di- and trimethylornithine lipids are formed by sequential methylation of the ornithine moiety by an N‑methyl transferase requiring S‑adenosylmethionine in Planctomycetes. The marine roseobacter Ruegeria pomeroyi contains an analogue of the ornithine lipids in which the amine component is aminopropane sulfonic acid, i.e., it is a sulfonolipid.
Other lipoamino acids and N-acylamides: Neisseria meningitidis is a Gram-negative opportunistic pathogen that is responsible for causing such human diseases as septicaemia and meningitis. It has been shown to contain lipoamino acids consisting of ornithine or lysine linked to saturated and monoenoic fatty acids, some with 3-hydroxyl groups. These bind to the G‑protein coupled receptor S1PR4 in host immune cells for which sphingosine-1-phosphate is the usual ligand.
In human intestines, the commensal microbiome produces N-acyl amino acids and amides (with various fatty acyl components) of which six main families were initially identified with the potential to interact with receptors in the intestinal tract that affect the metabolism of the host, although many more are now known. These are N‑acylglycine/serine, N‑acyloxyacyllysine, N‑acyloxyacylglutamine, N‑acyllysine/ornithine, N-acylalanine and N-acylserinol. Bacteroides vulgatus produces N-acyl-3-hydroxy-palmitoylglycine ('commendamide'), biosynthesis of which starts with N-acylation of glycine with a β‑hydroxy fatty acid via an N-acyltransferase (encoded by the glsB gene), followed by O-acylation of the free hydroxyl with a secondary fatty acid via an O‑acyltransferase (encoded by glsA) to produce a diacylated amino acid lipid. The mono-acylated forms resemble the long-chain N‑acyl-amides that are mammalian signalling molecules through activation of G-protein–coupled receptors (GPCR G2A/132) and may interact with the immune system of the human host. Many more such lipoamino acids have been detected by sensitive mass spectrometric methods in Escherichia coli and in Gram positive bacteria with C12 to C17 fatty acyl moieties, including N‑acylleucine (or isoleucine) derivatives in Deleya marina, N-acyl-D-asparagine in Bacillus pumilus and N‑acylserine in Serratia sp.
The primary function of these N-acyl lipoamino acids is presumably to interact with bacterial receptors, but there is evidence that they have structural similarity (chemical mimicry) to those of their eukaryotic hosts with some acting as antagonists for receptors in the host, e.g., N‑palmitoylserinol, N‑3‑hydroxypalmitoylornithine and N‑myristoylalanine activate GPR119, sphingosine-1-phosphate receptor 4 (S1PR4), and G2A, respectively.
 Among the simple fatty acyl amides, 13‑docosenamide or erucamide is secreted by many
bacteria when they are grown on a medium containing glucose, and it may be relevant to host-bacterial interactions.
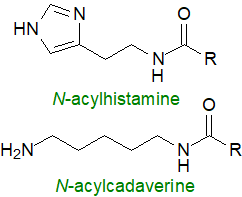
N-Acyl histamines have been found in the intestinal microbiome of patients with HIV and intestinal disorders, while cadaverine linked
to short-chain fatty acids contributes to neurological problems.
N-Acyl derivatives of homospermidine (undecane with amine groups at the 1-, 6- and 11‑positions) with fatty acids attached to
each of the amine groups have been found in fruiting bodies of Myxococcus Xanthus, where they may be part of its defence strategy.
Among the simple fatty acyl amides, 13‑docosenamide or erucamide is secreted by many
bacteria when they are grown on a medium containing glucose, and it may be relevant to host-bacterial interactions.
N-Acyl histamines have been found in the intestinal microbiome of patients with HIV and intestinal disorders, while cadaverine linked
to short-chain fatty acids contributes to neurological problems.
N-Acyl derivatives of homospermidine (undecane with amine groups at the 1-, 6- and 11‑positions) with fatty acids attached to
each of the amine groups have been found in fruiting bodies of Myxococcus Xanthus, where they may be part of its defence strategy.
