Glucosyl- and Galactosylceramides (Cerebrosides)
There are two natural monoglycosylceramides of special importance in animals, i.e., glucosylceramide and galactosylceramide. They are structural components of membranes, as in the brain where galactosylceramide is required for the maintenance of the structure and stability of myelin and the differentiation of oligodendrocytes. Glucosylceramide is a minor component of all cell types and is most abundant in human skin as part of the permeability barrier, but it is the key intermediate in the biosynthesis of lactosylceramide and thence of complex oligoglycosphingolipids including gangliosides; it is a major component of the membranes of plants and fungi. Although these monoglycosylceramides have very similar structures in that D‑galactose is an epimer of D‑glucose and they differ only in the configuration at C4, they have very different biological properties and many functions that are separate from their structural roles. Only a few other monoglycosyl analogues are produced in nature, notably by some bacteria of the order Sphingomonadales of the α‑proteobacteria.
1. Structure and Occurrence
β-D-Galactosylceramide (Galβ1-1'Cer or simply GalCer) is the principal glycosphingolipid in brain tissue, hence the trivial name "cerebroside", which was first conferred on it in 1874, although it was much later before it was properly characterized. In fact, galactosylceramides are found in all nervous tissues and indeed at low levels in all organs, but in brain they can amount to 2% of the dry weight of grey matter and 12% of white matter or 23% of myelin lipids, where they insulate the axons of neuronal cells and constitute a substantial component of the extended plasma membrane of oligodendrocytes. While galactosylceramide can be sulfated to form a sulfatide or sialylated to form ganglioside GM4, only a small proportion is subjected to further galactosylation to form Gal2Cer as the precursor for the minor gala-series of oligoglycosphingolipids. It is present in some fungi but not in plants (where galactosyldiacylglycerols are important structural lipids).
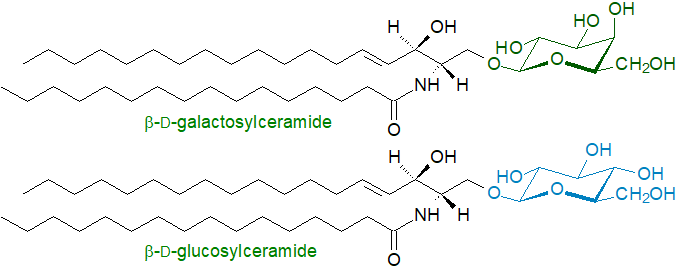 |
| Figure 1. Representative structures of galactosyl- and glucosylceramides |
β-D-Glucosylceramide (Glcβ1-1'Cer or simply GlcCer), trivial name "glucocerebroside", is a major constituent of skin lipids, where it is essential for the maintenance of the water permeability barrier of the skin. Otherwise, it is most abundant in animal tissues such as spleen and erythrocytes as well as in nervous tissues, mainly in the neurons if at low levels, but it is an appreciable component of plant membranes. Higher than normal concentrations of this glycosphingolipid have been reported for the apical plasma membrane domain of epithelial cells from the intestines (e.g., the absorptive villous cells) and urinary bladder. The relative proportions of glucosyl- and galactosylceramide reported for some mouse tissues are - small and large intestines (99:1), spleen (98:2), liver (90:10), lung (70:30) thymus (62:18), kidney (58:42), lymph nodes (15:85) and brain (3:97) (von Gerichten, J. et al. J. Lipid Res., 58, 1247–1258 (2017); DOI.). The d18:1/16:0 molecular species of the two lipids are illustrated.
Animals: In human brain, the galactosylceramides are reportedly enriched in very-long-chain fatty acids (C22 to C26, > 80%). Data from a comprehensive analysis of the fatty acid and long-chain base compositions of cerebrosides from intestines of the Japanese quail are listed in Table 1 for illustrative purposes. The fatty acid components resemble those of other sphingolipids, although the percentage of 2‑hydroxy acids is higher than that in sphingomyelin. They are exclusively saturated in this instance, though a small proportion of monoenoic components may be present in other tissues. Glucosylceramides tend to contain mainly non-hydroxylated fatty acids that are of relatively shorter chain-length. The proportion of trihydroxy bases listed is perhaps higher than in other many other tissues or species studied, probably reflecting the diet. Usually, sphingosine is the main long-chain base in cerebrosides of animal tissues.
Table 1. Composition of fatty acids and long-chain bases (wt % of the total) in cerebrosides of intestines from the Japanese quail.* |
||||
| Long‑chain bases | Fatty acids | Non-hydroxy acids |
2-Hydroxy acids |
|
|---|---|---|---|---|
| % | % | % | ||
| t18:0 | 43 | 16:0 | 5 | 6 |
| d18:0 | 9 | 18:0 | 3 | trace |
| d18:1 | 27 | 20:0 | 2 | 4 |
| t20:0 | 6 | 21:0 | trace | 2 |
| d20:0 | 3 | 22:0 | 4 | 43 |
| d20:1 | 11 | 23:0 | 1 | 13 |
| 24:0 | 3 | 12 | ||
| * The cerebrosides comprised 81% galactosylceramide and 19% glucosylceramide. From Hirabayashi, Y. et al., Lipids, 21, 710-714 (1986); DOI. |
||||
Interestingly, the proportion of galactosylceramides relative to glucosylceramides in myelin glycolipids increases greatly in the ascending phylogenetic tree, and the ratio of hydroxy- to nonhydroxy-fatty acids in cerebrosides increases with the complexity of the central nervous system. There is an intriguing sex difference in kidney, where it has been shown that galactosylceramide rather than glucosylceramide occurs in male mice only (or androgen-treated adult females). Only glucosylceramide is present in the nerves of the most primitive animals (protostomes).
