Bis(monoacylglycero)phosphate
Bis(monoacylglycero)phosphate ('BMP') was first isolated from pig lung in 1967 and is now known to be a common if minor constituent of all animal tissues, but mainly in the lysosomal compartment of cells and in exosomes. It was first incorrectly termed ‘lysobisphosphatidic acid’, although it is only superficially related to phosphatidic acid per se and might better be considered a structural analogue of phosphatidylglycerol. It has a unique stereochemistry, which makes this lipid of special interest, but only now are key many aspects of its structure, biosynthesis and function being revealed. It is not found in plants, and its occurrence in bacteria is problematic.
1. Structure and Occurrence
The stereochemical configuration of bis(monoacylglycero)phosphate differs from that of all other animal glycero-phospholipids in that the phosphodiester moiety is linked only to positions sn-1 and sn-1′ of glycerol, rather than to positions sn-3 and sn-3′, while the fatty acids may be linked to the sn-3/3'-positions.
 |
| Figure 1. Structural comparison of bis(monacylglycero)phosphate (3,3'- and 2,2'-forms) and phosphatidylglycerol |
There is doubt in the literature as to the positional distributions of the fatty acids on the glycerol moieties, and there is an influential school of thought to the effect that the fatty acids are esterified to position sn-2 and sn-2′ in the native molecule, as has been determined by 1H NMR spectroscopy with chiral shift reagents, but biosynthetic studies have not yet confirmed whether this is the case (see below). S,S-(2,2′-diacyl)-BMP forms a more tightly packed structure in membranes, but this is less stable thermodynamically than S,S-(3,3′-diacyl)-BMP.
BMP is usually a rather minor component of animal tissues (~1-2%), but it is highly enriched in the lysosomal membranes of liver and other tissues, where it can amount to 15% or more of the phospholipids, and it is now recognized as a marker for this organelle. Cellular constituents, including excess nutrients, growth factors and foreign antigens, are captured at the cell surface by receptors, such as the mannose-6-phosphate receptor, for uptake and delivery to an intermediate heterogeneous set of organelles known as endosomes, which act as a sorting station where the receptors are recycled before the hydrolases and other materials are directed to the lysosomes, the digestive organelles of the cell enriched in hydrolytic enzymes at an acidic pH (4.6 to 5). There, the hydrolases are activated, and the unwanted materials are digested and others are recycled. It is the internal membranes of mature or ‘late’ endosomes and lysosomes that contain bis(monacylglycero)phosphate, where it amounts to as much as 70% of the phospholipids or 11% of the phospholipids of some macrophage/microglial cell lines, reflecting the increased endo-lysosomal capacity of these cells.
Whatever the positions of the fatty acids on the glycerol molecule, their compositions can be distinctive with 18:1(n-9) and 18:2(n-6) and/or 20:4 and 22:6(n‑3) being abundant, although this is highly dependent on the tissue, cell type or organelle (see Table 1). For example, the testis lipid contains more than 70% 22:5(n‑6), and to my knowledge, no other natural lipid contains so much of this fatty acid. Lung alveolar macrophages contain BMP with mainly C18 fatty acids, and baby hamster kidney (BHK) fibroblast cells are very different in that their BMP has more than 80% of oleate. In contrast, the metabolically important lipid isolated from rat liver lysosomes specifically contains almost 70% 22:6(n-3). Such unusual compositions must confer distinctive properties in membranes and suggest quite special functions, most of which have yet to be revealed, but it would be of value to see new data from the improved separation technologies now available (published molecular species data cannot easily be tabulated for reproduction here). In plasma, the lipid is found at trace levels only where it is associated both with the lipoprotein fractions (40%) and the lipoprotein-deficient compartment (60%). A further finding of general interest is that this lipid alone of more than 1200 studied accumulated in the tissues of mice and humans during aging.
Table 1. Fatty acid composition (wt% of the total) of bis(monoacylglycero)phosphate from various tissues. |
||||||
| Fatty acid | Rat liver lysosomes | Human liver | THP-1 macrophages | Rat uterine stromal cells | Rat testis | BHK cells |
|---|---|---|---|---|---|---|
| 16:0 | 3 | 6 | 23 | 6 | 5 | 4 |
| 18:0 | 1 | 5 | 24 | 3 | 3 | trace |
| 18:1 | 5 | 57 | 27 | 30 | 5 | 83 |
| 18:2 | 6 | 10 | 5 | 2 | 1 | 6 |
| 20:4 | 6 | 4 | 7 | |||
| 22:4 | 6 | 5 | ||||
| 22:5(n-6) | } 4 | trace | 70 | |||
| 22:5(n-3) | trace | |||||
| 22:6(n-3) | 69 | 9 | 7 | 36 | 5 | |
| Reference | 1 | 1 | 2 | 3 | 3 | 4 |
| 1, Wherrett, J.R. and Huterer, S. Lipids, 8, 531-533 (1973);
DOI.
