Proteolipids and Protein Lipidation
In 1951, proteins that were soluble in organic solvents such as chloroform-methanol were found in rat brain myelin by Folch and Lees (perhaps better known for devising the most common method for lipid extraction), who coined the term ‘proteolipid’, but it was a further twenty years before it was shown that these contained covalently bound fatty acids and so differed from the plasma lipoproteins. Such lipid-modified proteins, formed in post-translational reactions, are now known to be widespread in nature with many vital functions in animals, plants and bacteria. Proteolipids can be defined as all proteins containing covalently bound lipid moieties such as fatty acids, isoprenoids, cholesterol and glycosylphosphatidylinositols (the last have their own web page). Protein lipidation provides an interface between the lipophilic membranes of cells and otherwise hydrophilic proteins that enables signalling and many other metabolic events at the membrane surface; dysregulation impacts upon physiology and disease. Here, proteolipids from animals and plants are discussed, but bacterial lipopeptides and proteolipids are sufficiently different to have their own web page. The term ‘lipoprotein’ is sometimes used to describe such compounds, but to avoid confusion this might be better reserved for the non-covalently linked lipid-protein complexes of the type found in plasma.
1. Introducing Protein-Lipid Modifications
It is a curious but noteworthy fact that the two main types of protein with long-chain fatty acid modifications to have been described from eukaryotic organisms contain saturated fatty acid components, i.e., those with only myristoyl and those with predominantly palmitoyl moieties, each with a distinctive type of linkage, amide or thiol ester, respectively, although a few O‑acylated proteins are known. These modifications can be differentiated by the main amino acid residues (glycine, cysteine or serine) that are involved. Prenylated lipids contain an isoprenoid group linked via a sulfur atom (thiol ether bond) to the protein. N‑Myristoylated and prenylated proteins often have one or more additional S-acylation residue. Covalent modification of proteins with lipids changes them from a generally hydrophilic nature to one that is hydrophobic at one end at least, thus facilitating the interaction with membranes.
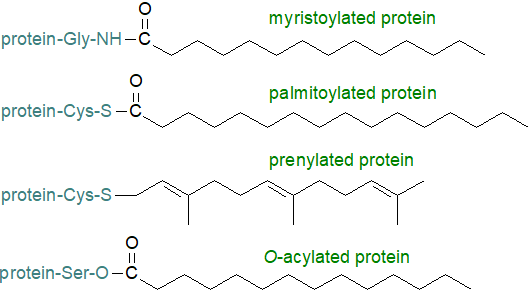 |
| Figure 1. The main types of proteolipids. |
Protein S-palmitoylation occurs post-translationally at the membrane surface, while N‑myristoylation takes place at the ribosomal level, and S-prenylation is brought about by cytoplasmic enzymes. It is now clear that such modifications can determine the properties of proteins and target them to specific subcellular membrane domains, including the rafts and caveolae in plasma membranes. Thus, both myristoylated and palmitoylated proteins are targeted to rafts (as are the GPI-anchored proteins), but prenylated lipids are not. It is significant that many signalling proteins, e.g., G-protein-coupled receptors and protein tyrosine kinases, and often their substrates are modified by lipids with implications for the relevant signalling events at the cell surface. Of these, S-palmitoylation is readily reversible so has drawn comparisons with such metabolic modifications as phosphorylation and ubiquitination.
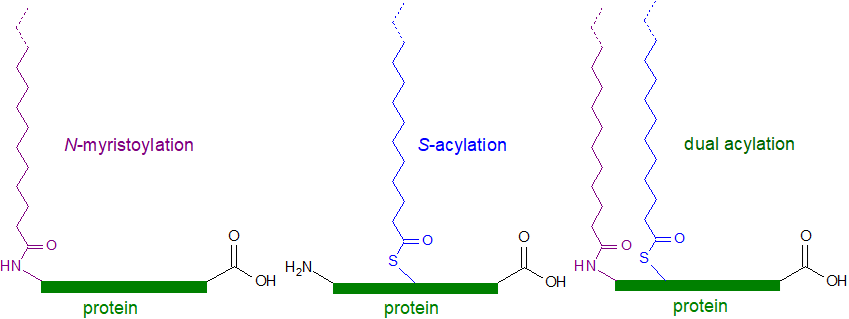 |
| Figure 2. Protein lipid acylation. |
Other protein-lipid links exist and are discussed below, but they tend to be less common. One class of proteolipids contains a linkage to cholesterol as well as N‑palmitoylation, the ‘hedgehog’ signalling proteins, while bacterial proteolipids contain N-acyl- and S‑diacylglycerol groups attached to an N‑terminal cysteine.
Such proteolipids are critical for all classes of eukaryotes (animals, plants and fungi) and bacteria, and they many influence human disease states so are potential pharmacological targets. Deregulation of palmitoylation has been associated with heart disease, cancer, mental retardation and schizophrenia, while some pathogenic organisms can hijack the protein acylation mechanism to increase the susceptibility of the host to infection.
2. N-Myristoylated Proteins
In most N-myristoylated proteins, myristic acid (14:0), a ubiquitous but usually minor component of cellular lipids (only 1% of the total fatty acids), is bound to the amino-terminal glycine (rarely cysteine) residue of a relatively conserved sequence of the protein via an amide linkage that is comparatively stable to hydrolysis. Therefore, for most proteins, N-myristoylation in this way is an irreversible stable modification in essence, and the half-life of a myristoylated protein is like that of the nascent polypeptide chain. These proteolipids constitute a large family of eukaryotic proteins (~2% of the total) with many different functions, and they are located either in the cytosol or in the cytosolic (inner) membrane of cells, or both. By means of N-glycine myristoylation, the modified proteins can be targeted to various membranes in the cell, including the plasma membrane, endoplasmic reticulum, Golgi, mitochondria and nuclei. The acyl group anchors the protein to membranes, although simultaneous binding to phospholipids or other membrane constituents increases the strength of the interaction. As a single N‑myristoylation is not sufficient to ensure membrane association, a second binding site is usually necessary, e.g., hydrophobic residues, another membrane-bound binding partner or further N‑myristoylation or S‑palmitoylation (see below).
During biosynthesis, the leader methionine residue of the nascent peptide chain is first removed by a methionine aminopeptidase (MAP) to expose the N‑terminal glycine before an N‑myristoyl-CoA:protein N-myristoyltransferase (NMT1 or NMT2 in humans) catalyses the formation of the stable amide bond as it begins to emerge from the ribosome, i.e., it is mainly a co-translational rather than a post-translational event. The mechanism involves an exchange of NMT1 with MAP at the ribosomal tunnel exit to form the co-translational complex together with the nascent polypeptide-associated complex (NAC). As NMT1 binding is sequence selective and is triggered by methionine excision to expose the N-myristoylation motif, the interactions of the three proteins explain how a specific subset of proteins can be efficiently N-myristoylated.
 |
| Figure 3. N-Myristoylation of proteins. |
In brain at least, myristic acid is obtained mainly from membrane phospholipids by the action of the phospholipase A1 isoform DDHD2. Myristoyl-CoA is supplied by an acyl-CoA binding protein (ACBD6) and this, together with the size of the NMT binding pocket controls the specificity of the reaction by forming a complex with the transferase thereby stimulating it and preventing it from utilizing the competitor palmitoyl-CoA. Prokaryotes do not have such enzymes, but most eukaryotes do, although lower eukaryotes, e.g., the yeast Saccharomyces cerevisiae, have only one isoform.
While NMT1, but not NMT2, is utilized for cell proliferation, both are required for cell survival. NMT1 is located mainly in the plasma membrane while NMT2 is cytosolic (nearly 77% peptide sequence identity), and both have an N‑terminal region with polybasic amino acid sequences, which target proteins to the ribosomes to facilitate the co-translational timing of the reaction. They have partial substrate overlap but differential roles during embryogenesis and tumour cell proliferation. The crystal structure of NMT1 has shown that it has a characteristic binding cleft where potential substrates for myristoylation are recognized (with some overlap with targets for N-acetylation), and there is an absolute requirement for the N-terminal glycine on the target protein with amino acid residues two through eight as the consensus motif (Met-Glc-X-X-X-Ser/Thr/Cys) used in substrate recognition.