Small amounts of glucosyl- and galactosylceramide that are O-acylated with a fatty acid in various positions of the carbohydrate moiety, most often position 6, have been found in brain tissue of some animals. Novel galactosylceramides acetylated at position 3 of the sphingosine moiety were first located in myelin from rat brain, and molecular species with further galactose O-acetyl modifications are now known to be present in this tissue; these have been termed 'fast-migrating cerebrosides' from their chromatographic properties. In addition, a galactosylceramide with a long-chain cyclic acetal at the sugar moiety, plasmalo-galactosylceramide, has been isolated from equine brain, i.e., the 4',6'-O-acetal derivative with a clearly defined stereochemistry. Some other less common but relevant molecules are described below, including the epimeric α-D-galactosylceramide, which has therapeutic properties.

Glucosylceramide has a vital role as the biosynthetic precursor of lactosylceramide in animals and thence of most of the complex neutral oligoglycolipids and gangliosides.
Plants: Glucosylceramide is the only glycosphingolipid common to animals, plants and fungi, and it is usually considered to be the end-product of the biosynthetic pathway in the last two. While it has often been described incorrectly as the main sphingolipid in plants, this has been because the more polar complex glycosylinositol phosphoceramides are not easily extracted and until relatively recently were not detected by the available methodology; these presumably occupy the biochemical niche for complex sphingolipids in plant membranes. Nonetheless, glucosylceramide is abundant in photosynthetic tissues and constitutes approximately a third of the total sphingolipids, where the main long-chain bases are C18 4,8‑diunsaturated (Z/Z and E/Z) (not sphingosine as illustrated above); it is a major component of the outer leaflet of the plasma membrane and is enriched in the late endosomes, plant tonoplasts and trans-Golgi network-associated secretory vesicles. Small amounts of monoglycosylceramides containing a β‑D‑mannopyranosyl unit may be present in non-photosynthetic tissues (but galactosylceramides are not).
The fatty acid and long-chain base compositions of glucosylceramides from two plant sources are listed in Table 2 (data for the sphingoid bases in glucosylceramide in Arabidopsis are listed elsewhere in this website). Perhaps surprisingly, the fatty acid components are not very different in nature from those in animal tissues in this instance, comprising mainly longer-chain saturated and monoenoic acids, with a high proportion being saturated and having a hydroxyl group in position 2. In the analyses selected for the table here, both di- and tri-hydroxy long-chain bases were found, mainly diunsaturated (Z/Z and E/Z) and almost entirely C18 in chain-length. Much higher concentrations of glucosylceramides are found in pollen than in leaves and with substantial compositional differences.
Table 2. Composition of fatty acids and long-chain bases (wt % of the total) in glucosylceramides of seeds from scarlet runner beans and kidney beans. |
|||||
| Fatty acidsa | Long-chain basesb | ||||
|---|---|---|---|---|---|
| Runner beans | Kidney beans | Runner beans | Kidney beans | ||
| % | % | % | % | ||
| 16:0 | 4 | 5 | t18:0 | trace | trace |
| Other non-hydroxy |
1 | 2 | t18:1-8t | 13 | 11 |
| 14:0-OH | 1 | 1 | t18:1-8c | 10 | 9 |
| 15:0-OH | 1 | 1 | d18:0 | trace | trace |
| 16:0-OH | 58 | 58 | d18:1-8c/t | 1 | 3 |
| 18:0-OH | trace | trace | d18:1-4t | trace | trace |
| 20:0-OH | trace | trace | d18:2-4t,8t | 45 | 60 |
| 22:0-OH | 7 | 6 | d18:2-4t,8c | 31 | 17 |
| 23:0-OH | 2 | 1 | |||
| 24:0-OH | 23 | 23 | |||
| 25:0-OH | 1 | 1 | |||
| 26:0-OH | 1 | 1 | |||
| From Kojima, M. et al., J. Agric. Food. Chem., 39,
1709-1714 (1991); DOI, but see
Yamashita, S. et al. for further data: DOI a including 2-hydroxy acids; b di- and tri-hydroxy bases with cis or trans double bonds in the positions indicated. |
|||||
In Arabidopsis leaves, the long-chain bases consist mainly of t18:1, with relatively little d18:1, t18:0 and d18:0 with 16:0, 24:0 and 24:1 hydroxy fatty acids mainly, although the chloroplast lipid contains mainly hydroxy-16:0 and the endoplasmic reticulum and nucleus have more C24-glucosylceramide. There is no d18:2 base in leaves, although this is 50% of those in pollen. While saturated 2‑hydroxy acids predominate in most plants, some cereal glucosylceramides contain high proportions of mono-unsaturated very-long-chain fatty acids of the n‑9 family. 6-O-Acylceramides ('oryzaceramides A-C') have been isolated from rice bran. Glucosylceramides from algae tend to resemble those from higher plants, although some novel structures have been reported from microalgae.
Fungi: Glucosylceramide is a common component of the lipids of yeast and other fungi, including most fungal pathogens, but it does not occur in the yeast Saccharomyces cerevisiae, which is often used as an experimental model, although trace levels of galactosylceramide have been detected and it may occur more widely. The latter is produced in the rice pathogen Magnaporthe oryzae and is required for cell development and its infectivity. Ceramide monohexosides in fungi are highly conserved molecules, with the ceramide moiety containing the distinctive sphingoid base, (4E,8E)‑9‑methyl-4,8-sphingadienine (or rarely phytosphingosine), linked to 2-hydroxy-octadecanoic or hexadecanoic acids (occasionally these with a trans-double bond in position 3) and with the carbohydrate portion consisting of one residue of either glucose or less often galactose (in contrast to plants). The methyl branch in the sphingoid base may be a recognition feature for plant and insect anti-fungal defences. During different stages of growth (yeast versus mycelial forms), the nature of these can vary dramatically. More complex glycosphingolipids, such as the mannosylinositol phosphorylceramides, have very different sphingoid base and fatty acid compositions, and it is established that the dihydroceramide precursors are generated by enzymes encoded by different genes. When deprived of phosphate, a few bacteria can produce ceramide monohexosides.

