Glycosphingolipid Sulfates
Glycosphingolipid sulfates (sometimes termed "sulfatides" or "sulfoglycosphingolipids") are glycosphingolipids carrying a sulfate ester group attached to the carbohydrate moiety. They were first identified in brain tissue by the pioneering lipid chemist Thudichum in 1884, although it was much later (1962) before the structure of the main component was fully characterized. Although sulfoglycosphingolipids tend to be minor components of animal tissues, 3'-sulfo-galactosylceramide illustrated is one of the more abundant glycolipid constituents of myelin, but many more sulfated glycosphingolipids occur in nature. Terminology is sometimes employed of the form galactosylceramide-I3-sulfate or 'cerebroside sulfate' or the short-hand 'SM4' (by analogy with the GM nomenclature used with gangliosides).
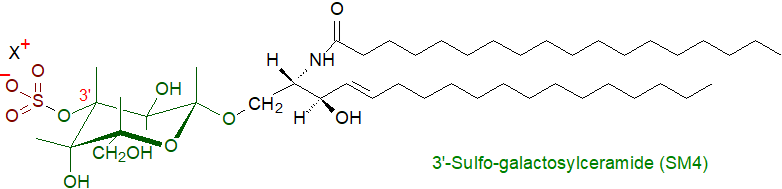
1. Occurrence and Structure
Glycosphingolipid sulfates are of particular importance in brain where galactosylceramide and its sulfatide comprise 23% and 4-6%, respectively, of the total lipid content of the myelin sheath, and in mouse brain, they are concentrated in the substantia nigra region. They are synthesised in oligodendrocytes in the central nervous system and in Schwann cells in the peripheral nervous system. However, they are vital constituents of many other organs, and especially the kidney, but also the gastrointestinal tract, islet of Langerhans, trachea and many cancer cell lines. 3'-Sulfo-galactosylceramide is present in the outer plasma membrane of most eukaryotic cells, including blood cells, such as erythrocytes, neutrophils and platelets, and it is a component of the plasma lipoproteins.
Many other sulfoglycosphingolipids have now been characterized but primarily from animal tissues (sea urchins to vertebrates). Sulfo-lactosylceramide or lactosylceramide-II3-sulfate (or 'SM3') is frequently found in tissues, and other sulfate esters derived from oligoglycosylceramides of the globo- and ganglio-series have been isolated from human kidney, where they show some structural kinship with the "brain-type" gangliosides. Such lipids with one to four hexose units and usually one but occasionally two sulfate groups have been isolated from the kidneys of rats and mice, while analogous lipids containing from one up to five hexose units and one sulfate were detected in a human renal carcinoma. There are substantial regional differences in the content and composition of the sulfatides in kidney, with particularly high concentrations in the distal nephron segments and the renal medulla that presumably reflect the functional requirements of these tissues. Sulfated forms of galactosylceramide, lactosylceramide and globopentaosylceramide are present on the surface membrane of undifferentiated human pluripotent stem cells. The bis-sulfo-gangliotetraosylceramide (SB1a) or gangliotetraosylceramide-II3,IV3-bis-sulfate from mouse kidney is illustrated.

As of a 2009 review, 24 such lipids with variations in the carbohydrate chain had been characterized in vertebrates alone. As further examples, two oligoglycosphingolipids with terminal glucuronic acid residues having sulfate ester moieties in the 3'-position occur in the peripheral nervous system, and other complex oligoglycosphingolipids, including gangliosides, with sulfate groups have been isolated from human, mouse and monkey kidney cells. With kidney cells from the African green monkey, nine distinct sulfated glycolipids were characterized. In most if not all of these, the sulfate ester moiety is attached to the C3 hydroxyl group of galactose and has an equatorial conformation. 3’‑Sulfo-glucosylceramide is only encountered at trace levels in tissues, although glucosylceramide-I6 sulfate has been isolated from the Ascidian Ciona intestinalis.
 The fatty acid
components of sulfolipids in animals vary with the nature of each lipid and the tissue.
