Miscellaneous Lipid Sulfates and Sulfonates
Sulfation is a common reaction in lipid biochemistry for many reasons, but especially in that it can enable solubilization of a lipid
for transport or elimination, or it can convert a complex lipid to a highly anionic state for particular purposes in membranes, or it can act
in the defence against predators.
Sulfonolipids have the sulfur atom linked directly to a carbon atom,
while in lipid sulfates the sulfur is linked to the lipid component by an oxygen atom.
Many interesting lipid sulfates and sulfonates, which are vital for the organisms that produce them, are described below,
 but other
important sulfur-containing lipids are discussed elsewhere on this website for reasons of their relevance to other topics.
For example, sphingolipid sulfates are essential for the function of the brain and kidney,
amongst other tissues, and they have their own web page on this site.
Other such molecules include seminolipid,
sterol sulfates, phosphatidylsulfocholine,
lipo-chitooligosaccharides (Nod factors)
and some Archaeal lipids (extremophiles such as those that tolerate high salinity can
contain mono- and bis-sulfated diglycosyl diphytanylglyceroldiethers and phosphatidylglycerosulfate).
Coenzyme A and lipoic acid are of course other important
sulfur containing lipids, though not sulfates.
but other
important sulfur-containing lipids are discussed elsewhere on this website for reasons of their relevance to other topics.
For example, sphingolipid sulfates are essential for the function of the brain and kidney,
amongst other tissues, and they have their own web page on this site.
Other such molecules include seminolipid,
sterol sulfates, phosphatidylsulfocholine,
lipo-chitooligosaccharides (Nod factors)
and some Archaeal lipids (extremophiles such as those that tolerate high salinity can
contain mono- and bis-sulfated diglycosyl diphytanylglyceroldiethers and phosphatidylglycerosulfate).
Coenzyme A and lipoic acid are of course other important
sulfur containing lipids, though not sulfates.
The best known and probably most abundant of the sulfonolipids in nature is sulfoquinovosyldiacylglycerol or 1,2-di-O-acyl-3-O-(6'-deoxy-6'-sulfo-α-D-glucopyranosyl)-sn-glycerol, a key component of the photosynthetic mechanism of higher plants and other photosynthetic organisms, and because of its relationship to the mono- and digalactosyldiacylglycerols, it is discussed in a separate web page.
1. Chlorosulfolipids
The phytoflagellate Ochromonas danica (a chrysophyte alga) contains linear alkanes (C22 and C24) substituted with sulfate groups and with both sulfate groups and chlorine atoms, i.e., they are chlorosulfolipids, the discovery and exploration of which are largely associated with Thomas H. Haines. Generally, there are two sulfate groups in the 1 and 14 positions of the C22 alkyl chain (positions 1 and 15 of the C24 compounds), and there can be one to six chlorine atoms in various positions. They constitute approximately 15% of the total lipids, but 90% of the polar lipids of the flagella. Eight chlorosulfolipids have now been characterized from this organism, and some representative structures are illustrated. Of these, 'danicalipin A' (the last formula in the figure) is the major component, and the position and stereochemistry of each of the substituents has been determined.
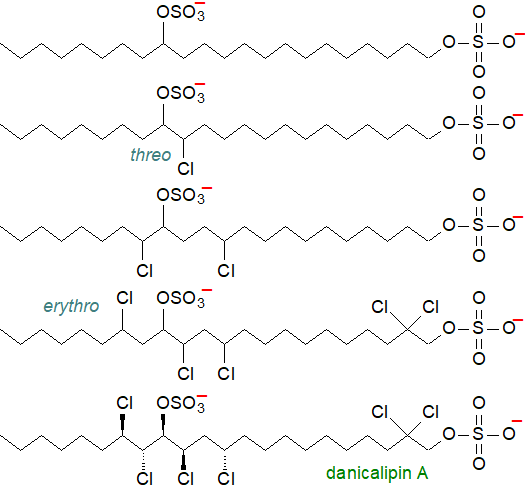 |
| Figure 1. Structures of some chlorosulfolipids of Ochromonas danica. |
Many aspects of the biosynthesis of these unusual lipids remain to be confirmed, but docosanoic acid is hydroxylated at C14, before reduction to the 1,14-diol and sulfation. Chlorine atoms are presumed to be inserted into the saturated alkyl chain of the 1,14-diol sulfate in a stepwise fashion by a free radical process involving chlorinases, which have yet to be characterized. The end points of the biosynthetic process are 2,2,11,13,15,16-hexachloro-1,14-docosanediol disulfate and 2,2,12,14,16,17-hexachloro-1,15-tetracosanediol disulfate. As O. danica is unable to remove the sulfate groups, the lipids are remarkably inert metabolically. When this organism is grown with excess bromide ions, bromosulfolipids are produced with the same positional and stereochemical selectivity as in danicalipin A.