Exceptions to these generalities are photoreceptor proteins, which are modified heterogeneously with the uncommon 12:0, 5‑14:1 and 5,8‑14:2 fatty acids as well as 14:0, and certain histone proteins can be modified by various fatty acids. In the model nematode Caenorhabditis elegans, N-terminal glycine is acylated almost exclusively with myristic acid, but lysine (see below) is acylated preferentially with isomethyl-branched-chain fatty acids. N‑Palmitoylated proteins have been found on occasion, usually where there is a dual lipid modification as with the cholesterol-linked hedgehog proteins (see below). As a further exception, it has become apparent that myristoylation can occur post-translationally in apoptotic cells on internal glycine residues exposed by caspase cleavage in partially hydrolysed proteins, a process that may have implications for health and disease.
N-myristoylated proteins with dual modifications can be dissociated from membranes by de‑S‑palmitoylation, by phosphorylation, by binding of the hydrophobic moieties to competing cytosolic proteins (if the myristoyl moiety is relatively exposed) or by conformational changes, and these reactions are forms of regulation. The low levels of myristic acid in tissues may be a further controlling factor. Macrophages contain at least one protease that targets certain N‑myristoylated proteins, and similar proteases must operate in other tissues. On the other hand, myristoylation promotes protein stability by inhibiting the proteasome degradation pathway.
 In humans,
569 proteins are now known to be myristoylated, and these include proteins that have roles in signalling networks, apoptosis,
oncogenesis and viral replication, all of critical physiological importance.
The endoplasmic reticulum-Golgi network and the plasma membrane are the main targets of myristoylated proteins, but mitochondria, cilia,
nuclei and the endolysosomal network are targeted in the same way.
Their lipid moieties influence protein efficiency perhaps by modifying or stabilizing their conformations,
by facilitating protein-protein interactions, and by targeting otherwise soluble proteins to the membranes and to appropriate receptors.
N‑Myristoylation is essential for erythrocyte formation and myelination, it is necessary for calcium release in mitochondria,
and for T cells and the immune response.
During apoptosis, NMTs are substrates for caspases, which regulate their location from the ribosomal and membrane
regions to the cytosol and vice versa.
In humans,
569 proteins are now known to be myristoylated, and these include proteins that have roles in signalling networks, apoptosis,
oncogenesis and viral replication, all of critical physiological importance.
The endoplasmic reticulum-Golgi network and the plasma membrane are the main targets of myristoylated proteins, but mitochondria, cilia,
nuclei and the endolysosomal network are targeted in the same way.
Their lipid moieties influence protein efficiency perhaps by modifying or stabilizing their conformations,
by facilitating protein-protein interactions, and by targeting otherwise soluble proteins to the membranes and to appropriate receptors.
N‑Myristoylation is essential for erythrocyte formation and myelination, it is necessary for calcium release in mitochondria,
and for T cells and the immune response.
During apoptosis, NMTs are substrates for caspases, which regulate their location from the ribosomal and membrane
regions to the cytosol and vice versa.
Increased levels of N-myristoylation have been observed in certain cancers, and there have been suggestions that NMTs could be a target for therapeutic intervention. NMT plays a part in innate immunity, the evolutionarily conserved host defence mechanism that responds immediately to pathogens but does not provide long-lasting immunity to the host (in contrast to adaptive immunity). It is relevant to infections by bacteria, e.g., Shigella flexneri, viruses, including human immunodeficiency virus-1 (HIV-1), fungi and parasitic protozoa, including such diseases as malaria. The Shigella virulence factor IpaJ has been identified as an irreversible demyristoylase, which can hydrolyse the myristoyl group from the N-terminal glycine of human ARF1.
Plants: 650 Arabidopsis thaliana proteins are potentially myristoylated, including many protein kinases, phosphatases, thioredoxins and transcription factors. As an example, the calcium-sensing molecule CBL1 uses N-myristoylation for association with the endoplasmic reticulum followed by S‑acylation to promote trafficking to the plasma membrane; in contrast CBL2 is not N-myristoylated but rather is triply S-acylated to direct it to the tonoplast.
Lysine fatty acylation: It is now recognized that a second form of protein N-acylation exists in which lysine residues are acylated (mainly with 14:0) as a post-translational event, again to increase the affinity of proteins for cellular membranes, although so far, a relatively limited number of proteins are known to be modified in this way that include tumour necrosis factor α (TNF-α), interleukin 1-α and some of the Ras family of GTPases. With these, different enzymes or iso-enzymes from those for N‑terminal myristoylation are involved, and the reaction occurs by a less efficient mechanism termed K-myristoylation. In this instance, the modification is reversible as sirtuins 2 and 6 (SIRT2/6), mammalian nicotinamide adenine dinucleotide (NAD)-dependent lysine deacylases, catalyse the removal of fatty acyl groups from lysine residues, and they are thus able to regulate the relevant proteins. Demyristoylation at two sites of gravin-α in adipocytes by the enzyme histone deacetylase 11 (HDAC11) prevents β-adrenergic (G protein-coupled) receptors from translocating to the membrane microdomains that are utilized for downstream protective signalling, in effect a control mechanism with relevance to obesity. In brain, lysine-myristoylation/demyristoylation may underpin synaptic plasticity and learning. Among reported ill effects of lysine fatty acylation are tumorigenesis and increased pathogenesis of bacteria. In plants, N‑acylation of lysine residues with various phytohormones, including jasmonic acid, has been reported. N-acetylation of lysine residues is discussed below.
 Lipoic acid (5R-(1,2-dithiolan-3-yl)pentanoic acid) is a cofactor
for five enzymes or classes of enzymes in mitochondria, all of which are concerned with energy metabolic complexes.
It is synthesised from octanoic acid and transferred as a thioester of acyl carrier protein to a terminal lysine residue of the
lipoyl domains of the mitochondrial proteins where it is linked by an amide bond.
This post-translational modification provides a “swinging arm”, which is able to move between different subunits within the enzyme complex to
facilitate substrate channelling and electron transfer during oxidation-reduction reactions, including hydrogen transfers, decarboxylation
and movement of other acyl groups.
In contrast, the sulfur unit of lipoic acid has a high affinity for copper ions, which can result in cell death through cuproptosis with
implications for many disease states.
Some sirtuin family members, including SIRT4 and SIRT2, can delipoylate these proteolipids, but most lipoic acid is released when the enzymes
are eventually degraded.
Lipoic acid (5R-(1,2-dithiolan-3-yl)pentanoic acid) is a cofactor
for five enzymes or classes of enzymes in mitochondria, all of which are concerned with energy metabolic complexes.
It is synthesised from octanoic acid and transferred as a thioester of acyl carrier protein to a terminal lysine residue of the
lipoyl domains of the mitochondrial proteins where it is linked by an amide bond.
This post-translational modification provides a “swinging arm”, which is able to move between different subunits within the enzyme complex to
facilitate substrate channelling and electron transfer during oxidation-reduction reactions, including hydrogen transfers, decarboxylation
and movement of other acyl groups.
In contrast, the sulfur unit of lipoic acid has a high affinity for copper ions, which can result in cell death through cuproptosis with
implications for many disease states.
Some sirtuin family members, including SIRT4 and SIRT2, can delipoylate these proteolipids, but most lipoic acid is released when the enzymes
are eventually degraded.
Analysis: Mass spectrometry is now widely used for characterization of N-myristoylated proteins, although the fatty acyl group can be released for analysis by conventional chromatographic means (e.g., gas chromatography) by the acidic hydrolysis conditions commonly employed to cleave peptide bonds. Treatment with 6M HCl or 2M HCl in 83% methanol at 100°C for several hours is needed to release the N-acyl group as the free fatty acid or methyl ester, respectively. New methods with chemical probes and mass spectrometry are now facilitating detection and analysis of N‑myristoylated proteins and other proteolipids.