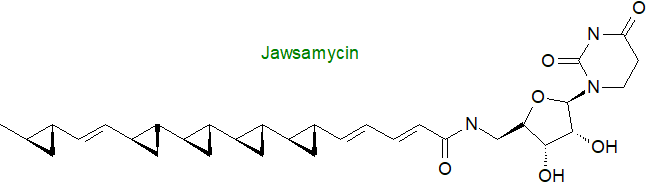
Marine bacteria of the family Rhodobacteraceae contain glutamine lipids, which are closely related in structure to the ornithine lipids (some contain both). Structurally related lipids with lysine, serine, glycine, glutamine and taurine residues occur in microorganisms such as the gliding bacterium Cytophaga (Flavobacterium) johnsonae and the Gram-negative marine Cyclobacterium marinus. (N‑(5‑Methyl)hexanoyltyrosine and N-(7-methyl)octanoyl-tyrosine or phenylalanine are reported from Olivibacter sp. (Bacteroidetes). Further lipoamino acid forms of increasing complexity have been characterized, including molecules with a long-chain alcohol moiety linked to the carboxyl group of the amino acid (such as siolipin A from Streptomyces sp.). Arguably, the most unusual fatty acid found in amide linkage is jawsamycin, which contains multiple cyclopropyl groups and is linked to a nucleoside. It is synthesised by the bacterium Streptoverticillium fervens by a polyketide pathway and has antifungal properties.
Such lipoamino acids/amides are necessary for many different purposes in bacteria depending upon species, and it does not seem possible to generalize. They have been implicated in temperature and stress tolerance, and they are produced when phosphorus is limiting; they may be recognized by plant defence systems or be necessary for symbiotic relationships. In Gram-negative bacteria, they may stabilize the outer membrane by counteracting the negative charge of the lipopolysaccharides. Some have interesting and potentially useful pharmacological properties as antibacterial and antifungal agents, as well as against some human metabolic diseases.
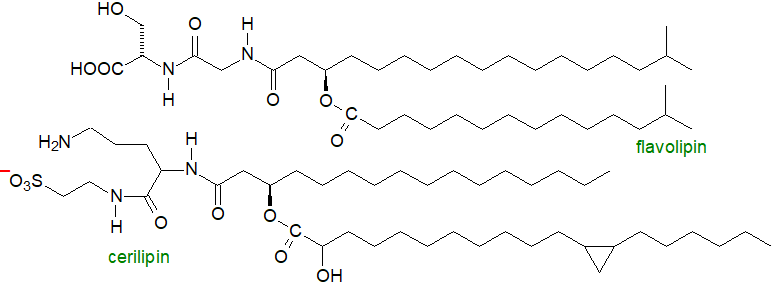
Dipeptido lipids: Among the lipids found in Flavobacterium sp. is one containing a glycine-serine dipeptide linked to branched chain acids in a similar manner to the ornithine lipids and termed 'flavolipin'. Biosynthesis is by a comparable mechanism to that of the ornithine lipids. The same lipids (but termed 'lipid 654' from the molecular weight of the main isomer, and 'lipid 430' with a single fatty acid constituent) together with mono-amino analogues, have been found in common oral and intestinal Bacteroidetes, and they have been detected in serum and brain samples from healthy subjects, presumably after uptake via the intestines. The former is an agonist for human and mouse Toll-like receptor 2 and it may be beneficial towards atherosclerosis. Intriguingly, it may be a biomarker of multiple sclerosis, as it is present at significantly lower levels in the serum of MS patients in comparison to healthy individuals. Cerilipin is a taurine-containing ornithine lipid from Gluconobacter cerinus. A related glycine-serine-ornithine tripeptide family of lipids has been isolated from the Bacteroidetes genus Chitinophaga.
Many pathogenic bacteria, including Actinobacteria (including Streptomyces sp.) and Mycobacteria, produce lipopeptides in the form usually of diacyl dipeptides linked to fatty acids with isonitrile substituents (kupyaphores), and these are described in our web page on isonitriles. The more complex microbial lipopeptides have their own web page.
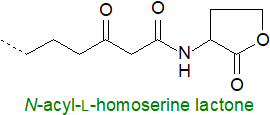 N-Acyl-L-homoserine lactones
are produced by many Gram-negative bacteria.
The fatty acid components can vary in chain length from C4 to C18, sometimes with one or two double bonds (in position 2
(trans) and/or more central positions, e.g., n‑7 (cis)), with hydroxyl or keto groups in position 3 and/or
with methyl branches.
The first molecule of this type to be identified was N‑3‑oxohexanoyl-L-homoserine lactone, which induces
bioluminescence, from the marine bacterium Vibrio fischeri, although different strains of this organism are now known to produce
other isomers with variable chain-lengths and the presence or absence of 3‑oxo or 3‑hydroxyl groups.
Some bacteria produce one molecular form only, but others can secrete many different forms.