Other glycosyl ceramides: Other monoglycosylceramides found in nature
include fucosylceramide, which has been isolated from a colon carcinoma, a xylose-containing cerebroside from an avian salt gland,
mannosylceramides from certain microorganisms, and neuraminic acid linked to ceramide in the phototrophic green sulfur bacterium,
Chlorobium limicola.
 The genus Sphingomonas is unique among gram-negative bacteria in that it lacks
lipopolysaccharides in its outer membrane and instead has two sphingolipids, a tetraglycosylceramide and α‑galacturonosylceramide,
i.e., with a galacturonic acid moiety with an α- rather than a β-linkage to the ceramide unit.
Sphingomonas yanoikuyae contains a similar lipid, but with the sphingoid base component having a cyclopropyl ring.
α‑Glucuronosylceramide from Zymomonas mobilis, a Gram-negative bacterium, contains 2‑hydroxy-myristic acid
as the main fatty acid with sphinganine as the long-chain base.
The genus Sphingomonas is unique among gram-negative bacteria in that it lacks
lipopolysaccharides in its outer membrane and instead has two sphingolipids, a tetraglycosylceramide and α‑galacturonosylceramide,
i.e., with a galacturonic acid moiety with an α- rather than a β-linkage to the ceramide unit.
Sphingomonas yanoikuyae contains a similar lipid, but with the sphingoid base component having a cyclopropyl ring.
α‑Glucuronosylceramide from Zymomonas mobilis, a Gram-negative bacterium, contains 2‑hydroxy-myristic acid
as the main fatty acid with sphinganine as the long-chain base.
Cerebrosides linked to α-D- rather than β-D-galactose occur in a marine sponge (Agelas mauritianus). A few bacteria contain a similar lipid, and it may be of significance that α-galactosylceramides are produced by Bacteroides fragilis, a well-known component of the human intestinal microflora (see the note on this stereoisomer below). In the latter organism, the fatty acid and long-chain bases are saturated and contain iso-methyl-branches. It is now recognized that trace amounts of α-glycosylceramides are present in mammalian cells and are part of the immune response system (0.02% of the galactosylceramides in RBL-CD1d cells, for example).
2. Biosynthesis of Monoglycosylceramides
Ceramides synthesised both de novo and by catabolism of sphingomyelin are used for the biosynthesis of monoglycosylceramides in animal tissues. The biosynthetic mechanism resembles that discussed elsewhere on this website for glycosyldiacylglycerols, i.e., there is a direct transfer of the carbohydrate moiety from a sugar-nucleotide, e.g., uridine 5-diphosphate(UDP)-galactose, UDP-glucose, etc, to a ceramide unit synthesised in the endoplasmic reticulum. During the transfer, which is catalysed by specific glycosyl-transferases, inversion of the glycosidic bond occurs from the alpha to beta configuration.
Synthesis of β-D-galactosylceramide takes place on the lumenal surface of the endoplasmic reticulum, although it has free access to the cytosolic surface by an energy-independent flip-flop process. Expression of the UDP-galactose:ceramide galactosyl transferase (galactosylceramide synthase) is restricted to oligodendrocytes, Schwann cells, kidneys and testes. Prior to biosynthesis of its sulfatide, galactosylceramide is transported to the trans-Golgi compartment.
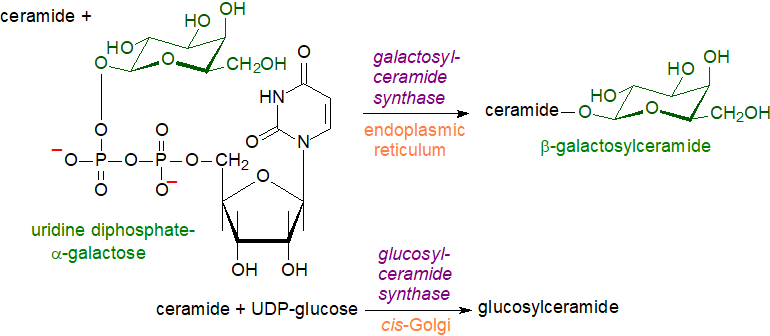 |
| Figure 2. Biosynthesis of galactosyl- and glucosylceramides. |
After transfer of the precursor ceramides from the endoplasmic reticulum to the cytosolic side of the early Golgi membranes with the aid of the CERT protein, glucosylceramide is produced by a glucosylceramide synthase present in this membrane (with the possible exception of neuronal tissues). If this is to be converted to more complex oligoglycosylceramides, it must be translocated to the luminal leaflet of the trans-Golgi membranes, a process that occurs both by vesicular and by non-vesicular transport. The latter is mediated by a conserved clade of integral membrane proteins, i.e., phospholipid flippases (P4-ATPases) designated ATP10A and ATP10D, together with the phosphate adapter protein-2 (FAPP2) and glycolipid transfer protein (GLTP) in humans with related enzymes in fungi, which utilize the energy from ATP to translocate lipids across cellular membranes. The human enzymes are entirely specific for glucosylceramide and not galactosylceramide, and indeed, the galactosyl- and glycosylceramide synthases have no significant sequence homology, indicating different evolutionary origins.
To enable protein interactions and signalling, both galactosyl- and glucosylceramide must be transported to and then across the plasma membrane. Some glucosylceramide is carried by lipoproteins (VLDL, LDL and HDL) in the circulation and presumably requires a transport mechanism for absorption and distribution across the membranes of target tissues.
In plants, glucosylceramides are formed by an evolutionarily conserved glucosylceramide synthase (GCS1) using UDP-glucose and ceramides in the endoplasmic reticulum, although an alternative mechanism has been described that utilizes sterol glucoside as the immediate glucose donor to ceramide. After transport through the Golgi, the final destination is the outer leaflet of the plasma membrane. There is evidence for a requirement for ceramides containing Δ4 trans-double bonds for synthesis of glucosylceramides but not for other sphingolipids in some plant and fungal tissues. During phosphate deficiency, the expression of glucosylceramide synthase is increased, while synthesis of glycosylinositol phosphoceramide is decreased.