3. S-Acylated (Palmitoylated) Proteins
In the S-acylated proteins, long-chain fatty acids are linked to one or more (up to six) internal cysteine residues via labile high-energy thioester bonds. As palmitic acid (16:0) is the most common acyl component, these are often termed S-palmitoylated proteins, but this is something of a misnomer as other fatty acids are often present, including 16:1, 18:0 and 18:1 (one screen found 218 proteins acylated with 16:0 and 308 proteins with C18 fatty acids in humans), while S-18:0 is as common as S-16:0 in plants, and viral proteins are often linked to C18 fatty acids; in the nematode C. elegans, 15‑methylhexadecanoic acid is preferred. This may reflect the relative availabilities of these fatty acids at each acylation site, enzyme specificities or specific features of the modified substrate protein. There is some evidence that the nature of the acylating fatty acid can affect the efficiency of the resulting proteolipid and might contribute to the development and progression of disease, so the term 'S-acylation' is more accurate. In contrast to N-myristoylation and isoprenylation, there is no defined peptide target sequence, although proteomic methods now enable prediction of which cysteine residue is likely to be palmitoylated. It is now apparent that protein palmitoylation is essential for intracellular signalling and for the folding, trafficking and function of such disparate proteins as Src-family kinases, Ras family GTPases, G proteins and G protein-coupled receptors. As S-acylation is reversible by deacylation, it is an on/off switch, analogous to phosphorylation, acetylation and ubiquitylation.
The archetypal proteolipid protein (‘PLP’) or lipophilin is the main protein in the myelin of the central nervous system and was the first of its type to be identified and properly characterized. It is a highly conserved hydrophobic protein of 276 to 280 amino acids with four transmembrane segments that binds at least six palmitoyl groups via thioester bonds. A wide variety of different palmitoylated proteins are now known, and this is most prevalent type of protein modification by lipids with more than 500 different palmitoylated proteins identified in humans (10-20% of the proteome), 1094 in A. thaliana (6% of the proteome) and over 50 in S. cerevisiae. These can be grouped into three broad categories - poly-acylated membrane proteins (e.g., some receptors and rhodopsin), mono-acylated membrane proteins (some receptors and viral proteins) and hydrophilic proteins (such as certain protein kinases). In brain, 490 palmitoylation sites have been identified on 342 synaptic proteins, 44% of which are integral membrane proteins. A comparison of orthologues between such distant phyla as mammals and Drosophila melanogaster suggests that S-palmitoylated proteins are more conserved than are non-S-palmitoylated proteins.
 Thio-acylation: The reaction occurs post-translation of the protein and is catalysed by
endomembrane-bound acyltransferases at specific cysteine residues exposed to the cytosol.
Although there is some evidence for occasional non-enzymatic palmitoylation, enzymatic mechanisms predominate,
and palmitoyl S-acyltransferases were identified definitively first from yeasts and subsequently from mammalian cells.
Families of such enzymes (23 in humans coded by more than 20 separate genes, 24 in mice, a similar number in Arabidopsis and 7 in yeasts)
have now been characterized with a conserved cysteine-rich domain containing zinc and a distinctive aspartate-histidine-histidine-cysteine
motif (DHHC or ZDHHC) within a cysteine-rich region, where the catalytic reaction occurs.
They are membrane proteins with many subcellular locations that span the bilayer at least four times,
with the DHHC domain and the N- and C‑terminal domains on the cytosolic face.
In most palmitoyl transferases, there is a conserved C‑terminus (PaCCT) domain, and this is required for their function.
With human DHHC20, it has been established that the fatty acyl chain of palmitoyl CoA inserts into a hydrophobic pocket within the
transmembrane spanning region of the protein, while the CoA headgroup is recognized by the cytosolic domain through polar and
ionic interactions.
Thio-acylation: The reaction occurs post-translation of the protein and is catalysed by
endomembrane-bound acyltransferases at specific cysteine residues exposed to the cytosol.
Although there is some evidence for occasional non-enzymatic palmitoylation, enzymatic mechanisms predominate,
and palmitoyl S-acyltransferases were identified definitively first from yeasts and subsequently from mammalian cells.
Families of such enzymes (23 in humans coded by more than 20 separate genes, 24 in mice, a similar number in Arabidopsis and 7 in yeasts)
have now been characterized with a conserved cysteine-rich domain containing zinc and a distinctive aspartate-histidine-histidine-cysteine
motif (DHHC or ZDHHC) within a cysteine-rich region, where the catalytic reaction occurs.
They are membrane proteins with many subcellular locations that span the bilayer at least four times,
with the DHHC domain and the N- and C‑terminal domains on the cytosolic face.
In most palmitoyl transferases, there is a conserved C‑terminus (PaCCT) domain, and this is required for their function.
With human DHHC20, it has been established that the fatty acyl chain of palmitoyl CoA inserts into a hydrophobic pocket within the
transmembrane spanning region of the protein, while the CoA headgroup is recognized by the cytosolic domain through polar and
ionic interactions.
Each of the mammalian DHHC proteins is associated with characteristic subcellular locations but mainly the endoplasmic reticulum and Golgi, with a few at the plasma membrane and some at more than one location, often with some preference for particular tissues. Simplistically, Golgi-located palmitoyl acyltransferases promote the palmitoylation and forward trafficking of substrates to the plasma membrane, while those in other organelles tend to generate the products for local use. In plants, many of the enzymes are located at the plasma membrane.
DHHC acyltransferases operate by a two-step mechanism in the first step of which the enzymes are auto-acylated by a nucleophilic attack of the active site cysteine on the thioester of palmitoyl CoA with generation of an acyl-enzyme intermediate and release of CoA. In the second step, the acyl group is transferred to a protein substrate in a trans-acylation reaction. Cysteine palmitoylation forms a thioester bond that is similar in energy to that in the palmitoyl donor, palmitoyl-CoA, so the reaction does not need an energy source such as ATP. There is an absolute requirement for long-chain acyl-CoA esters, mainly 16:0, as fatty acyl donors, though some members of the DHHC protein family have different acyl specificities and at least one can use a wide variety of fatty acyl-CoA substrates. As these can have characteristic tissue distributions, there can be differences in the nature of the thio-esterified fatty acids between organs. The acyl chain binds in a lengthy cavity containing hydrophobic residues formed by the transmembrane domains, and the acyl chain-length selectivity depends upon cavity variants with preferences for particular acyl chains.
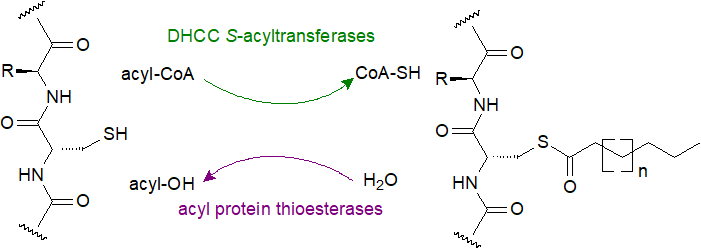 |
| Figure 4. Protein palmitoylation-depalmitoylation. |
It is not yet fully understood how substrate recruitment and specificity are regulated for palmitoylation. One DHHC enzyme can often palmitoylate many different protein substrates, while a given protein may be palmitoylated by more than one acyl transferase. On the other hand, there are examples where certain proteins are palmitoylated by only one DHHC enzyme. One common feature is that the catalytic sites on the enzymes reside at the membrane-cytosol interface so that membrane-proximal cysteines are candidates for palmitoylation. While in general there is no preferred substrate-sequence motif in target proteins, some DHHCs contain further sequences of amino acids (SH3 and Akr domains, PDZ binding motifs) that may be associated with enzyme-substrate recruitment. There is a suggestion that acyl-CoAs do not diffuse freely in the cytosol but are channelled into appropriate pathways by mechanism that may involve compartmentalization and intracellular transport. Some DHHCs must be palmitoylated by another before encountering their substrate protein. Prior to S-acylation, modification by N‑myristoylation or prenylation is often required, and with the former, target sequences for subsequent palmitoylation have been identified with further regulation by post-translational phosphorylation or ubiquitylation.