In myelin from the central nervous system, 24:0, 24:1 and 2‑hydroxy saturated fatty acids predominate in the sulfo-galactosylceramide,
and hydroxy fatty acids occur in high concentrations in grey matter but not in the white.
During myelination and development of the brain in rats, short-chain sulfatides with C16 non-hydroxylated fatty acids and
C18 non-hydroxy and hydroxy fatty acids are synthesised first in restricted regions of the early embryonic spinal cord,
while C22 hydroxy fatty acids accumulate later in oligodendrocyte development;
C24 fatty acids are most abundant in adulthood.
The corresponding lipid from peripheral tissues such as the pancreas often contains a high proportion of 16:0 and 18:0 fatty acids,
and this composition is present likewise in some of the sulfo-oligoglycolipids of brain such as the cortical grey matter.
On the other hand, 22:0 together with 23:0 and 24:0 are the main fatty acids of kidney sulfolipids,
although there are appreciable variations in the long-chain base compositions within structures
in this organ that are presumably related in some manner to their functions.
Thus, the renal papillae contain sulfatides with C20 bases, whereas conventional C18-sphingosine-containing compounds
predominate in the medulla, and sulfatides with phytosphingosine (t18:0/C22:0-2OH and t18:0/C24:0-2OH) are present in
intercalated cells.
The fatty acid
components of sulfolipids in animals vary with the nature of each lipid and the tissue.
In myelin from the central nervous system, 24:0, 24:1 and 2‑hydroxy saturated fatty acids predominate in the sulfo-galactosylceramide,
and hydroxy fatty acids occur in high concentrations in grey matter but not in the white.
During myelination and development of the brain in rats, short-chain sulfatides with C16 non-hydroxylated fatty acids and
C18 non-hydroxy and hydroxy fatty acids are synthesised first in restricted regions of the early embryonic spinal cord,
while C22 hydroxy fatty acids accumulate later in oligodendrocyte development;
C24 fatty acids are most abundant in adulthood.
The corresponding lipid from peripheral tissues such as the pancreas often contains a high proportion of 16:0 and 18:0 fatty acids,
and this composition is present likewise in some of the sulfo-oligoglycolipids of brain such as the cortical grey matter.
On the other hand, 22:0 together with 23:0 and 24:0 are the main fatty acids of kidney sulfolipids,
although there are appreciable variations in the long-chain base compositions within structures
in this organ that are presumably related in some manner to their functions.
Thus, the renal papillae contain sulfatides with C20 bases, whereas conventional C18-sphingosine-containing compounds
predominate in the medulla, and sulfatides with phytosphingosine (t18:0/C22:0-2OH and t18:0/C24:0-2OH) are present in
intercalated cells.
Sponges have been found to contain many novel lipid compounds, and the freshwater sponge Ephydatia syriaca contains a strange, sulfated ceramide glycoside, termed ‘syriacin’, in which a fucose residue is linked to ceramide via a sulfate bridge, together with a novel fatty acid linked to the ceramide, i.e., (all Z)‑34S‑methylhexatriaconta-5,9,12,15,18,21-hexaenoic acid. Similarly, ceramides linked directly to sulfate have been isolated from marine Zoanthids ('palyosulfonoceramides'). Oligoglycosphingolipid sulphates containing glucuronic acid as one of the carbohydrate units are present in several species of marine invertebrates and in the insect genus Arthropoda. Sphingolipid sulfates do not appear to have been detected in plants, but non-animal exceptions are the parasitic protozoa Plasmodium falciparum, and the trypanosomatid parasite, Trypanosoma cruzi, from which a sulfated dihexosylceramide has been characterized.
Other complex glycolipid sulfates: Many parallels can be drawn between the biosynthesis, metabolism and function of seminolipid and those of sphingolipid sulfates. There are separate web pages dealing with other sulfur-containing lipids, including the plant sulfonolipid, sulfoquinovosyldiacylglycerol, the microbial sulfo- and sulfonolipids, e.g., the trehalose containing glycolipids from Mycobacterium tuberculosis, and some gangliosides.