Other quorum sensing molecules include the lipidic 2-heptyl-4-quinolone and 2-heptyl-3-hydroxy-4(1H)-quinolone, while the photosynthetic
bacterium Rhodopseudomonas palustris synthesises p‑coumaroyl-homoserine lactone.
N-Acyl-L-homoserine lactones
are produced by many Gram-negative bacteria.
The fatty acid components can vary in chain length from C4 to C18, sometimes with one or two double bonds (in position 2
(trans) and/or more central positions, e.g., n‑7 (cis)), with hydroxyl or keto groups in position 3 and/or
with methyl branches.
The first molecule of this type to be identified was N‑3‑oxohexanoyl-L-homoserine lactone, which induces
bioluminescence, from the marine bacterium Vibrio fischeri, although different strains of this organism are now known to produce
other isomers with variable chain-lengths and the presence or absence of 3‑oxo or 3‑hydroxyl groups.
Some bacteria produce one molecular form only, but others can secrete many different forms.
Other quorum sensing molecules include the lipidic 2-heptyl-4-quinolone and 2-heptyl-3-hydroxy-4(1H)-quinolone, while the photosynthetic
bacterium Rhodopseudomonas palustris synthesises p‑coumaroyl-homoserine lactone.
N-Acyl-L-homoserine lactones are used in a form of intercellular signalling termed 'quorum sensing', which controls gene expression in response to the population density of the species to result in coordinated regulation of a range of group-level behaviours, including production of secondary metabolites and virulence factors, bioluminescence and biofilm formation. These molecules can diffuse freely through the bacterial membrane, and when they reach a threshold concentration in a particular environment (nanomolar range), they bind to their intracellular receptor/activator proteins (LuxR-type) in the inner membrane or in the cytoplasm to induce the expression of innumerable genes, one consequence of which is to establish a feed-forward loop that promotes synchronous gene expression to allow the bacterial population to act in effect as a multicellular organism. Homoserine lactones (and their hydrolysis products by ring opening) can promote large-scale remodelling in lipid membranes to generate long microtubules in a reversible manner on the surface of the bilayer and thereby enable molecular transport across the bilayer.
For biosynthesis, an acyl carrier protein-linked fatty acid, such as a 3-oxo isomer illustrated, is reacted with S-adenosylmethionine (SAM) by means of an acyl-homoserine lactone (AHL) synthase (LuxI). The mechanism begins with nucleophilic attack on the 1‑carbonyl carbon of the fatty acyl moiety by the amine of SAM, followed by nucleophilic attack on the γ-carbon of SAM by its own carboxylate oxygen to produce the lactone.
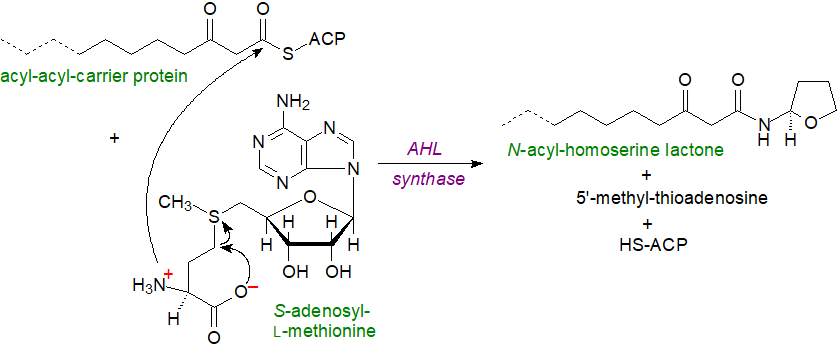 |
| Figure 4. Biosynthesis of an N-acyl-homoserine lactone. |
Homoserine lactones produced by the intestinal microbiota serve as mediators in host-microbe communication to influence both microbial behaviour and the cellular responses of their hosts by interacting with intestinal epithelial cells to regulate inflammation, barrier integrity and immune modulation. As they are de-activated by lactonases, acylases and/or oxidoreductases (quorum quenching), it is hoped that drugs will be found to stimulate these enzymes and reduce the virulence of pathogenic bacteria. In contrast, certain plants are able to detect quorum sensing signals with the potential to permit them to alter the outcomes to their own benefit via the signalling action of oxylipins and salicylic acid, and they can hydrolyse N‑acyl-L-homoserine lactones with fatty acid amide hydrolases to generate L‑homoserine, which encourages plant growth.