De-acylation: In contrast to irreversible N-myristoylation, hydrolysis of S-palmitoylated proteins occurs readily and is catalysed by thioesterases, two of which have been characterized in the cytosol, i.e., acyl protein thioesterase 1 (APT1 or LYPLA1) and 2 (APT2 or LYPLA2) each with specificities for particular classes of protein conjugates. Among other such enzymes, two lysosomal enzymes, palmitoyl protein thioesterases 1 and 2 (PPT1 & 2) and a mitochondrial enzyme (ABHD10) are known. All of these belong to the ‘metabolic’ serine hydrolase superfamily, with a catalytic serine residue for substrate hydrolysis and the ability to cleave ester, amide, or thioester bonds on many classes of molecules. Thus, most proteolipids of this type undergo cycles of acylation-deacylation, with a half-life (seconds to several minutes) that is much shorter than that of the peptide per se, and this permits proteins to shuttle between membranes and other cellular compartments, such as between the plasma membrane and Golgi in both directions. For example, after extracellular stimulation, Ras-proteins produce signals that lead to cell proliferation, differentiation and apoptosis; they are palmitoylated for transport to the plasma membrane then de-palmitoylated for transport back to the Golgi (in this instance by a family of ABHD17 serine hydrolases) so at the steady state, these proteins can signal at both organelles in a continual and essential cycle. Some proteins are prenylated and S-acylated in an active form, while the inactive form is only prenylated. Such acylation-deacylation cycles are regulatory, and synthetic and hydrolytic enzymes are themselves regulated dynamically by extracellular factors like phosphorylation, with the level of palmitoylation determined by a finely tuned balance between the enzymes in various cellular locations.
Function:  As with the myristoylated proteins, palmitoylation modifies protein behaviour partly by altering their
conformations, but mainly in targeting otherwise soluble proteins to the relevant membranes or receptors with the number of bound
fatty acyl groups controlling the strength of the interaction with membranes.
A hydrophobic protein with a single acylation can bind only loosely to membranes and is easily displaced, but a second or further acylation
ensures strong targeting of a protein to the cytoplasmic face of the membrane and guarantees that it is firmly bound to a site
where an appropriate receptor may be located.
While many reported S-acylation substrates are already transmembrane proteins, acylation on a cytosolic cysteine brings it to the
membrane.
As with the myristoylated proteins, palmitoylation modifies protein behaviour partly by altering their
conformations, but mainly in targeting otherwise soluble proteins to the relevant membranes or receptors with the number of bound
fatty acyl groups controlling the strength of the interaction with membranes.
A hydrophobic protein with a single acylation can bind only loosely to membranes and is easily displaced, but a second or further acylation
ensures strong targeting of a protein to the cytoplasmic face of the membrane and guarantees that it is firmly bound to a site
where an appropriate receptor may be located.
While many reported S-acylation substrates are already transmembrane proteins, acylation on a cytosolic cysteine brings it to the
membrane.
Palmitoylation of integral membrane proteins may be regulatory, changing their conformation to increase their stability through protecting them from degradation by preventing ubiquitinylation and increasing their resistance to proteases. It is a factor in the process of trafficking proteins between organelles and in directing them to appropriate membrane compartments. In neurons, palmitoylation targets proteins for transport to nerve terminals and may regulate trafficking at synapses; it enables the growth and integrity of neuronal axons and is needed to convey axonal signals to influence learning and memory. Disturbance of these processes can result in brain disorders associated with cognitive defects.
The saturated acyl moiety in palmitoylation facilitates transfer of proteins to lipid rafts, subdomains of the plasma membrane that are enriched in sphingolipids and cholesterol. Thus, S-palmitoylation enables flotillins to bind to the cytosolic leaflet of the plasma membrane in raft domains, where they oligomerize to serve as scaffolds that facilitate the assembly of multiprotein complexes at the membrane–cytosol interface. Proteins with many S-acylated cysteines near the membrane have the potential to induce formation of membrane nanodomains de novo through a phase-separation mechanism, and one such is the SARS-CoV-2 spike protein, which forms trimers and carries ten acylation sites per protomer. In association with nanodomains, proteolipids can participate in cell signalling events such as in T-cells of the immune system, where the unique feature of reversibility in lipid S-palmitoylation modifications is advantageous.
In general, S-palmitoylation stimulates signalling by the lipidated proteins. Protein S-palmitoylation is a basic mechanism for control of the properties and operation of ion channels, both directly and indirectly via other signalling pathways, and via stimulation of calcium channels in the plasma membrane and endoplasmic reticulum and its influence upon the location and function of key regulators, this regulates apoptosis. Similarly, in necroptosis, S-acylation can enhances membrane binding and membrane permeabilization, so contributing to cell death and inflammatory responses, and it is a factor in autophagy. In the heart, S-palmitoylation is implicated in the regulation of cardiac electrophysiology, including the sodium-calcium exchanger, phospholemman, the cardiac sodium pump and the voltage-gated sodium channel. Mitochondrial morphology and function are regulated by S‑stearoylation of human transferrin receptor 1. S‑Palmitoylation enables macrophages to respond appropriately in processes such as endocytosis, intracellular signalling, lysosomal sorting and chemotaxis. Among many further examples that could be listed, S-acylation regulates components of the insulin secretion and insulin response pathways, including channels, transporters and receptors, and it influences osteoblast and osteoclast differentiation, so contributing to skeletal homeostasis.
Palmitoylation is a factor in lipoprotein metabolism. Lipoprotein particles containing apolipoprotein B (apo B), such as chylomicrons, VLDL and LDL, transport triacylglycerols and cholesterol esters in plasma, and it has been established that palmitoylation of apo B regulates the biogenesis of the nascent lipoprotein particles that contain this apolipoprotein and may control the amount available for lipid transport.
Dysregulation of DHHC palmitoyl acyltransferases is known to be associated with many human diseases, including schizophrenia, X‑linked mental retardation, Huntington's disease and cancer, and these enzymes are now seen as targets for drug development. Protein palmitoylation is crucial for many signalling pathways in tumours that affect their occurrence and progression and the therapeutic response, so that both palmitoylases and depalmitoylases are seen as potential targets for anti-cancer treatment. In relation to cardiovascular diseases, S-palmitoylation influences cardiac myocytes by regulating ion channels and signal transduction and may be a factor in arrhythmia.
 Infections: The innate immune system responds to invasion by microorganisms by signalling through pattern
recognition receptors (PRRs) that bind conserved microbial features.
For example, NLRP3 is an inflammasome seeding PRR that initiates a pro-inflammatory signalling cascade in response
to bacterial infection or tissue damage, and S-acylation is an important modifier of its function.
Many other receptors, including the Toll-like receptors, are S‑palmitoylated, and this may be a required
modification for much innate immune signalling.
Infections: The innate immune system responds to invasion by microorganisms by signalling through pattern
recognition receptors (PRRs) that bind conserved microbial features.
For example, NLRP3 is an inflammasome seeding PRR that initiates a pro-inflammatory signalling cascade in response
to bacterial infection or tissue damage, and S-acylation is an important modifier of its function.
Many other receptors, including the Toll-like receptors, are S‑palmitoylated, and this may be a required
modification for much innate immune signalling.