In the yeast Pichia pastoris, there is one ceramide synthase, which produces ceramides of defined composition exclusively for the production of glucosylceramides (see our web page on long-chain bases), while a second ceramide synthase with different specificities produces the ceramide precursors for ceramide phosphorylinositol, which contains only phytosphingosine as the long-chain base. In some fungi, glucosylceramide synthases have been characterized (e.g., Hsx11 in Candida albicans), and the products are exported by vesicular trafficking pathways to the plasma membrane and cell wall, or they are secreted to the extracellular space in vesicles. A galactosylceramide synthase has yet to be identified. Enzymes responsible for the biosynthesis of glucuronosylceramide and α‑galactosylceramide in some bacteria have been characterized.
3. Function
Galactosylceramides: A remarkable property of cerebrosides is that their 'melting point' is well above physiological body temperature, so that glycolipids have a para-crystalline structure at this temperature. Each cerebroside molecule may form up to eight inter- or intramolecular hydrogen bonds by lateral interaction with neighbouring molecules between the polar hydrogens of the sugar and the hydroxy and amide groups of the sphingosine base of the ceramide moiety, and this dense network of hydrogen bonds contributes to the high transition temperature and the compact alignment of cerebrosides in membranes. As with sphingomyelin, monoglycosylceramides tend to be concentrated in the outer leaflet of the plasma membrane together with cholesterol and thence in myelin in the membrane domains termed 'rafts'. Indeed, the latter facilitate segregation to a greater extent than sphingomyelin via the combination of hydrogen bonds and hydrophobic interactions, and these forces are necessary for binding to the wide range of proteins, including enzymes and receptors, which are found in raft domains.
Galactosylceramide is essential to myelin structure and function, and it is involved in oligodendrocytes differentiation and the development of proper axo-glial interactions. While molecular species with 2’‑hydroxy fatty acid constituents are not necessary for myelin formation, they are critical for its long-term stability, presumably because increased hydrogen bonding with neighbouring lipids in membranes stabilizes the phase structure. Galactosylceramide is the precursor of 3’‑sulfo-galactosylceramide, which is required for brain development and for numerous purposes in other tissues. By interacting with sulfatide located in the membrane of opposing layers in the myelin sheath by carbohydrate-carbohydrate interaction, it forms what is known as a glycosynapse to provide a necessary contribution to the long-term stability of myelin.
Glucosylceramides: Glucosylceramides have similar physical properties in membranes to the galactose analogue, and they are also concentrated in raft domains in the outer leaflet of the plasma membrane. As mentioned briefly above, they are the primary precursor for most of the more complex oligoglycosphingolipids in animal tissues, especially in brain, where synthesis is vital for production of most neuronal oligoglycosphingolipids, while glucosylceramide per se is required for axonal growth. They are major constituents of skin lipids, where they are necessary for lamellar body formation in the stratum corneum and to maintain the water permeability barrier of the skin. In addition, the epidermal glucosylceramides (together with sphingomyelin) are the source of the unusual complex ceramides that are found in the stratum corneum including those with terminal hydroxyl groups and estolide-linked fatty acids. Some of the glucosylceramide in skin is linked covalently to proteins via terminal hydroxyl groups, presumably to strengthen the epidermal barrier.
 Much of the evidence for
the requirement for monoglycosylceramides in animals has been derived from cell lines in which synthesis has been suppressed by various
means in vitro.
Glucosylceramide is not critical for the viability of certain cell lines in culture, but disruption of the global synthase gene in mice results
in the death of embryos.
It is critical for the survival of cancer cells, and deletion from other cell types can lead to abnormalities.
As well as being an intermediate in the biosynthesis of more complex glycosphingolipids and its role in the permeability
barrier of the skin (discussed above), glucosylceramide is required for intracellular membrane transport, cell proliferation and survival,
and for various aspects of the immune system.
In contrast, there are indications that it may have adverse implications for various disease states.
Over-expression of glucosylceramide synthase in cancer cells has been linked to tumour progression with a reduction in ceramide
concentration that results in an increased resistance to chemotherapy, and the lipid has been associated with drug resistance in a wider context.
In the nematode Caenorhabditis elegans, glucosylceramide is the main glycosphingolipid and that containing the fatty acid 22:0 with an
iso-methyl-branched sphingoid base is reported to be as a critical mediator of the response to glucose and a longevity metabolite via
the membrane localization of clathrin, a protein that regulates membrane budding.
Much of the evidence for
the requirement for monoglycosylceramides in animals has been derived from cell lines in which synthesis has been suppressed by various
means in vitro.
Glucosylceramide is not critical for the viability of certain cell lines in culture, but disruption of the global synthase gene in mice results
in the death of embryos.
It is critical for the survival of cancer cells, and deletion from other cell types can lead to abnormalities.
As well as being an intermediate in the biosynthesis of more complex glycosphingolipids and its role in the permeability
barrier of the skin (discussed above), glucosylceramide is required for intracellular membrane transport, cell proliferation and survival,
and for various aspects of the immune system.
In contrast, there are indications that it may have adverse implications for various disease states.
Over-expression of glucosylceramide synthase in cancer cells has been linked to tumour progression with a reduction in ceramide
concentration that results in an increased resistance to chemotherapy, and the lipid has been associated with drug resistance in a wider context.
In the nematode Caenorhabditis elegans, glucosylceramide is the main glycosphingolipid and that containing the fatty acid 22:0 with an
iso-methyl-branched sphingoid base is reported to be as a critical mediator of the response to glucose and a longevity metabolite via
the membrane localization of clathrin, a protein that regulates membrane budding.