8. Analysis
The main problems in the analysis of simple lipoamino acids relate to the low levels at which they occur naturally, and there is a concern that artefactually high results might be obtained because of the physiological effects of sampling methods and/or artefact formation during sample storage. Until recently, high-performance liquid chromatography with fluorescent detection or gas chromatography-mass spectrometry with selected ion monitoring were most used for the purpose, but liquid chromatography allied to tandem mass spectrometry with electrospray ionization would probably be the preferred method now.
Recommended Reading
- Blair, I.A. Analysis of endogenous glutathione-adducts and their metabolites. Biomed. Chromatogr., 24, 29-38 (2010); DOI.
- Bortoletto, R., Comacchio, C., Garzitto, M., Piscitelli, F., Balestrieri, M. and Colizzi, M. Palmitoylethanolamide supplementation for human health: A state-of-the-art systematic review of Randomized Controlled Trials in patient populations. Brain Behav. Immun.-Health, 43, 100927 (2025); DOI.
- Burstein, S.H. N-Acyl amino acids (elmiric acids): endogenous signaling molecules with therapeutic potential. Mol. Pharmacol., 93, 228-238 (2018); DOI.
- Cai, F.J. and Wang, C.H. Comprehensive review of the phytochemistry, pharmacology, pharmacokinetics, and toxicology of alkamides (2016-2022). Phytochemistry, 220, 114006 (2024); DOI.
- Cohen, L., and 17 others. Commensal bacteria make GPCR ligands that mimic human signalling molecules. Nature, 549, 48–53 (2017); DOI.
- Dembitsky, V.M. Microbiological aspects of unique, rare, and unusual fatty acids derived from natural amides and their pharmacological profile. Microbiol. Res., 13, 377-417 (2022); DOI.
- Greger, H. Alkamides: a critical reconsideration of a multifunctional class of unsaturated fatty acid amides. Phytochem. Rev., 15, 729-770 (2016); DOI.
- Hamzah, K.A., Turner, N., Nichols, D. and Ney, L.J. Advances in targeted liquid chromatography-tandem mass spectrometry methods for endocannabinoid and N-acylethanolamine quantification in biological matrices: A systematic review. Mass Spectrom. Rev., 44, 513-538 (2025); DOI.
- Ivashkevich, D., Ponomarenko, A., Manzhulo, I. and Dyuizen, I. Mechanistic insights into N-oleoylethanolamide-mediated hepatoprotection via PPAR-α. Biocell, 49, 607-627 (2025); DOI.
- Kumar, L. and others. Molecular mechanisms and applications of N-acyl homoserine lactone-mediated quorum sensing in bacteria. Molecules, 27, 7584 (2022); DOI.
- Liu, S.W. and others. Aminolipids in bacterial membranes and the natural environment. ISME J., 18, wrae229 (2024); DOI.
- Mannochio-Russo, H. and others. The microbiome diversifies long-to short-chain fatty acid-derived N-acyl lipids. Cell, 188, 4154-4169 (2025); DOI.
- Mock, E.D., Gagestein, B. and van der Stelt, M. Anandamide and other N-acylethanolamines: A class of signaling lipids with therapeutic opportunities. Prog. Lipid Res., 89, 101194 (2023); DOI.
- Nani, M.F., Rinaldi, M.M., Miraglia, M., Amico, R., De Cicco, P. and Romano, B. N-Acylethanolamines in cancer: mechanisms and therapeutic potential of lipid regulators of tumor behavior. Prog. Lipid Res., 100, 101358 (2025); DOI.
- Prakash, S.A. and Kamlekar, R.K. Function and therapeutic potential of N-acyl amino acids. Chem. Phys. Lipids, 239, 105114 (2021); DOI.
- Verhoeckx, K.C.M., Voortman,T., Balvers,M.G.J., Hendriks,H.F.J., Wortelboer,H.M. and Witkamp,R.F. Presence, formation and putative biological activities of N‑acyl serotonins, a novel class of fatty-acid derived mediators, in the intestinal tract. Biochim. Biophys. Acta, Lipids, 1811, 578-586 (2011); DOI.
- Wellner, N., Diep, T.A., Janfelt, C. and Hansen, H.S. N-Acylation of phosphatidylethanolamine and its biological functions in mammals. Biochim. Biophys. Acta, Lipids, 1831, 652-662 (2013); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: December 2025 | ||
© The LipidWeb is open access and fair use is encouraged but not text and data mining, AI training, and similar technologies.