Such a high proportion of chlorosulfolipids (and an absence of phospholipids) in the flagella of O. danica implies that they must be the major constituents of the membranes of this organelle and must be differentiated in some manner from the contiguous exterior surface membranes. At first glance, it is not easy to understand how such lipids, which are highly soluble in water and carry a polar substituent in the centre of the hydrocarbon chain, can form a membrane bilayer. This can only be possible if there are some positively charged ions buried deep in the hydrocarbon layer that shield the negative sulfate groups, and Haines has postulated that an unidentified molecule, possibly a divalent metal ion or protein bearing charged residues, offsets the negative charge of the sulfate group at the physiological pH. Physical chemical studies suggest that danicalipin A inserts into lipid bilayers in the headgroup region to thin the bilayer and fluidize it, permitting even saturated lipids to form fluid bilayers. The anionic lipid head groups may serve as a proton-conducting pathway along the surface of membranes.
Since the initial studies, a range of further chloro- and bromosulfolipids have been found in algae and other organisms, some as toxins affecting shellfish. Many of the chlorosulfolipids found in fresh-water algae are the same as those of O. danica, but others have some distinctive features, including a chlorovinylsulfate group. Complex multi-chloro-sulfolipids isolated from the digestive glands of toxic mussels are causative agents of diarrhetic shellfish poisonings, which tend to be associated with marine algal blooms, and mytilipin B isolated from the culinary mussel Mytilus galloprovincialis contains an ester-linked fatty acid in addition to eleven chlorine atoms and a sulfate moiety; the stereochemistry of this molecule is obviously highly complex.

2. Taurine-containing Lipids (Taurolipids)
 Several lipids have been found that are conjugated to taurine (2-aminoethanesulfonic acid), of which the best known are certain
bile acids (discussed elsewhere on this site).
Taurine itself is synthesised from cysteine via oxidation and decarboxylation reactions and is plentiful in animal tissues, especially the brain,
but is very rare in plants.
Several lipids have been found that are conjugated to taurine (2-aminoethanesulfonic acid), of which the best known are certain
bile acids (discussed elsewhere on this site).
Taurine itself is synthesised from cysteine via oxidation and decarboxylation reactions and is plentiful in animal tissues, especially the brain,
but is very rare in plants.
N-acyltaurines: These lipids are endogenous bioactive acyl-amino acids and were first encountered in marine invertebrates, but there is increasing interest in a range of fatty N‑acyltaurines isolated from both the central nervous system and peripheral tissues of mice and the islets of Langherans and in plasma of humans that are signalling mediators. In brain, the fatty acyl groups are largely long-chain saturated, but in liver and kidney, arachidonoyl and docosahexaenoyl species predominate, while N-oleoyltaurine is most abundant in plasma. They are synthesised through enzyme-dependent conjugation of fatty acyl-CoA esters with taurine via the action of the peroxisomal bile acid-CoA:amino acid N‑acyltransferase in the liver of mice and humans. Like other fatty acyl amides in animals, the levels of these metabolites are controlled by the fatty acid amide hydrolase (FAAH).

N-Acyltaurines, and specifically N-oleoyltaurine, improve insulin sensitivity and augment the secretion of the anti-diabetic hormone GLP‑1, insulin and glucagon through stimulation of GPR119, an abundant receptor in pancreatic and intestinal endocrine cells. In kidney, N‑acyltaurines have been shown to activate receptors that control calcium channels. N‑Arachidonoyl- and N‑oleoyltaurine induce a significant inhibition of a cancer cell line in vitro, while the 20:0 and 24:0 fatty acyl analogues are produced endogenously in vivo and regulate the healing of skin wounds in mice. Arachidonoyltaurine is an excellent substrate for lipoxygenases, but the functions of the resulting hydroxyeicosatetraenoyltaurines have yet to be determined. Docosahexaenoyl-taurine accumulates in bile and plasma in mice and in plasma in humans fed a diet enriched in omega-3 fatty acids; intestinal hydrolysis of triacylglycerols and lipid absorption was inhibited and resulted in lower plasma triacylglycerol levels and protection against hepatic accumulation of lipids.