Protein S-palmitoylation is critical for the pathogenicity of many human and plant pathogens, including fungal, bacterial and viral infections. While parasitic protozoa and fungi possess their own palmitoyltransferases, viruses and bacteria hijack the enzymes in their hosts to favour their internalization, survival and replication inside the cells. Palmitoylation of viral proteins is essential for their life cycle, and three types of membrane proteins in viruses, including many that are highly pathogenic, are known to be S-acylated, a factor that is necessary for the immune response. Palmitoylated 'spike' proteins are the main transmembrane proteins in the viral envelope, and they assist the entry of viruses into cells by catalysing receptor binding and/or membrane fusion. The viroporins are a second group, which are freely expressed in infected cells but not into virus particles per se to any appreciable extent; they possess one or two membrane-spanning regions, which serve as hydrophilic pores in membranes. A third diverse group of palmitoylated proteins produced by viruses are peripheral membrane proteins in which the fatty acid component simply anchors the modified protein to a membrane. In contrast to other proteins, glycoproteins of viral membranes are palmitoylated at or near the cytoplasmic face and then remain palmitoylated. S‑Palmitoylation is necessary for the survival, growth and infectivity of the trypanosomatids (protozoal parasites) in humans.
Plants: Numerous S-acylated proteins are known to be present in higher plants, and many are now being described, although progress appears to have been slower than with mammalian systems. As in animals, S‑acylation alters protein interactions with membranes together with their activation state, conformations and sub-cellular distributions, and the cycle of S-acylation and de-S-acylation provides a molecular mechanism for membrane-associated proteins to undergo cycling and trafficking between different cellular compartments. SNARE and G proteins and the cellulose synthase complex are among several proteins known to be modified in this way with effects on fertility, Ca2+ signalling, movement of potassium ions, stress signalling (e.g., the immune response), root hair and pollen tube formation, cell death, production of reactive oxygen species, cell expansion and division, gametogenesis and salt tolerance. The 18 subunits of the cellulose synthase A family have numerous S‑acyl groups (70–110), rendering it the most heavily S-acylated complex ever described. As this complex is integral to the plasma membrane and extrudes cellulose microfibrils into the extracellular environment to form the cell wall, the extrusion process enables the complex to pass through the plane of the plasma membrane to form microdomains and recruit accessory proteins for unhindered transport across the membrane.
Few S-acylated proteins have been matched to the any of the 24 S-acyl transferases in Arabidopsis, so each enzyme may have multiple substrates (as many as 100) participating in diverse processes. Protein thioesterases comparable to those in animals that reverse the reaction have not yet been characterized in plants, although two candidate enzymes have been identified that unexpectedly may not be in the serine hydrolase superfamily. S-Palmitoylation may be more important at the plasma membrane in plants than in animals.
Analysis: Modern mass spectrometric methods identification of S-palmitoylated proteins and permit location of the acyl group to defined cysteine residues when used in concert with a site-specific acyl-biotin-exchange reaction in which palmitoyl-thioester bonds are cleaved with hydroxylamine for labeling of newly exposed thiols with biotin for affinity purification and detection. In contrast to the N-acylated proteins, the fatty acids are easily released from thiol linkage by base-catalysed transesterification for analysis by gas chromatography.
4. Prenylated Proteins
Prenylated proteins are formed by attachment of isoprenoid lipid units, farnesyl (C15) or geranylgeranyl (C20), via cysteine thio-ether bonds, which are more stable than ester or thioester bonds, at or near the carboxyl terminus. Such proteins were first detected in fungi, but they are ubiquitous in mammalian cells where they can amount to up to 2% of the total proteins, and they are increasingly being found in other eukaryotic organisms. Some proteins from pathogenic bacteria and viruses can be prenylated by their hosts, apparently as a protective measure. Isoprenylation is a stable (non-reversible) modification, which targets proteins to membranes, aids protein-protein interactions and enables their functions. In contrast to the acylated proteins, the bulky branched nature of the lipid moiety of isoprenylated proteins ensures that the latter cannot be incorporated into ordered raft microdomains.

Whether a protein is prenylated is determined by characteristic amino acid sequence motifs at the carboxyl terminus, principally a CAAX sequence with cysteine (C) attached to two aliphatic amino acids (A) then to a variable carboxyl-terminal amino acid residue (X). The nature of the X residue determines whether a protein will be farnesylated or geranylgeranylated; with a few exceptions, proteins ending in serine, methionine, alanine or glutamine are farnesylated, while proteins ending in leucine or isoleucine are geranylgeranylated. A single farnesylation is usual, but some dual geranylgeranylation can occur. Proteins that participate in cellular signalling and trafficking pathways are the main targets, and the best known of these are probably the Ras super-family (discussed above).
The isoprenoid units are produced by the mevalonate pathway, as discussed on our web page dealing with cholesterol with farnesyl pyrophosphate as the branch-point in sterol/isoprene synthesis. Subsequent biosynthesis of prenylated proteins involves a concerted series of reactions in which the proteins are transported through various cellular organelles, ending mainly but not only at the plasma membrane. Prenylation occurs in the cytoplasm of the cell after synthesis of the protein per se, with farnesyl or geranylgeranyl pyrophosphate as the isoprenoid substrate, each catalysed by its own transferases, four of which are known, i.e., protein farnesyltransferase (FTase) and protein geranylgeranyl transferases (three types GGTase-1/2/3). The enzymes transfer the isoprenoid group to the cysteine residue in the CAAX box, and enzyme-bound Zn2+ is necessary for the proper transfer of the lipid anchor. Cleavage of the terminal tripeptide (AAX) occurs in the endoplasmic reticulum via a protease, before the new terminal cysteine is enzymically methylated at the carboxyl group with S-adenosyl methionine as the methyl donor (further increasing the hydrophobicity of the proteolipid). GGT-2 is known to transfer two geranylgeranyl units but only to the C-terminal double-cysteine motif (CC or CXC) of the 'Rab' family of proteins and requiring Rab escort proteins.
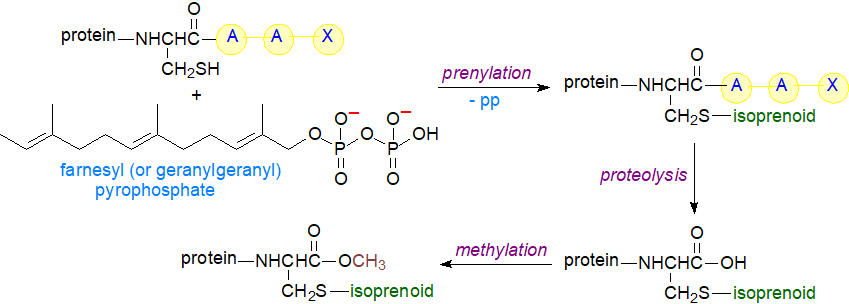 |
| Figure 5. Prenylation of proteins. |
Prenylation alone tends to target proteins from the cytosol to endomembranes, such as the endoplasmic reticulum and Golgi, a process facilitated by several binding or chaperone proteins, but S-acylation (palmitoylation) can then occur to increase the affinity for membranes before proteins are directed from the endomembranes to the plasma membrane; Ras proteins are transported from the Golgi to the plasma membrane by this means. As the second modification is reversible, it may be part of a control mechanism. After they have been fully processed, these proteolipids have a high affinity for cellular membranes and possess a unique structure at their carboxyl termini, which is a recognition motif in some protein-protein interactions. Rho proteins are an exception in that they can be anchored to the plasma membrane, endomembranes or endosomes by geranylgeranylation alone. Although the prenylation reaction is irreversible, some geranylgeranylated Rho proteins can be removed from membranes and de-activated by binding via protein-protein interactions; prenylated proteins can also be trafficked between membranes in this way. Binding between prenylated and non-prenylated proteins can serve to increase the activity of the latter, and many examples of this have now been documented.
Protein farnesylation (Ras) and geranylgeranylation (Rho) are critical for Treg cell differentiation, maintenance and function. Prenylated proteins of the Ras family are attached to the cytoplasmic face of the cell membranes, where they transfer the signal from surface receptors to transcription factors for gene expression in the nucleus. In C. elegans, geranylgeranylation of the small G protein RAB-11.1 for interaction with the nuclear hormone receptor NHR-49 is part of a mechanism that enables cells to sense metabolic demand due to lipid depletion and respond by increasing nutrient absorption and lipid metabolism.