In Arabidopsis, glucosylceramides are critical for cell differentiation and organogenesis, but not necessarily for the viability of cells. It has been proposed that glycosphingolipids could impose positive curvature to membranes, thereby facilitating vesicle fusion. Glycosylceramides (but not glycosyldiacylglycerols) together with sterols are located in 'rafts' in plant membranes in an analogous manner to sphingolipids in animal tissues, and they are associated with specific proteins. There is evidence that glucosylceramide is necessary for the synthesis of cellulose, an abundant component of plant cell wall matrices. Correlative studies suggest that glucosylceramides help the plasma membrane in plants to withstand stresses brought about by cold and drought, and molecular species containing 2-hydroxy monounsaturated very-long-chain fatty acids and long-chain bases with 4-cis double bonds are present in higher concentrations in plants that are more tolerant of chilling and freezing. While fungal glucosylceramides with a 9-methyl group within the sphingosine backbone elicit defence responses in rice, cerebrosides with double bonds in positions 4 and/or 8 of the long-chain bases are utilized in the defence of some plants against fungal attack. A small amount only of glucosylceramide is utilized in plants as a precursor for oligoglycosylceramides, of which little appears to be known of the biosynthesis and properties.
The requirements for glucosylceramides in fungi are less studied, although they are certainly major constituents of the plasma membrane and cell wall, and they are involved in such processes as cell wall assembly, cell division and differentiation, and signalling. The presence of a methyl branch in the long-chain base is required for cell division and alkali-tolerance, and in the case of fungal pathogens, this feature is recognized by the host immune system and regulates virulence, often after the lipid is exported into the external environment as extracellular vesicles. In contrast to animals (and like plants), glucosylceramides are not precursors for oligoglycosylceramides in fungi. Some molecular species of this lipid from plants induce fruiting in the fungus Schizophyllum commune, and in this instance, a Δ8 double bond in the long-chain base is essential.
4. α-D-Galactosylceramide
 Cerebrosides linked to an α-D- rather
than a β-D-galactosyl unit such as that found in the marine sponge Agelas mauritianus, human gut microflora
and even in cow's milk are potent stimulators of mammalian immune systems by binding to the protein CD1d on the surface of antigen-presenting
cells and activating invariant natural killer T cells.
Indeed, this was one of the first pieces of evidence to show that glycolipids, like glycoproteins, could invoke an immune response.
Subsequently, it was demonstrated that α‑galactosylceramide with a 24:1 fatty acid, though present in very small amounts,
is loaded onto the CD1d or CD40 protein and is presented as the natural endogenous ligand for NKT cells in the thymus and the periphery.
Once activated, NKT cells proliferate and differentiate into producers of cytokines such as interleukin-10 (IL-10) to modulate the innate
and adaptive immune responses, with synthetic lipids containing straight-chain components more efficient than the branched molecules in
B. fragilis, which induce a different NKT phenotype.
The α‑glucosyl and α‑psychosine analogues are comparable in this respect.
Cerebrosides linked to an α-D- rather
than a β-D-galactosyl unit such as that found in the marine sponge Agelas mauritianus, human gut microflora
and even in cow's milk are potent stimulators of mammalian immune systems by binding to the protein CD1d on the surface of antigen-presenting
cells and activating invariant natural killer T cells.
Indeed, this was one of the first pieces of evidence to show that glycolipids, like glycoproteins, could invoke an immune response.
Subsequently, it was demonstrated that α‑galactosylceramide with a 24:1 fatty acid, though present in very small amounts,
is loaded onto the CD1d or CD40 protein and is presented as the natural endogenous ligand for NKT cells in the thymus and the periphery.
Once activated, NKT cells proliferate and differentiate into producers of cytokines such as interleukin-10 (IL-10) to modulate the innate
and adaptive immune responses, with synthetic lipids containing straight-chain components more efficient than the branched molecules in
B. fragilis, which induce a different NKT phenotype.
The α‑glucosyl and α‑psychosine analogues are comparable in this respect.
It is not certain whether α-galactosylceramide is synthesised in animal tissues, and it seems likely that is derived primarily from organisms in the gut microbiome such as Bacteroides fragilis and its relatives (although in general few bacteria produce sphingolipids). Ceramide-galactosyltransferases responsible for synthesis of this lipid in two species of bacteria from the intestinal microbiome have been identified, and in mouse gut, the main molecular form consists of a 2‑R‑hydroxylated hexadecanoyl chain linked to C18-sphinganine, while that in B. fragilis contains longer-chain components with iso-methyl-branches in the sphingoid base and often fatty acid moieties. With the latter, the sphinganine chain branching is a critical determinant of NKT cell activation. A decrease in production of this lipid was observed in mice exposed to stress conditions that alter the composition of the gut microbiota, including Western type diet, colitis and influenza A virus infection, with potential consequences upon the systemic immune responses.
Its concentration within animal tissues is controlled by catabolic enzymes in a two-step mechanism: removal of the acyl chain by an acid ceramidase followed by hydrolysis of the sugar residue by an α-glycosidase. From the initial studies with animal models, it is reported that treatment with α‑D‑galactosylceramides is effective against lung and colorectal cancers, melanomas and leukaemia, and pre-clinical trials of this lipid and synthetic analogues so far have shown that these are safe and efficacious as an anti-tumour immunotherapeutic agents and vaccine adjuvants against viruses and intracellular bacteria. Indeed, phase I/II trials of patients with high-risk melanoma or non-small cell lung cancer have given promising preliminary results. Synthetic analogues with anti-viral properties are also under development.