 Another class of sulfonolipids has a substituted taurine component, i.e., an N,O-acylated cysteinolic acid-containing head group carrying
various different (α‑hydroxy)carboxylic acids, and now termed cysteinolides from a marine Roseobacter sp., was once erroneously
identified as homotaurine analogues (they have structural and functional relationships to certain of the capnoids discussed below).
Comparable lipids, tentatively identified as N-butyramide cysteine glycerols, have been found in the anaerobic sulfate-reducing bacterium
Desulfatibacillum alkenivorans grown in phosphorus-deprived conditions.
Another class of sulfonolipids has a substituted taurine component, i.e., an N,O-acylated cysteinolic acid-containing head group carrying
various different (α‑hydroxy)carboxylic acids, and now termed cysteinolides from a marine Roseobacter sp., was once erroneously
identified as homotaurine analogues (they have structural and functional relationships to certain of the capnoids discussed below).
Comparable lipids, tentatively identified as N-butyramide cysteine glycerols, have been found in the anaerobic sulfate-reducing bacterium
Desulfatibacillum alkenivorans grown in phosphorus-deprived conditions.
Other taurolipids: Aside from the bile acids, the first taurolipids to be recognized were novel C18 hydroxy acids (3, 4 or 5 hydroxyl groups) with an amide link to taurine, which were isolated from the ciliated protozoan Tetrahymena. The hydroxyl on carbon 3 is acylated with normal fatty acids (approx. 30% 16:0), i.e., it is an estolide, and in one variant, carbon 7 is acylated in the same way. The deacylated backbone has been termed ‘lipotaurine’. Biosynthesis involves conjugation of stearic acid with taurine, with subsequent sequential insertion of hydroxyl groups.
 |
||||
| Taurolipid | R1 | R2 | R3 | R4 |
|---|---|---|---|---|
| Taurolipid A | OH | OH | H | H |
| 7-Acyltaurolipid A | CH3(CH2)14COO | OH | H | H |
| Taurolipid B | OH | OH | OH | H |
| Taurolipid C | OH | OH | OH | OH |
Marine invertebrates are a rich source of unusual lipids, including some containing taurine. For example, irciniasulfonic acid B, isolated from a marine sponge, Ircinia sp., comprised 3-methyl-8-hydroxy-dec-2-enoic acid conjugated to taurine with various unusual fatty acids linked to the hydroxyl group; carteriosulfonic acids and taurospongin A are related lipids found in sponges. In addition, compounds termed copepodamides and consisting of taurine connected by an amide linkage to isoprenoid hydroxy fatty acids, to which polyunsaturated fatty acids are linked as estolides, are synthesised by marine and fresh-water copepods (zooplankton). At minute concentrations, these act as a signal to bloom-forming dinoflagellates (phytoplankton) and induce production of paralytic shellfish toxins, presumably as a defence response. An unusual N-acyltaurine, linked to a dihydroxy acid, has been found in a sea urchin. Cerilipin is a taurine-containing ornithine lipid from the bacterium Gluconobacter cerinus (see our web page on lipoamino acids).

A tauroglycolipid, 1,2-diacyl-3-glucuronopyranosyl-sn-glycerol taurineamide, was isolated from a seawater bacterium Hyphomonas jannaschiana, which has another unusual feature in that it lacks phospholipids. The main fatty acyl chains are saturated and monoenoic (C16 to C20). Other unusual sulfolipids from bacteria are described below.

A novel ganglioside with the carboxyl group of N-acetylneuraminic acid amidated by taurine has been isolated from brain samples from patients with Tay-Sachs disease, a well-known glycosphingolipid (GSL) storage disease. As this lipid is not present in normal brains, it seems probable that it is associated with the pathogenesis of the disease, possibly as a means of removing the excess of ganglioside GM2 from the tissue.