2. Biochemistry and Function
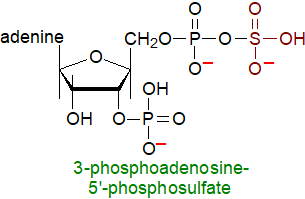 The galactosylceramide precursor
of sulfatide is synthesised in the endoplasmic reticulum and is transported to the Golgi lumen where sulfation of galactosylceramide is catalysed
by the enzyme glycosylceramide sulfotransferase with 3'‑phosphoadenosine-5'-phosphosulfate as the activated sulfate donor.
Lactosylceramide and oligoglycosphingolipids are sulfated by the same enzyme,
which prefers a β-glycoside and mainly a β-galactoside at the non-reducing termini of sugar chains attached to ceramide and
was first isolated and then cloned from a cell line derived from human renal carcinoma, where it is especially active.
The same enzyme and sulfate donor is utilized for the biosynthesis of the glycoglycerolipid
seminolipid.
A second sulfotransferase from rat brain catalyses the transfer of sulfate to glucuronylglycolipids.
Within the cell, sulfatides are redistributed by means of a glycolipid transfer protein (GLTP), which transfers glycolipids
from the cytosolic leaflet of the endoplasmic reticulum or plasma membrane and acts as a sensor of glycolipid levels.
Peroxisome proliferators-activated receptor alpha (PPARα) is a regulator of sulfatide biosynthesis.
The galactosylceramide precursor
of sulfatide is synthesised in the endoplasmic reticulum and is transported to the Golgi lumen where sulfation of galactosylceramide is catalysed
by the enzyme glycosylceramide sulfotransferase with 3'‑phosphoadenosine-5'-phosphosulfate as the activated sulfate donor.
Lactosylceramide and oligoglycosphingolipids are sulfated by the same enzyme,
which prefers a β-glycoside and mainly a β-galactoside at the non-reducing termini of sugar chains attached to ceramide and
was first isolated and then cloned from a cell line derived from human renal carcinoma, where it is especially active.
The same enzyme and sulfate donor is utilized for the biosynthesis of the glycoglycerolipid
seminolipid.
A second sulfotransferase from rat brain catalyses the transfer of sulfate to glucuronylglycolipids.
Within the cell, sulfatides are redistributed by means of a glycolipid transfer protein (GLTP), which transfers glycolipids
from the cytosolic leaflet of the endoplasmic reticulum or plasma membrane and acts as a sensor of glycolipid levels.
Peroxisome proliferators-activated receptor alpha (PPARα) is a regulator of sulfatide biosynthesis.
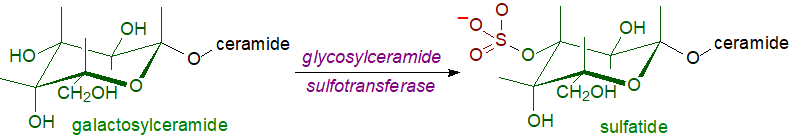 |
| Figure 1. Biosynthesis of glycosphingolipid sulfates. |
Galactosylceramide-I3-sulfate is located exclusively on the outer leaflet of the myelin sheath, which is a lipid-rich membrane produced as an extension of the plasma membrane and consists of multilamellar and spirally wrapped sheaths around neuronal axons; it is an integral component of the axo-glial junction. Simplistically, the lipid coating acts as an insulator, although each of the various lipid constituents may be required for other purposes. The period when sulfatide synthesis is most rapid in brain coincides with myelin formation during foetal development in animal models, and there is considerable evidence pointing to a direct role in this process. During development, there is a rapid increase in the relative concentration of molecular species with C24 as opposed to C18 fatty acid constituents. Experiments with genetically modified animals and targeted enzyme deletion have confirmed that sphingolipid sulfates are indispensable for myelin development and function and for its maintenance. Mice in which the galactosylceramide sulfotransferase in the brain was eliminated in this manner appeared normal at birth, but soon developed neurological disorders. In addition, there is evidence that sulfatide is a key regulator of the terminal differentiation of oligodendrocytes.