5. Catabolism of Glycosphingolipids
In animal tissues, the main sites for the degradation of all glycosphingolipids, including the monoglycosylceramides, oligoglycosphingolipids and gangliosides, are the lysosomes. These are membrane-bound organelles that comprise a limiting external membrane and internal lysosomal vesicles, which contain soluble digestive enzymes that work at the acidic pH of this organelle. All membrane components are transported to the lysosomes to be broken down into their various primary components. In the case of glycosphingolipids, this means to fatty acids, sphingoid bases and monosaccharides, which can be recovered for re-use or further degraded. Thus, sections of the plasma membrane enter the cell by a process of endocytosis, and they are then transported through the endosomal compartment to the lysosomes. The compositional and physical arrangement of the lysosomal membranes is such that they are themselves resistant to digestion with bis(monoacylglycero)phosphate (lysobisphosphatidic acid) as a characteristic component of the inner membrane. A glycocalyx of highly N-glycosylated integral membrane proteins protects the perimeter membrane with the aid of the ganglioside GM3, which is resistant to degradation. This glycocalix forms an efficient hydrophilic barrier at the luminal surface of the lysosomal perimeter membrane to protect it from degradation by proteases and hydrolases and to prevent lipids and their hydrolysis products from escaping from the lumen of the lysosome.
Degradation of oligoglycosylceramides and gangliosides occurs by sequential removal of monosaccharide units via the action of exohydrolases from the non-reducing end until a monoglycosylceramide unit is reached, when glucosylceramide β-glucosidases or an analogous β-galactosidase (one isoform) removes the final carbohydrate moiety. Several glucosylceramidases are known; GBA1 is a lysosomal hydrolase, GBA2 is a ubiquitous non-lysosomal enzyme and GBA3 is a cytosolic β-glucosidase. The last is found in the kidney, liver, spleen and a few other tissues of mammals, but its function is not clear.
As glycolipids with fewer than four carbohydrate residues are embedded in intralysosomal membranes, while the degradative enzymes are soluble, the process requires the presence of negatively charged lipids and activator proteins, which are water-soluble glycoproteins of low molecular weight. These are not catalysts themselves but are required as cofactors either by directing the enzyme to the substrate or by stimulating the enzyme by binding to it in some manner. Five such proteins are known, the GM2-activator protein (specific for gangliosides) and Sphingolipid Activator Proteins or saposins A, B, C and D, which perturb the membranes sufficiently to enable the degradative enzymes to reach the glycolipid substrates. The four saposins are derived by proteolytic processing from a single precursor protein, prosaposin, which is synthesised in the endoplasmic reticulum, transported to the Golgi for glycosylation and then to the lysosomes. Of these, saposin C is needed for the degradation of galactosyl- and glucosylceramide, while saposin B is required for hydrolysis of sulfatide, globotriaosylceramide and digalactosylceramide. The products of the hydrolysis reaction with monoglycosylceramides are ceramides and monosaccharides with net retention of the stereochemistry of the latter in the process.
 |
| Figure 3. Catabolism of glycosylceramides. |
The reactions are aided by the presence of anionic lipids such as bis(monoacylglycero)phosphate, which increases the ability of the GM2-activator to solubilize lipids and stimulates the hydrolysis of membrane-bound GM1, GM2 and some of the kidney sulfatides. Saposin D enables degradation of lysosomal ceramide to fatty acids and sphingoid bases by acid ceramidase and participates in the solubilization of negatively charged lipids at an appropriate pH. β-Glucosylceramidase and saposin C are required for the generation of the structural ceramides from glucosylceramide in the outer region of the skin, a key process to optimize the skin barrier and ensure survival.
Some glucosylceramide is hydrolysed by the enzyme GBA2 at the plasma membrane, where the ceramide formed is rapidly converted to sphingomyelin by the sphingomyelinase 2, which may be co-located with the glucosidase. Cellular β‑glucosidases are able to transfer the glucose moiety from glucosylceramide to and from other lipids as in the formation of cholesterol glucoside.
Small but significant amounts of glucosyl- and galactosylceramides are ingested as part of the human diet. They are not hydrolysed by pancreatic enzymes but are degraded in the brush border of the intestines by the enzyme lactase-phlorizin hydrolase (which also hydrolyses the lactose in milk) to ceramides and thence to sphingosine (see our web page on sphingomyelin).
Plants: An Arabidopsis homologue of human glucosylceramidase (AtGCD3) preferentially hydrolyses glucosylceramides that contain long acyl chains in vitro, and three further isoforms exist based on sequence homology. Under osmotic stress, these enzymes operate at the plasma membrane to generate ceramides, which are utilized for synthesis of glycosylinositol phosphoceramides to stabilize the membrane. A glucocerebrosidase designated Os3BGlu6 catalyses the hydrolysis of glucosylceramide to ceramide in rice.
6. Genetic Disorders and Disease
Harmful quantities of glucosylceramide accumulate in the spleen, liver, lungs and bone marrow, and in the brain of patients with the rare Gaucher disease, the most common of the inherited metabolic disorders (autosomal recessive) with storage of excessive amounts of complex sphingolipids. Three main clinical forms (phenotypes) of the disease are recognized of which by far the most dangerous are those affecting the brain (Types 2 and 3). All patients exhibit a deficiency in the lysosomal glucosylceramide-β‑glucosidase (GBA1), which catalyses the first step in the catabolism of glucosylceramide. The enzyme may be present, but a mutation prevents it assuming its correct conformation, although there must be other factors as patients with a defective saposin C, the lysosomal activator protein, develop similar symptoms.
In brain, glucosylceramide accumulates when complex lipids turn over during brain development and during the formation of the myelin sheath of nerves. Other than brain, excess glucosylceramide arises mainly from the biodegradation of old red and white blood cells. The result is that the glucosylceramide remains stored within the lysosomes of macrophages, i.e., the specialized cells that remove worn-out cells by degrading them to simple molecules for recycling, thus preventing them from working normally and often leading to chronic inflammation. Enlarged macrophages containing undigested glucosylceramide are termed Gaucher cells, which over express and secrete certain proteins into the circulation that can be used as biomarkers. Further, glucosylceramide is converted more rapidly to gangliosides in these cells, leading to an increase in ganglioside GM3 in plasma and spleen of patients with Gaucher disease. Fortunately, there are now enzyme replacement therapies for patients with the milder (non-neurological or Type 1) form of Gaucher disease that successfully reverse most manifestations of the disorder, including decreasing liver and spleen size and reducing skeletal abnormalities. Two oral drugs that inhibit glucosylceramide synthesis have been approved. Galactosylceramide is essential for the maintenance of proper structure and stability of myelin and differentiation of oligodendrocytes, and disruption of its synthesis and metabolism may be relevant to neurological disorders.