3. Other Lipid Sulfates
The first sulfated fatty acids to be identified were the caeliferins, which were found in the oral secretions of a type of grasshopper and elicit the release of volatile organic compounds as a defence response when the insects graze upon plants. Caeliferin A (illustrated) has two sulfate groups, while caeliferin B has a carboxyl conjugated to glycine replacing the terminal sulfate. Synthetic caeliferin A (16:0) was found to induce significant production of jasmonic acid and ethylene in Arabidopsis at concentrations equivalent to those found in grasshopper saliva. When under attack by predators, nematodes can emit fatty acid sulfates, iso-methyl-tetradecanoic acid with sulfate moieties at positions 9 or 10, for defence purposes. The marine sponge Antho ridgwayi contains anthonoic acids with both sulfate and amine groups. Sulfamisterin from the fungus Pycnidiella sp., an analogue of the sphingoid bases, has antibiotic properties and it is a specific inhibitor of serine palmitoyltransferase, the key enzyme in the biosynthesis of such bases, while sulfate esters of oleate, linoleate and linolenate have now been found in some fungi, where they may be antifungal agents. The lipid designated IOR-1A is produced by bacteria to inhibit rosette development in choanoflagellates.

The outer membranes of the cell walls of virulent strains of Mycobacterium tuberculosis contain a number of trehalose-containing glycolipids. Some of these are sulfated and consist of a sulfated trehalose moiety to which up to four fatty acids are linked, including palmitate or stearate and two or three of the very-long-chain multi-branched phthioceranic acids that are characteristic of Mycobacteria. There are two main types; sulfolipid-I comprises a family of homologous 2‑palmitoyl(stearoyl)-3-phthioceranoyl(C37)-6,6'-bis(hydroxyphthioceranoyl)trehalose 2'-sulfates, while sulfolipid-II constitutes a family of homologous 2‑stearoyl(palmitoyl)-3,6,6'-tris(hydroxyphthioceranoyl)(C40)trehalose 2'-sulfates (see our web page on mycolic acids, where mycobacterial cell wall constituents are discussed at greater length).

Biosynthesis is initiated by a sulfotransferase that converts trehalose into trehalose-2-sulfate. As trehalose sulfates and especially sulfolipid-I increase the virulence of M. tuberculosis in human cells in vitro, an action enhanced by cord factor, a means of inhibition of their biosynthesis is a pharmacological target. The immunogenicity is dependent on the sulfate moiety, although the number of methyl substituents is a relevant factor. In an infection, they are reported to hijack the host cell membrane, affecting its order, fluidity and stiffness as well as interacting with the underlying cytoskeleton with profound effects on downstream signalling and on membrane-associated immune processes in the host. These lipids do not appear to be present in other Mycobacteria.
4. Sulfono-Analogues of Sphingolipids (Capnoids)
1-Deoxyceramide-1-sulfonate was first isolated from the Bryozoa Watersipora cucullata and consists of a long chain-base analogous to sphinganine but with a sulfonate moiety attached to carbon 1. The predominant fatty acid (64%) in this lipid in Nitzschia alba is 3E‑hexadecenoic acid, which is normally associated with the phosphatidylglycerol of plant chloroplasts. 1-Deoxyceramides where the fatty acid is sulfated have been characterized from anaerobic bacteria from the Black Sea.
The sulfonic acid analogue of ceramide, N-fatty acyl capnine is a major lipid component of gliding bacteria of the genera Cytophaga, Capnocytophaga, Sporocytophaga and Flexibacter where it may have a role in the motility of the organisms. These are organisms that can move over solid surfaces, but not through liquids, although they do not appear to have flagella or other organs of propulsion. Capnine is 2‑amino-3-hydroxy-15-methylhexadecane-1-sulfonic acid and occurs in the organisms both in the free state and as the N-acylated derivatives, though up to 20% of other homologues can occur, depending on species. Although it was not the first to be discovered, the generic term 'capnoid' is widely used for such lipids. The N-acyl fatty acids are much more heterogeneous and vary from C14 to C16 in chain-length, a high proportion with iso- or anteiso-methyl branches and hydroxyl groups in positions 2 or 3.