Ultimately, degradation of prenylated proteins in animals occurs in the lysosomal compartment of the cell and is catalysed by a prenylcysteine lyase (also known as prenylcysteine oxidase 1), which is a flavin-containing monooxygenase that converts prenylcysteine to prenyl aldehyde by a novel mechanism. This may regulate the protein prenylation pathway and may be especially relevant in disease conditions.
As many of these proteins take part in the development of cancer, they are the subject of much pharmaceutical interest, focusing especially on the inhibition of the prenylation reaction with Ras proteins and a farnesyltransferase inhibitor for patients with acute myeloid leukaemia is undergoing clinical trials. Inhibitors of protein farnesyltransferase have been shown to be efficacious in the treatment of protozoal pathogens and other parasitic diseases in animal models, and they may be of value in the treatment of viral (e.g., hepatitis D), bacterial and fungal infections. Defects in prenylation or its regulation have been implicated in the rare disease progeria, and in cardiovascular disease, neurodegenerative disorders and metabolic diseases. Prenylations are abundant in the retina, especially photoreceptor cells, where disruption can lead to retinopathies.
In plants, protein prenylation is required for plant growth, meristem development and environmental (stress) responses, including the control of abscisic acid and auxin signalling. At least 250 proteins in Arabidopsis have the CAAX sequence and have the potential to be prenylated, although this has been demonstrated experimentally for only a few. Some prenylated proteins do not use the prenyl group to bind to membranes, but this does permit otherwise hydrophilic proteins to operate as peripheral lipid membrane proteins or as a signal for interaction with other proteins. Three major types of prenyltransferases have been characterized in plants, i.e., protein farnesyltransferase, protein geranylgeranyltransferase I and protein geranylgeranyltransferase II, all of which are heterodimeric enzymes.
5. O-Acylated Proteins and Peptides
Wnt proteins: In a few proteolipids, serine or threonine residues are acylated such that an O-acyl rather than an S-acyl linkage is formed, and the family of 'Wingless' or Wnt proteins in animals (19 members in humans) falls into this category. These are central mediators of embryonic development and tissue renewal that influence cell proliferation, differentiation and migration, and they need O‑acylation for secretion and activity. They are secreted glycoproteins, which share a conserved sequence of cysteine residues and an N‑terminal signal sequence that targets them for secretion. In all, there is an unusual O‑acyl modification with palmitoleic acid (9‑cis‑hexadecenoic or 9‑16:1), which has been termed a lipokine (i.e., a lipid hormone), at a conserved serine residue (and not with S‑palmitoylation on a conserved cysteine residue as originally reported, although some members of the Wnt family undergo a further S‑acylation with palmitate at a cysteine residue). Following synthesis, Wnt proteins are processed in the endoplasmic reticulum, where the conserved signal sequence is cleaved before glycosylation and fatty acylation.
Desaturation of palmitoyl-CoA to generate a dedicated pool of palmitoleoyl-CoA occurs in the endoplasmic reticulum by the action of stearoyl-CoA desaturase (SCD1), and the subsequent O-acylation uses an O-acyltransferase termed 'porcupine' (PORCN), which is entirely distinct from the S‑acyltransferases (one inhibitor of this enzyme is undergoing clinical trials for treatment of Wnt-driven solid tumours). The specificity of the reaction is due to a structural feature in the enzyme that will only permit the use of the cis-9-monoenoic fatty acid; only those Wnt proteins esterified in this way can separate from the PORCN-Wnt complex. PORCN is a member of the MBOAT family (membrane-bound O-acyl transferase) of multi-pass transmembrane proteins like GOAT and Hhat (see below) that carries out fatty acylation of secreted signalling proteins as well as having a role in glycerolipid metabolism.
The O-palmitoleoyl residue is essential for intracellular trafficking and operation of Wnt proteins within cells and for maintaining their structural integrity, and it assists their secretion by facilitating movement from the trans Golgi network to the plasma membrane in signalling cells. The transport mechanism involves transfer to the co-chaperone 'Wntless' or 'WLS' (G protein-coupled receptor 177 (GPR177)), a conserved membrane protein that binds to the palmitoleoyl residue of Wnt proteins and facilitates transport by vesicular or non-vesicular means to the plasma membrane and then across it in complex with SFRP and WIF1 carriers. Serine acylation may be required for extracellular long-range transport of Wnt proteins in lipoprotein particles with protein chaperones or in extracellular vesicles. In turn to initiate signalling, Wnt proteins are handed over to cell-surface receptors of the 'Frizzled' receptor family with a co-receptor, glypicans, where the palmitoleoyl residue fits neatly within a hydrophobic cleft in the receptor to enable ligand-receptor interactions. Wnt proteins are deactivated by an extracellular carboxylesterase ('Notum'), which removes the palmitoleoyl group.
The Wnt signalling pathway is often deregulated in cancer and other diseases with effects upon lipid metabolism, particularly lipid mobilization in that it reduces lipid accumulation in adipocytes (studied in Drosophila) by boosting lipolysis and inhibiting lipogenesis and fatty acid β-oxidation through effects upon target genes thereby regulating homeostasis of triacylglycerols.
Wnt proteins are relatively hydrophobic and are stabilized in the aqueous environment of the cell by binding in a hydrophobic space created by glypicans, i.e., heparan sulfate proteoglycans that are bound to the outer surface of the plasma membrane by a glycosyl-phosphatidylinositol anchor. These serve as a reservoir from which Wnt proteins can be handed over to signalling receptors.
 Ghrelin: Ghrelin is an O-acylated proteolipid, i.e., a circulating 28-amino acid
peptide hormone, which is octanoylated (C8) at a serine residue (third amino acid from the N-terminus).
It is a hunger-stimulating (anorexigenic) hormone produced in endocrine cells of the human stomach and pancreas
that increases caloric intake, decreases energy expenditure and promotes fat deposition.
Only the octanoylated protein binds to the single receptor characterized to date, i.e., growth hormone secretagogue receptor GHS‑R1a,
which results in increased intracellular Ca++, release of growth hormone and recruitment
of multiple proteins to initiate a variety of cell signalling cascades associated with the physiological and behavioural effects of ghrelin.
This receptor can stimulate the phospholipase C pathway with accumulation of diacylglycerols and
inositol phosphates and thence signalling for further downstream events.
The picture is complicated by a finding that GHS-R1a can dimerize and alter the signalling of other G protein-coupled receptors,
while the liver-expressed antimicrobial peptide 2 (LEAP2) is an endogenous antagonist for GHS-R1a and is associated with the regulation of
ghrelin signalling.
Ghrelin: Ghrelin is an O-acylated proteolipid, i.e., a circulating 28-amino acid
peptide hormone, which is octanoylated (C8) at a serine residue (third amino acid from the N-terminus).
It is a hunger-stimulating (anorexigenic) hormone produced in endocrine cells of the human stomach and pancreas
that increases caloric intake, decreases energy expenditure and promotes fat deposition.
Only the octanoylated protein binds to the single receptor characterized to date, i.e., growth hormone secretagogue receptor GHS‑R1a,
which results in increased intracellular Ca++, release of growth hormone and recruitment
of multiple proteins to initiate a variety of cell signalling cascades associated with the physiological and behavioural effects of ghrelin.
This receptor can stimulate the phospholipase C pathway with accumulation of diacylglycerols and
inositol phosphates and thence signalling for further downstream events.
The picture is complicated by a finding that GHS-R1a can dimerize and alter the signalling of other G protein-coupled receptors,
while the liver-expressed antimicrobial peptide 2 (LEAP2) is an endogenous antagonist for GHS-R1a and is associated with the regulation of
ghrelin signalling.