The physical properties of sulfatides are relevant here, as sulfatides interact with galactosylceramides and gangliosides in myelin via the carbohydrate moieties to maintain a compact myelin structure and axo-glial integrity. They may take part in the lateral organization of myelin membranes to influence sorting, lateral assembly and dynamics, as well as upon the operation of proteins in different substructures of the myelin sheath.
 Like the gangliosides,
sulfoglycosphingolipids are acidic and relatively soluble in aqueous systems, properties that must have a bearing on their functions
in tissues, especially in ion transport.
Indeed, stable layers of water up to 44 Å thick can form around the polar head group.
The free hydroxyl groups in the fatty acid and sphingoid base constituents greatly strengthen hydrogen bonding
in surface membranes, where sulfoglycosphingolipids may be necessary as amphiphilic donors of negative charges.
There is evidence that they are concentrated in the microdomains known as rafts in the plasma membrane,
which serve as signalling platforms.
In the salt glands of ducks and organs associated with osmoregulation in fish, sulfatides have been shown to be required for the transport
of sodium and potassium ions.
There are strong indications that they may have a similar role in kidney as targeted gene deletion has demonstrated that sulfoglycosphingolipids
are critical for renal ammonium handling, urinary acidification and acid-base homeostasis, i.e., to maintain a stable blood pH.
A high content of sulfatides in the gastric and duodenal mucosa, where membranes can be attacked by acid, pepsin and bile salts,
may be a factor in mucosal protection.
Like the gangliosides,
sulfoglycosphingolipids are acidic and relatively soluble in aqueous systems, properties that must have a bearing on their functions
in tissues, especially in ion transport.
Indeed, stable layers of water up to 44 Å thick can form around the polar head group.
The free hydroxyl groups in the fatty acid and sphingoid base constituents greatly strengthen hydrogen bonding
in surface membranes, where sulfoglycosphingolipids may be necessary as amphiphilic donors of negative charges.
There is evidence that they are concentrated in the microdomains known as rafts in the plasma membrane,
which serve as signalling platforms.
In the salt glands of ducks and organs associated with osmoregulation in fish, sulfatides have been shown to be required for the transport
of sodium and potassium ions.
There are strong indications that they may have a similar role in kidney as targeted gene deletion has demonstrated that sulfoglycosphingolipids
are critical for renal ammonium handling, urinary acidification and acid-base homeostasis, i.e., to maintain a stable blood pH.
A high content of sulfatides in the gastric and duodenal mucosa, where membranes can be attacked by acid, pepsin and bile salts,
may be a factor in mucosal protection.
Sulfatides participate in many different cellular processes throughout the body including trafficking of proteins, cell adhesion and aggregation, immune responses and signal transmission. Many of these effects are a consequence of binding to characteristic proteins, which usually have a hydrophobic cavity that interacts with the ceramide component with the hydrophilic moiety exposed for further intermolecular associations. Among many such interactions, sulfatides induce the coagulation factors FXII and FXI to initiate the endogenous coagulation pathway, and they are involved in platelet aggregation by binding to selectins as stable aggregates.
They can act as antigens in the immune system by binding to clusters of differentiation 1 (CD1) molecules as complexes recognized by dendritic cells, macrophages and natural killer T (NKT) cells; long-chain sulfatides (24:0 and 24:1) are most efficient. Sulfatide-reactive type II NKT cells regulate type I NKT cells by modulating the operation of antigen-presenting cells to protect from autoimmune disorders. They are ligands for Toll-like receptor 4 (TLR4), an innate immune receptor that initiates inflammation when stimulated by the bacterial lipopolysaccharide and endotoxin (lipid A). Depending on the particular tissue and pathological context, they can be pro- or antioxidants.
In the pancreas, sulfatide is located in the same cellular compartment as insulin, and it takes part in insulin processing and secretion through activation of ion channels. It promotes folding of proinsulin and may serve as a molecular chaperone for insulin, where molecular species containing palmitic acid are important. Antibodies to sulfatide are often present in serum before the onset of diabetes, and sulfatide may influence the progression of the disease.