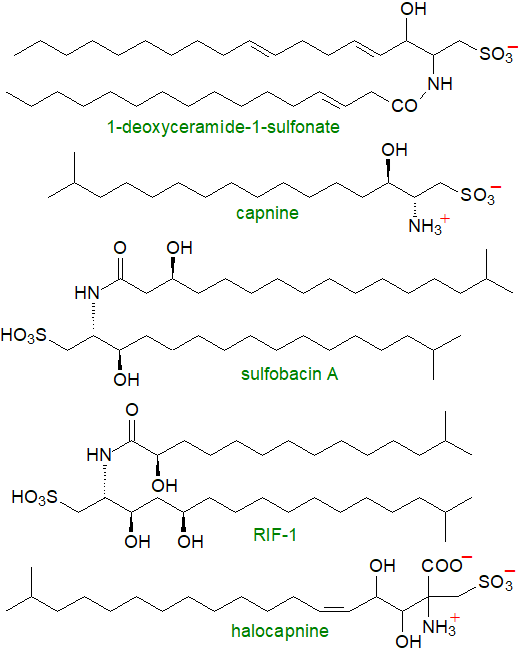 |
| Figure 2. Structures of some representative capnoids. |
The first steps in the biosynthesis of in capnine Bacteroidetes occur by a reaction of O-phospho-L-serine with sulfite catalysed by a cysteate synthase (CapA) to form cysteic acid (2R‑amino-3-sulfopropanoic acid); different types of CapA have been characterized from anaerobic and aerobic species. The product is then condensed with 13-methylmyristoyl-CoA by a cysteate-C-fatty acyltransferase (CapB) with pyridoxal phosphate as a cofactor to generate 3-ketocapnine as an intermediate before a reductase (CapC) requiring NADPH converts this to capnine. The reaction can be compared to the condensation of palmitoyl-coenzyme A with serine during the biosynthesis of sphingoid bases.
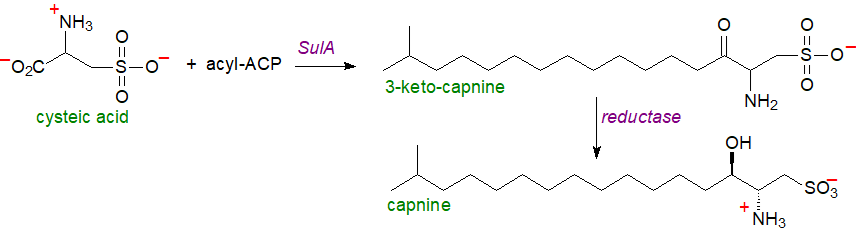 |
| Figure 3. Biosynthesis of capnine (Liu, Y. and others. DOI). |
Essentially the same lipids but termed sulfobacins A and B (differing in the presence of a hydroxyl group), i.e., (2R,3R)-3-hydroxy-2-[3R-hydroxy-15-methylhexadecanamido]-15-methylhexadecanesulfonic and (2R,3R)-3-hydroxy-15-methyl-2-[13-methyltetradecanamido]-hexadecanesulfonic acids, respectively, were found in several bacteria from the genera Chryseobacterium, Alistipes and Odoribacterin (all from the Bacteroidetes phylum) in the gut microbiome in mice. Indeed, capnine lipids are now known to be common in the genus Bacteroidetes, including environmental, pathogenic and human oral and intestinal microbiome bacteria, but their function is obscure. In animals, they are known to be antagonists of the von Willebrand receptor of importance in platelet aggregation and thrombosis, and they are pro-inflammatory with mice primary macrophages. Both sulfobacins from gut bacteria act on Toll-like receptor 4 (TLR4) to block lipopolysaccharide binding, suppressing inflammation and macrophage M1 polarization in inflammatory bowel disease.
Similar lipids termed flavocristamides A and B were isolated from a marine bacterium Flavobacterium sp. and the Gram-negative sea-water bacterium Cyclobacterium marinus, and these have been shown to inhibit DNA polymerase α. Subsequently, a new sulfonolipid with some structural affinity to the capnoids was isolated from the halophilic bacteria Salinibacter ruber and Salisaeta longa, i.e., it is 2‑carboxy-2-amino-3,4-hydroxy-17-methyloctadec-5-ene-1-sulfonic acid for which the trivial name halocapnine is suggested. As its 3-O-acyl derivative (and not N-acyl), it represents about 10% of the total cellular lipids of the former.