The addition of the octanoyl chain is necessary to induce a conformational change in ghrelin with formation of a hydrophobic core that promotes access to the receptor ligand-binding pocket. Octanoic acid is presumably obtained by β-oxidation of long-chain fatty acids in ghrelin-producing cells, before the ghrelin O‑acyltransferase (GOAT), a member of the MBOAT family of acyltransferases, catalyses octanoylation of pro-ghrelin in the Golgi. This reaction is unique in that ghrelin is the only known octanoylated protein and the only known substrate for this enzyme. After translocation to the endoplasmic reticulum, ghrelin is generated from octanoyl-proghrelin by the action of prohormone convertase 1/3. Mature ghrelin is stored within secretory granules of X/A-like cells, and upon fasting, it is released into the circulation to stimulate appetite. GOAT is now perceived as a target for pharmaceutical intervention in the treatment of the metabolic syndrome. Some circulating esterases can deacylate ghrelin, including the extracellular carboxylesterase ('Notum'), which removes the palmitoleic acid group from Wnt proteins.
Other O-acylated proteins: Aside from ghrelin and the Wnt proteins, relatively few proteins are known to be O-acylated with long-chain fatty acids in animals. One exception is O‑palmitoylation of a serine residue on the protein histone H4 catalysed by the acyl-CoA:lysophosphatidylcholine acyltransferase I (LPCAT1), better known for generating the pulmonary surfactant dipalmitoylphosphatidylcholine from lysophosphatidylcholine. This is presumed to be a mechanism for regulating mRNA synthesis that may lead to changes in global transcription in the cell. O-Palmitoylation of the threonine residue in the C-terminus of the spider venom neurotoxin PLTX-II may regulate toxin potency. In the model nematode C. elegans, serine can be O-acylated with many different fatty acids, including eicosapentaenoic acid. Corynebacteria (e.g., C. glutamicum) contain as many as 30 proteins O-linked to the multi-branched mycolic acids in the cell envelope, a unique post-translational modification in bacteria.
6. Hedgehog Proteins - linked Covalently to Cholesterol and Palmitate
Hedgehog (Hh) proteins have a major role in signalling during the differentiation of cells in the development of all embryos from fruit flies to fish to humans, while after embryogenesis, Hh signalling coordinates reparative and regenerative responses in many tissues. They were first found and studied in the insect model Drosophila melanogaster (and named for an anomalous cuticular feature reminiscent of a hedgehog's spines in a mutant), but they are now known to occur in all higher organisms. Vertebrates express three hedgehog family proteins designated 'Sonic' (Shh), 'Indian' (Ihh) and 'Desert' (Dhh) hedgehog, of which Shh has attracted most interest, and they are required for an extensive range of processes from the control of left-right asymmetry of the body to the specification of individual cell types within the brain and to limb development. In endocrine tissues, Hh signalling triggers conversion of cholesterol to progesterone and estradiol. A unique feature is that these proteins contain both palmitic acid and cholesterol in covalent linkage, further confirmation if needed of the vital importance of these occasionally controversial lipids in animal tissues. Proteins that operate in the same way but are structurally distinct are found in nematodes.

Hedgehog proteolipids are formed post-translationally by attachment of cholesterol via an ester bond to glycine at the C-terminus, a highly conserved region of the protein, while a palmitoyl moiety is attached to a cysteine residue at the N-terminus (N-palmitoylation). The signalling proteins are synthesised initially as ~45 kDa inert propeptides with two characteristic domains, i.e., an N-terminal 'hedge' domain and a dual-purpose C-terminal autoprocessing and cholesterol recognition domain. After the signalling sequence is removed (1), there is a unique reaction (2) in the endoplasmic reticulum (ER) in which there is an intramolecular attack of an internal cysteine on the carbonyl of an adjacent glycine, resulting in a rearrangement whereby a peptide bond is replaced with a labile thioester bond. This is followed by a transesterification reaction of the thioester bond with the hydroxyl group of a cholesterol moiety (3) facilitated by the processing domain. Cholesterol is thus bound covalently to the C-terminus of the N-terminal signalling domain and simultaneously cleaves this from the processing domain at the cholesteroylation site. Finally, the nascent Hh-N is further modified by the addition of an N-palmitoyl group at Cys-24 (4) at the ER, a reaction mediated by a palmitoyl acyltransferase (Hedgehog acyltransferase or Hhat, another MBOAT family member) to create a highly hydrophobic molecule that is often described as 'Hh-Np for Hh-N-processed' and is transported by a secretory pathway to the plasma membrane.
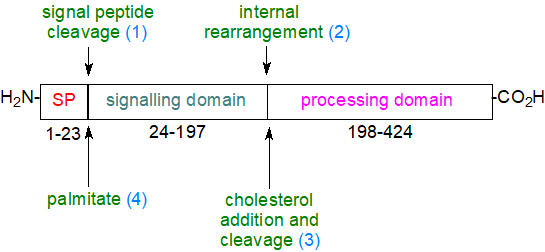 |
| Figure 6. Maturation of a hedgehog protein. |
Unlike the other lipid-modified proteins discussed above, but like the GPI-anchored proteins, the lipid moieties are located in the exoplasmic or exterior leaflet of the plasma membrane with the protein component in the extracellular region. The process is regulated both positively and negatively by various oxy-cholesterol derivatives including vitamin D3 and downstream metabolites of 7‑dehydrocholesterol, possibly by competing for binding with sterol-sensing domains (like those in proteins involved in cholesterol homeostasis) in receptor and other proteins, and the interactions between sterol metabolism and hedgehog signalling are increasingly a focus for research.
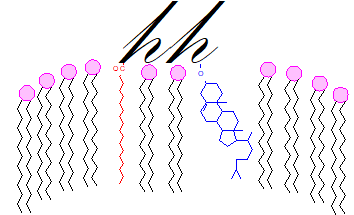 |
| Figure 7. A hedgehog protein in the outer leaflet of a membrane. |
Following biosynthesis in the endoplasmic reticulum and Golgi, there are mechanisms to transport the final proteolipid from the plasma membrane to other cells often for an appreciable distance from the site of synthesis (as much as 30 cell diameters), but much remains to be learned of how this is accomplished. There are reports that both lipid components are essential for the proper tissue distribution and efficient operation of the attached proteins, and the N‑palmitoyl moiety causes the proteins to form active multimeric complexes, while the cholesterol modification contributes to the partitioning of hedgehog proteins into plasma lipoprotein complexes for long-range transport. On the other hand, there is an alternative proposal that the N-palmitoyl moiety may be removed during translocation into the extracellular compartment and that the residual N-terminal peptide may enhance signalling of depalmitoylated hedgehog to the patched receptor.
In Drosophila and vertebrates, a protein termed 'Dispatched' (DISP), which contains a sterol-sensing domain, is used for transport through the plasma membrane and is dependent on the trans-membrane sodium (Na+) gradient across the membrane, while cell-surface heparan sulfate proteoglycans, consisting of a glycosylphosphatidyl-inositol (GPI)-linked protein core to which varying numbers of linear glycosaminoglycan chains are attached, may be required. Extracellular extraction from the membrane and subsequent distribution of cholesterol-modified Shh is enhanced by its interaction with the secreted protein SCUBE2 (and analogues) with the cholesterol component essential for this purpose. Cholesterol-modified Shh is also shed from the surface of producing cells in exovesicles or "exosomes” derived from the budding of cellular membranes. In insect models, these proteolipids are transported in the form of lipoprotein complexes (lipophorins) with the lipid moieties in a phospholipid monolayer that surrounds a core of triacylglycerols and cholesterol esters.
The mammalian Shh receptor termed 'PTCH1 or Patched' is a transmembrane protein located on the primary cilium, an antennae-like projection of the plasma membrane from the cytoplasm of the cell. Although the ciliary membrane is contiguous with the plasma membrane, it has a distinctive lipid composition and is relatively enriched in phosphatidylinositol 4-phosphate. PTCH1 contains a sterol-sensing domain and controls the cholesterol composition of the ciliary membrane. When it binds to hedgehog proteins, a second G protein-coupled receptor designated 'SMO or Smoothened' is enabled to enter the primary cilium and initiate down-stream signalling by triggering the expression of target genes through a complex network of post-translational processes and translocations. Cholesterol and oxysterols, including 20S‑hydroxycholesterol, 24S,25‑epoxycholesterol and 25R,26-hydroxy-7-oxocholesterol, can bind to the extracellular cysteine-rich domain of SMO and induce hedgehog signalling. In this process, it has been determined that human SMO is modified by cholesterol through an ester bond between the 3β‑hydroxyl group of the latter and the carboxyl group on the side chain of Asp95 of SMO.