Catabolism: The principles of lysosomal degradation of sphingolipids are outlined in our web page dealing with mono-glycosylceramides. In the catabolism of cerebroside sulfate within lysosomes, there is an initial hydrolysis of the sulfate bond by an arylsulfatase (ARSA), which must be activated by an enzyme in the endoplasmic reticulum that catalyses the conversion of cysteine to formylglycine at the catalytic site. Formation of the product galactosylceramide requires a non-enzymic protein, known as ‘saposin B’ (or as the ‘cerebroside sulfate activator’ or ‘sphingolipid activator protein-1’), which is one of a group of four cysteine-rich proteins with a common ability to interact with membranes. Saposin B acts by binding to the lipid, extracting it from the membrane and presenting it to the hydrolase in a manner that facilitates reaction. Structural studies have revealed that the molecule is a dimer with a large hydrophobic cavity into which the lipid fits and is presumably orientated so attack by the hydrolase is facilitated. Finally, the galactose unit is removed by a β‑galactosidase.
3. Sulfatides and Disease
Aberrant sulfoglycosphingolipid metabolism has been associated with various pathogenic conditions, including cancer, autoimmune diseases and sphingolipid storage disorders. Sulfoglycosphingolipids accumulate as a result of elevated galactosylceramide sulfotransferase activity in a number of human cancers, including renal cell carcinoma, adenocarcinoma of colon and lung, and ovarian cancer, where they promote metastasis. They are known to be a crucial factor in the development of cardiovascular disease, and the levels in plasma can be indicators of vascular injury, atherosclerosis and even myocardial infarction. In contrast, reduced levels of brain sulfatides have been found at the earliest stages of Alzheimer’s disease, possibly due to an impaired sulfatide transport mechanism mediated by apolipoprotein E. Substantial changes in brain sulfatide levels have been noted in other neurodegenerative diseases, including Parkinson's and Lewy body diseases, and the Guillain-Barré syndrome, and reduced formation of sulfatides has been found during chronic alcohol consumption and polycystic kidney disease. With Parkinson's disease, lipidomic studies have revealed highly specific distributions of sulfatide molecular species based on the chain-length, number of double bonds, and importantly the presence of 2‑hydroxyl groups in the fatty acid components in different regions of the brain (primate model). Loss of sulfatides in the liver is associated with a chronic inflammatory disorder known as primary sclerosing cholangitis in which bile ducts deficient in sulfatides have increased permeability, resulting in the spread of bile into the liver parenchyma.
Sulfatide has been implicated in infection by viruses, including the human immunodeficiency virus (HIV-1), noroviruses and the hepatitis C and influenza A viruses, by facilitating entry into cells. With the influenza A virus, sulfatide is recognized by the glycoprotein hemagglutinin of the viral envelope and binds to it on the surface membrane of infected cells to induce apoptosis and enable the virus to interact with the nucleus to facilitate virus replication. Noroviruses bind to the 3-O-sulfated galactose moiety of sulfatides via certain of their capsid proteins. Similarly, many types of pathogenic bacteria or bacterial protein toxins bind to sulfatide at the mucosal surface with effects upon the development of disease. In contrast, sulfoglycolipids have been associated with protection against Chagas disease and the vaccinia virus.
Lysosulfatide (sulfogalactosylsphingosine), i.e., without the fatty acid constituent, as well as the acylated state accumulates in the brain of patients with the metabolic disease - metachromatic leukodystrophy. The N-acyl-moiety is hydrolysed the by the fatty acid amide hydrolase (FAAH), first characterized for the catabolism of anandamide, but surprisingly experimental deletion of the enzyme in mice exacerbates the disease, suggesting that it may be normally be protective. The main cause of the syndrome is that the enzyme arylsulfatase A (or saposin B) is lacking in lysosomes, leading to fatal de-myelination of both central and peripheral nervous systems (large amounts of sulfatides may then be present in the cerebrospinal fluid and urine). Lysosulfatide is cytotoxic in vitro but occurs naturally at low levels in tissues such as brain, where it serves in signalling in a manner that may oppose the influence of sphingosine 1-phosphate and sphingosine phosphocholine. It inhibits protein kinase C and cytochrome c oxidase, and it retards the migration of neuronal precursor cells.