Choanoflagellates are motile microbial eukaryotes that live in aquatic environments and feed on bacteria. They are believed to be the closest living unicellular relatives of animals, but on exposure to sulfonolipids related to the capnoids from Algoriphagus machipongonensis, the choanoflagellate Salpingoeca rosetta, a marine bacterium that serves as its prey, assembles multicellular 'rosettes' in a manner that may provide insights into how multicellularity evolved in animals. Two such lipids have been isolated and characterized and they have been termed 'Rosette-Inducing Factors' - RIF-1 (illustrated) and RIF-2. Both have capnoid bases attached to 2‑hydroxy,iso-methylbranched fatty acids, but RIF-2 differs from RIF-1 in that the first has a double bond and the second hydroxyl is in a different position of the capnoid component. S. rosetta is extraordinarily sensitive to RIF-1 and is induced to form rosettes at femtomolar (10‑15M) concentrations. Lysophosphatidylethanolamines synthesised also by the symbiotic bacteria elicit no response on their own but act synergistically with the RIFs to maximize the activity of the latter. Sulfobacins D and F (initially termed 'Inhibitor of Rosettes (IOR-1)') from the same bacteria are inhibitors of rosette assembly in a concentration-dependent manner; presumably, they compete for the same cellular target. The cysteinolides described above should be considered in the same context. It has been determined that there is an absolute requirement for the observed stereochemistry for all of these metabolites to work efficiently.
Recommended Reading
- Baronio, M., Lattanzio, V.M.T., Vaisman, N., Oren, A. and Corcelli, A. The acylhalocapnines of halophilic bacteria: structural details of unusual sulfonate sphingoids. J. Lipid Res., 51, 1878-1885 (2010); DOI.
- Bedke, D.K. and Vanderwal, C.D. Chlorosulfolipids: Structure, synthesis, and biological relevance. Nat. Prod. Rep., 28, 15-25 (2011); DOI.
- Bisseret, P., Ito, S., Tremblay, P.A., Volcani, B.E., Dessort, D. and Kates, M. Occurrence of phosphatidylsulfocholine, the sulfonium analog of phosphatidylcholine in some diatoms and algae. Biochim. Biophys. Acta, 796, 320-327 (1984); DOI.
- Corcelli, A., Lattanzio, V.M.T., Mascolo, G., Babudri, F., Oren, A. and Kates, M. Novel sulfonolipid in the extremely halophilic bacterium Salinibacter ruber. Appl. Env. Microbiol., 70, 6678-6685 (2004); DOI.
- Gong, Y., Wang, J., Li, F. and Zhu, B.D. Polysaccharides and glycolipids of Mycobacterium tuberculosis and their induced immune responses. Scand. J. Immun., 97, e13261 (2023); DOI.
- Grevengoed, T.J. and others. An abundant biliary metabolite derived from dietary omega-3 polyunsaturated fatty acids regulates triglycerides. J. Clin. Invest., 131, e143861 (2021); DOI.
- Haines, T.H. Anionic lipid headgroups as a proton-conducting pathway along the surface of membranes: A hypothesis. Proc. Natl Acad. Sci. USA, 80, 160-164 (1983); DOI.
- Kaya, K. Chemistry and biochemistry of taurolipids. Prog. Lipid Res., 31, 87-108 (1992); DOI.
- Leichnitz, D., Peng, C.C., Raguz, L., Rutaganira, F.U.N., Jautzus, T., Regestein, L., King, N. and Beemelmanns, C. Structural and functional analysis of bacterial sulfonosphingolipids and rosette-inducing factor 2 (RIF-2) by mass spectrometry-guided isolation and total synthesis. Chem. Eur. J., 28, e202103883 (2022); DOI.
- Li, Y.T., Maskos, K., Chou, C.W., Cole, R.B. and Li, S.C. Presence of an unusual GM2 derivative, taurine-conjugated GM2, in Tay-Sachs brain. J. Biol. Chem., 278, 35286-35291 (2003); DOI.
- Liu, Y. and others. Identification and characterization of the biosynthetic pathway of the sulfonolipid capnine. Biochemistry, 61, 2861-2869 (2022); DOI.
- Radka, C.D., Miller, D.J., Frank, M.W. and Rock, C.O. Biochemical characterization of the first step in sulfonolipid biosynthesis in Alistipes finegoldii. J. Biol. Chem., 298, 102195 (2022); DOI.
- Trammell, S.A.J and others. Identification of bile acid-CoA:amino acid N-acyltransferase as the hepatic N-acyl taurine synthase for polyunsaturated fatty acids. J. Lipid Res., 64, 100361 (2023); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: January 2026 | ||
© The LipidWeb is open access and fair use is encouraged - but not text and data mining, AI training, and similar technologies (text scraping).