Abnormalities in the expression and/or signalling of the Shh hedgehog proteins have been implicated in developmental (morphological) abnormalities, and these can be fatal to embryos and have been linked to disease states in humans. In adults, aberrant metabolism of Hedgehog proteins, especially of Hhat, promotes tumorigenesis in many different cancers, including those of the pancreas, breast and lung, and it can impede myelin development and repair via cholesterol dysregulation in oligodendrocytes. Inhibition of these proteins has therapeutic potential.
7. Other Proteolipids
N-Acetylation of the primary amine in the ε-position of lysine residues of histone proteins in eukaryotes is a process that leads to neutralization of the position’s positive electrostatic charge in a dynamic mechanism for regulation of chromatin formation and behaviour. The acetylated lysine residues interact with a group of proteins, the so-called "readers”, which contain specific acetyl-lysine binding domains and couple acetylation to various down-stream events including gene transcription. The reaction is reversible; acetate is added from acetyl-coA by lysine acetyl transferases and removed by lysine deacetylases, while some lysine acetyl transferases can catalyse reaction with other short-chain acyl moieties, including crotonylation, succinylation and propionylation. Although the histone proteins, such as the sirtuins, were the first to be identified, it is now recognized that many other proteins are acetylated in this way in both prokaryotes and eukaryotes. N‑Terminal acetylation of proteins by acetyl transferases can occur, but in contrast this is an irreversible reaction, which determines the subcellular localization for certain proteins and modulates protein-protein interactions for normal development of bone, blood vessels and other tissues.
N-Butyrylation of lysine residues of proteins has also been observed in plants and animals, and in the latter has been linked to the onset and progression of several diseases, such as cancer and cardiovascular diseases. In rice, butyrylation is implicated in gene expression and thence in growth, development and metabolism.
For practical reasons, the GPI-anchored proteins are discussed elsewhere in these web pages, as are the ceramides and related lipids bound to proteins in skin. In yeasts, a covalent conjugate of phosphatidylethanolamine with a protein designated ‘Atg8’ is a factor in the process of autophagy (controlled degradation of cellular components) by promoting the formation of membrane vesicles containing the components to be degraded.
Recommended Reading
- Ashok, S. and Rao, S.R. Updates on protein-prenylation and associated inherited retinopathies. Front. Ophthal., 4, 1410874 (2024); DOI.
- Buszka, A., Pytys, A., Colvin, D., Wlodarczyk, J. and Wojtowicz, T. S-Palmitoylation of synaptic proteins in neuronal plasticity in normal and pathological brains. Cells, 12, 387 (2023); DOI.
- Busquets-Hernández, C., Tsiotsia, A., Pipitò, L., Chamberlain, L.H., Greaves, J. and Triola, G. Different chains for different gains: How acyl chain diversity shapes S-acylated protein function. Prog. Lipid Res., 100, 101354 (2025); DOI.
- Ding, W.L., Gu, J.Y., Xu, W.Y., Wu, J., Huang, Y.W., Zhang, S. and Lin, S.X. The biosynthesis and applications of protein lipidation. Chem. Rev., 124, 12176-12212 (2024); DOI.
- Eftekhari, A., Sabir, U. and Kasumov, T. The role of lysine acetylation in metabolic sensing and proteostasis. Pharmacol. Therapeut., 274, 108908 (2025); DOI.
- Giglione, C. and Meinnel, T. Mapping the myristoylome through a complete understanding of protein myristoylation biochemistry. Prog. Lipid Res., 85, 101139 (2022); DOI.
- Kallemeijn, W.W., Lanyon-Hogg, T., Panyain, N., Grocin, A.G., Ciepla, P., Morales-Sanfrutos, J. and Tate, E.W. Proteome-wide analysis of protein lipidation using chemical probes: in-gel fluorescence visualization, identification and quantification of N-myristoylation, N- and S-acylation, O‑cholesterylation, S-farnesylation and S-geranylgeranylation. Nature Protocols, 16, 5083-5122 (2021); DOI.
- Kumar, M., Carr, P. and Turner, S.R. An atlas of Arabidopsis protein S-acylation reveals its widespread role in plant cell organization and function. Nature Plants, 8, 670-681 (2022); DOI.
- Li, J.C., Zhang, M.Q. and Zhou, L.J. Protein S-acyltransferases and acyl protein thioesterases, regulation executors of protein S-acylation in plants. Front. Plant Sci., 13, 956231 (2022); DOI.
- Lin, C.H., Chin, Y., Zhou, M., Sobol, R.W., Hung, M.C. and Tan, M. Protein lipoylation: mitochondria, cuproptosis, and beyond. Trends Biochem. Sci., 49, 729-744 (2024); DOI.
- Liu, H.C., Wen, S., Xu, C., Kang, X.H. and Kong, E.Y. Mechanisms and functional implications of ZDHHC5 in cellular physiology and disease. J. Lipid Res., 66, 100793 (2025); DOI.
- Mesquita, S.F., Abrami, L., Linder, M.E., Bamji, S.X., Dickinson, B.C. and van der Goot, F.G. Mechanisms and functions of protein S-acylation. Nat. Rev. Mol. Cell Biol., 25, 488-509 (2024); DOI.
- Resh, M.D. Open Biology: overview for special issue on dynamics of protein fatty acylation. Open Biol., 11, 210228 (2021); DOI - Introduction to a series of publication on this topic.
- Siebold, C. and Rohatgi, R. The inseparable relationship between cholesterol and hedgehog signaling. Annu. Rev. Biochem., 92, 273-298 (2023); DOI.
- Tian, C. and Wang, Q. Protein prenylation in plants: mechanisms and functional implications. Plants-Basel, 14, 1759 (2025); DOI.
- Wang, B., Dai, T., Sun, W.H., Wei, Y.J., Ren, J., Zhang, L., Zhang, M.D. and Zhou, F.F. Protein N-myristoylation: functions and mechanisms in control of innate immunity. Cell. Mol. Immun., 18, 878-888 (2021); DOI.
- Wu, W., Zhu, L.,Dou, Z., Hou, Q., Wang, S., Yuan, Z. and Li, B. Ghrelin in focus: dissecting its critical roles in gastrointestinal pathologies and therapies. Curr. Issues Mol. Biol., 46, 948-964 (2024); DOI.
- Xu, M.K. and Xu, B. Protein lipidation in the tumor microenvironment: enzymology, signaling pathways, and therapeutics. Mol. Cancer, 24, 138 (2025); DOI.
- Yu, J. and Virshup, D.M. Functional regulation of Wnt protein through post-translational modifications. Biochem. Soc. Trans., 50, 1797-1808 (2022); DOI.
- Zhang, J.W., Wu, S.Y., Xu, Y.T., Zhang, L., Cong, C., Zhang, M.H., Jiang, Y.H. and Liu, Y. Lipid overload meets S-palmitoylation: a metabolic signalling nexus driving cardiovascular and heart disease. Cell Commun. Signal., 23, 392 (2025); DOI.
- and in relation to the history of the topic -
- Lees, M.B. A history of proteolipids: a personal memoir. Neurochem. Res., 23, 261-271 (1998); DOI.
An S-palmitoylation database is accessible via Blanc, M., David, F., Abrami, L., Migliozzi, D., Armand, F., Bürgi, J. and van der Goot, F.G. SwissPalm: protein palmitoylation database. F1000Res., 4, 261 (2015); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: November 2025 | ||
© The LipidWeb is open access and fair use is encouraged but not text and data mining, AI training, and similar technologies.