4. Analysis
Sulfoglycolipids tend not to be quite as water-soluble as the gangliosides, but they resemble them in some of their physical properties, and comparable methods are used for analysis. There is a report that they can be isolated from complex lipid samples by binding to titanium dioxide. One advantageous analytical strategy is to remove the sulfate ester moiety to reduce the polarity, so that the methodology devised for neutral glycosphingolipids can be employed. Modern mass spectrometric methods are now being used increasingly for characterization of intact sulfatides.
Recommended Reading
- Blomqvist, M., Zetterberg, H., Blennow, K. and Mansson, J.E. Sulfatide in health and disease. The evaluation of sulfatide in cerebrospinal fluid as a possible biomarker for neurodegeneration. Mol. Cell. Neurosci., 116, 103670 (2021); DOI.
- Chen, L., Elizalde, M. and Alvarez-Sola, G. The role of sulfatides in liver health and disease. Front. Biosci.-Landmark, 30, 25077 (2025); DOI.
- Christie, W.W. and Han, X. Lipid Analysis - Isolation, Separation, Identification and Lipidomic Analysis (4th edition), 446 pages (Oily Press, Woodhead Publishing and now Elsevier) (2010) - see Science Direct.
- Grassi, S., Prioni, S., Cabitta, L., Aureli, M., Sonnino, S. and Prinetti, A. The role of 3-O-sulfogalactosylceramide, sulfatide, in the lateral organization of myelin membrane. Neurochem. Res., 41, 130-143 (2016); DOI.
- Honke, K. Biosynthesis and biological function of sulfoglycolipids. Proc. Jpn Acad. Ser. B, Phys. Biol. Sci., 89, 129-138 (2013); DOI.
- Ishizuka, I. Chemistry and functional distribution of sulfoglycolipids. Prog. Lipid Res., 36, 245-319 (1997); DOI.
- Kaya, I. and others.Spatial lipidomics reveals brain region-specific changes of sulfatides in an experimental MPTP Parkinson’s disease primate model. npj Parkinsons Dis., 9, 118 (2023); DOI.
- Li, T.T., Li, G., Hu, R., Ji, X., Liu, C. and Shao, Y.T. Relationship of sulfatides physiological function and peroxisome proliferator-activated receptor α. Neurochem. Res., 48, 2059-2065 (2023); DOI.
- Marsching, C. and others. Renal sulfatides: sphingoid base-dependent localization and region-specific compensation of CerS2-dysfunction J. Lipid Res., 55, 2354-2369 (2014); DOI.
- Merrill, A.H. Sphingolipid and glycosphingolipid metabolic pathways in the era of sphingolipidomics. Chem. Rev., 111, 6387-6422 (2011); DOI.
- Rožman, M., Fabris, D., Mrla, T. and Vukelic, Ž. Database and data analysis application for structural characterization of gangliosides and sulfated glycosphingolipids by negative ion mass spectrometry. Carbohydr. Res., 400, 1-8 (2014); DOI.
- Takahashi, T. and Suzuki, T. Role of sulfatide in normal and pathological cells and tissues. J. Lipid Res., 53, 1437-1450 (2012); DOI.
- Takahashi, T. and Suzuki, T. Role of sulfatide in influenza A virus replication. Biol. Pharm. Bull., 38, 809-816 (2015); DOI.
- Xiao, S., Finkielstein, C.V. and Capelluto, D.G.S. The enigmatic role of sulfatides: new insights into cellular functions and mechanisms of protein recognition. In: Lipid-Mediated Protein Signaling (Adv. Exp. Med. Biol., Vol. 991), pp. 27-40 (edited by D.G.S. Capelluto, Springer Science+Business Media, Dordrecht) (2013); DOI.
- Yu, R.K., Yanagisawa, M. and Ariga, T. Glycosphingolipid structures. In: Comprehensive Glycoscience. Vol. 1, pp. 73-122 (ed. J.P. Kamerling, Elsevier, Oxford, UK) (2007); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: January 2026 | ||
© The LipidWeb is open access and fair use is encouraged - but not text and data mining, AI training, and similar technologies (text scraping).