Oligoglycosylceramides and gangliosides especially are known to be involved in the pathology of several cancers, and glucosylceramide is the precursor of these, but it is a bioactive lipid in its own right in that it modulates membrane microdomains and affects intracellular trafficking and cell signalling to oppose ceramide-induced apoptosis and promote cellular survival. Aberrant upregulation of glucosylceramide synthase drives tumour growth, metastasis and multidrug resistance in humans, while inhibition of this enzyme leads to a marked arrest of cell growth in some cancer cells in vitro; this may have potential for treatment of colorectal and other cancers. Those molecular species linked to 16:0, 18:0, 24:1 fatty acids have greater oncogenic potential than others, depending upon tissue. Further, a defective glucocerebrosidase GBA1 enzyme in humans has been implicated in an increased risk of multiple myeloma and other cancers.
A deficiency in the latter enzyme may predispose individuals to disorders such as Lewy body dementia and Parkinson's disease, although there seems to be no unequivocal evidence that glucosylceramide accumulates in the brains of these patients. Excess glucosylceramide production and thence of more complex glycosphingolipids is a factor in polycystic kidney disease. It appears to be a general rule that the mere process of lysosomal substrate accumulation in all lysosomal storage disorders impairs lysosome integrity and results in more general disruptions to lipid metabolism and membrane structure and function. On the other hand, inhibition of glucosylceramidases may be of benefit in cystic fibrosis. Krabbe disease is discussed in the next section.
Galactosylceramide is the initial receptor for the human immunodeficiency virus (HIV) in mucosal epithelial cells, and it controls the early infection-independent phase of HIV transfer to T cells. Glucosylceramide levels regulate the uptake of viruses that rely upon the late endosomal compartment for fusion, including the influenza A and Ebola viruses, while glucosylceramide in bunyavirus particles is essential for virus binding to host cells.
7. Galactosylsphingosine (Psychosine) and Glucosylsphingosine
Psychosine is the trivial name for galactosylsphingosine, the non-acylated or lyso form of galactosylceramide, sometimes termed lyso-galactosylceramide, which is unusual in being a basic (cationic) lipid with binding properties that differ from those of other sphingolipids. It is present in bovine but not normal human brain at very low concentrations, and it is a minor intermediate in the catabolism of monoglycosylceramides. Some psychosine is synthesised, together with galactosylceramide, by the action of UDP-galactose:ceramide galactosyltransferase on sphingosine in the oligodendrocytes, but most is generated catabolically through the deacylation of galactosylceramide by acid ceramidase (ACDase). As receptors have been identified in animal cells, it may be a factor in pathophysiology or in signalling, and in sensory neurons of skin, it causes itching (pruritus) by activation of 5-HT2A, TRPA1 or TRPV4 receptors.

Psychosine accumulates in brain in the genetic lysosomal storage disorder Krabbe disease (globoid cell leukodystrophy), leading to widespread degeneration of oligodendrocytes and then to demyelination. Genetic ablation of ACDase in the mouse model of this disease eliminates psychosine accumulation and is reported to be a cure; selective inhibitors of the enzyme are now being sought. It inhibits cytokinesis, i.e., the last stage in the process by which a single cell divides to produce two daughter cells, with production of multinucleate cells instead. While it can inhibit protein kinase C, psychosine may exert its effects primarily through perturbation of membranes, especially of raft domains.
In the same way, excess glucosylceramide formed in a deficiency of lysosomal glucosylceramide-β‑glucosidase (GBA1) is acted upon by the lysosomal enzyme acid ceramidase to produce glucosylsphingosine, which is cytotoxic and can exert pathological effects by stimulating the release of Ca2+ from the endoplasmic reticulum in the brain. It accumulates with related molecules in severe forms of Gaucher disease (discussed above), and plasma levels correlate strongly with the disease burden and response to treatment. Studies in vitro suggest that it exerts ill effects on cellular energy metabolism by inhibiting the TCA cycle, ATP production, mitochondrial function, glycolysis and protein ubiquitination. Some new-born babies with a genetic defect in which this lipid accumulates do not have an effective water barrier in the epidermis and die shortly after birth. It can accumulate in Niemann Pick type C disease, caused by deficient intracellular cholesterol transporter proteins, and it is present in elevated concentrations in glaucoma. Glucosylsphingosine induces modifications to melanoma cells that could contribute to cancer development in patients with sphingolipidoses.
O-Acyl and plasmalogen forms of psychosine with hexadecanal or octadecanal linked to the carbohydrate moiety through 4,6- or 3,4‑cyclic acetal bonds, termed 'plasmalopsychosines', have been detected in brain tissues of some animals, including humans (see the structure of the fully acylated plasmalo-galactosylceramide in Part 1 above). They are not cytotoxic, but 4,6‑plasmalopsychosine displays distinctive neurological effects. Although two stereoisomers can exist in theory, only the endo form is known to occur naturally. An analogous glycero-plasmalopsychosine has been characterized from brain tissue.
8. Phosphoglycoceramides
Glycosphingolipids in which the carbohydrate moiety is phosphorylated have been described, i.e., where the ceramide is linked directly to carbohydrate moieties and only then to phosphate, i.e., comparable to some diacylglycerol-linked phosphoglycolipids. As examples, cholinephosphoryl–6-Gal-(β1–1)-Cer and cholinephosphoryl–6-Gal-(β1–6)-Gal-(β1–1)-Cer have been isolated and characterized from the earthworm, Pheretima hilgendorfi. In this instance, the main fatty acids are 22:0 and 24:0, and the sphingoid bases are octadeca- and nonadeca-4-sphingenine. Subsequently, related triglycosylsphingophospholipids with either a terminal mannose or galactose unit linked to phosphorylcholine were found in the same species, while a lipid akin to that illustrated was found in a clam worm, Marphysa sanguinea. Analogous phosphonolipids, i.e., with a carbon-phosphorus bond, have been found in some marine invertebrates.
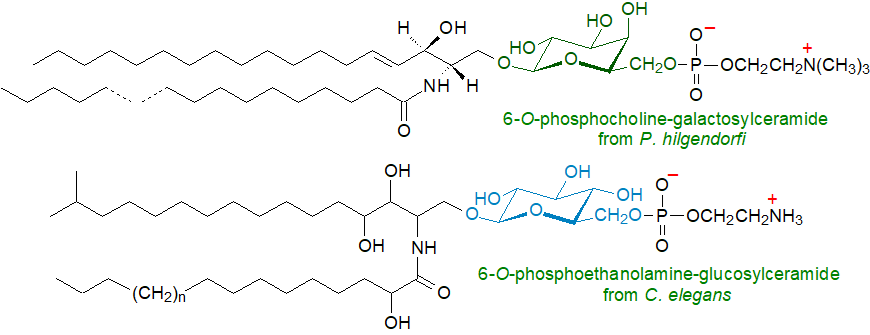 |
| Figure 4. Two representative phosphoglycoceramides. |
The model nematode C. elegans contains a novel glucosylceramide with phosphoethanolamine or its monomethylated form attached to carbon 6 of the glucose moiety. The ceramide moiety contained an iso-branched C17 sphingoid base of the phytosphinganine type (i.e., with a 4-hydroxyl group) and amide-linked 2‑hydroxy long-chain fatty acids with variable chain lengths (C22, C23 and C24). This lipid is vital for the development of C. elegans through its regulation of sterol mobilization (the organism requires an exogenous source of cholesterol). It can rescue larval arrest that has been induced by sterol starvation.
9. Analysis
High-resolution thin-layer chromatography and high-performance liquid chromatography (HPLC) are well established for the separation and analysis of the two types of monoglycosylceramides, although these methods may not be suitable for high-throughput lipidomics where data are often reported for a combined monohexoside class. Given the very different biological properties of the two lipids, this can be a severe limitation in the methodology. While separation of α-D-galactosylceramides from the β-form is more of an analytical challenge, it has been accomplished by HPLC. HPLC in the reversed-phase mode was for many years the standard method for separation of molecular species, often after benzoylation for sensitive UV detection, but modern mass spectrometric methods are now being used increasingly for characterization purposes. O‑Acylated forms may be missed if an alkaline purification step is included in analytical protocols.
Recommended Reading
- Aerts, J.M.F.G., Kuo, C.L., Lelieveld, L.T., Boer, D.E.C., van der Lienden, M.J.C., Overkleeft, H.S. and Artola, M. Glycosphingolipids and lysosomal storage disorders as illustrated by Gaucher disease. Curr. Opinion Chem. Biol., 53, 204-215 (2019); DOI.
- Birkholz, A.M., Howell, A.R. and Kronenberg, M. The alpha and omega of galactosylceramides in T cell immune function. J. Biol. Chem., 290, 15365-15370 (2015); DOI.
- Breiden, B. and Sandhoff, K. The role of sphingolipid metabolism in cutaneous permeability barrier formation. Biochim. Biophys. Acta, Lipids, 1841, 441-452 (2014); DOI.
- Cameron, G., Nguyen, T., Ciula, M., Williams, S.J. and Godfrey, D.I. Glycolipids from the gut symbiont Bacteroides fragilis are agonists for natural killer T cells and induce their regulatory differentiation. Chem. Sci.., 14, 7887-7896 (2023); DOI.
- Castillo-Ribelles, L. and others. Evaluation of a liquid chromatography-tandem mass spectrometry method for the analysis of glucosylceramide and galactosylceramide isoforms in cerebrospinal fluid of Parkinson's disease patients. Anal. Chem., 96, 12875-12882 (2024); DOI.
- Dubot, P., Astudillo, L., Therville, N., Carrié, L., Pettazzoni, M., Cheillan, D., Stirnemann, J., Levade, T., Andrieu-Abadie, N. and Sabourdy, F. Potential role of sphingolipidoses-associated lysosphingolipids in cancer. Cancers, 14, 48588 (2022); DOI.
- Fernandes, C.M., Goldman, G.H., and Del Poeta, M. Biological roles played by sphingolipids in dimorphic and filamentous fungi. mBio, 9, e00642-18 (2018); DOI.
- Merrill, A.H. Sphingolipids. In: Biochemistry of Lipids, Lipoproteins and Membranes (5th Edition). pp. 363-397 (Vance, D.E. and Vance, J. (editors), Elsevier, Amsterdam) (2008) - see Science Direct.
- Merrill, A.H. Don't be surprised when these surprise you: some infrequently studied sphingoid bases, metabolites, and factors that should be kept in mind during sphingolipidomic studies. Int. J. Mol. Sci., 26, 650 (2025); DOI.
- Michaelson, L.V., Napier, J.A., Molino, D. and Faure, J.-D. Plant sphingolipids: Their importance in cellular organization and adaption. Biochim. Biophys. Acta, Lipids, 1861, 1329-1335 (2016); DOI.
- Reza, S., Ugorski, M. and Suchanski, J. Glucosylceramide and galactosylceramide, small glycosphingolipids with significant impact on health and disease. Glycobiology, 31, 1416-1434 (2022); DOI.
- Yamaji, T. and Hanada, K. Sphingolipid metabolism and interorganellar transport: localization of sphingolipid enzymes and lipid transfer proteins. Traffic, 16, 101-122 (2015); DOI.
- Zhao, X.H. and Pandey, M.K. Central roles of glucosylceramide in driving cancer pathogenesis. Int. J. Mol. Sci., 26, 9879 (2025); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: December 2025 | ||
© The LipidWeb is open access and fair use is encouraged - but not text and data mining, AI training, and similar technologies (text scraping).