2, Besson, N. et al. Lipids, 41, 189-196 (2006);
DOI.
3, Luquain, C. et al., Biochem. J., 351, 795-804 (2000);
DOI.
4, Brotherus, J. and Renkonen, O. Chem. Phys. Lipids, 13, 11-20 (1974);
DOI. Molecular species data have been published, see - DOI1, DOI2 and DOI3. |
||||||
This lipid may not be uniquely of animal origin, as the plant bacterial-pathogen Agrobacterium tumefaciens can take up lyso-phosphatidylglycerol and convert it to two distinct isoforms of BMP, and it has been reported from some alkalophilic strains of Bacillus sp., although it is not known whether any of these have the distinctive stereochemistry of the animal equivalent.
2. Biochemistry and Function
Biosynthesis:Many questions remain to be answered questions on the biosynthesis of bis(monacylglycero)phosphate, and there is no doubt that the last word has yet to be written on the subject.
One recent proposal suggests that bis(monacylglycero)phosphate is synthesised from phosphatidylglycerol via lysophosphatidylglycerol (LPG) in the endosomal system. In the first rate-limiting step, lysosomal phospholipase A2 removes the fatty acid from position sn-2 of phosphatidylglycerol, before in the second step, the lysophosphatidylglycerol produced is acylated on the sn-3 position of the head group glycerol moiety by means of an energy-independent transacylase reaction from a fatty acid donor catalysed mainly by a protein designated CLN5 to generate R,S-bis(monacylglycero)phosphate. Lysosomal lipases remove the fatty acid from position sn-1 of the glycerol from the starting phosphatidylglycerol moiety, before this is exchanged for a 1-monoacylglycerol in a reaction catalysed by phospholipase D (PLD3/4) to produce S,S-bis(monacylglycero)phosphate, i.e., with stereo-inversion. Studies with CNL5-knockout cells show a deficiency of BMP and a substantial accumulation of lysophosphatidylglycerol (LPG) in the lysosomes. It is noteworthy that both of the original fatty acids are exchange during this process.
 |
| Figure 2. Proposed mechanism for biosynthesis of bis(monacylglycero)phosphate. |
An alternative proposal is that R,S-lysophosphatidylglycerol is first acylated to generate (R,S-BMP) by an enzyme CLN8 in the endoplasmic reticulum, during which stereo-inversion occurs via a cyclic intermediate to form S,S-lysophosphatidylglycerol. This is then converted to S,S-BMP by CLN5, or an unidentified transacylation reaction occurs following stereoinversion to generate S,S-BMP directly. Presumably, the acylation reactions are responsible for the distinctive fatty acid composition of BMP. Cells in which CLN8 is deleted lack BMP.
 |
| Figure 3. Alterative proposed mechanism for biosynthesis of bis(monacylglycero)phosphate. |
A more recent proposal is that the reaction proceeds via a deacylated intermediate after stereoinversion has occurred. However, the primary source of phosphatidylglycerol and thence lysophosphatidylglycerol has yet to be determined, but possibly mitochondria via crosstalk with the endoplasmic reticulum, nor is the origin of the monoacylglycerol required in the first proposed mechanism known. The intermediate BMP with the sn-3:sn-1′ configuration has been isolated from BHK and rat uterine stromal cells. While other biosynthetic routes may be possible, cardiolipin has been ruled out as a potential precursor.
While the biosynthetic studies confirm the stereochemistry of the glycerol moieties, they provide no information on the positional distributions of the fatty acid components, i.e. 3,3' as illustrated in Figures 2/3 (following the relevant publications) or as many believe the 2,2'-positions. Alix(adaptor protein)-dependent multi-vesicular liposomal membranes are formed in the presence of S,S-(2,2′-diacyl)-BMP and not S,S-(3,3′-diacyl)-BMP, and there is a report that the 2,2′-dioleoyl form rather than the more stable sn‑3,3′‑isomer is essential for the function of BMP in cholesterol metabolism in lysosomes. The 3/3'-form is more rapidly degraded by lipases than 2,2'-BMP. In the presence of acid (slow) or base (more rapid) in vitro, 2,2'-diacyl-BMP spontaneously isomerizes to the thermodynamically more stable 3,3'-diacyl-BMP via a 2,3'-intermediate, possibly as part of a regulatory system in which four factors, proteins, metal ions, nucleotides and pH are involved, with the last of these most important in vivo. Isomerization may be the first step in catabolism of BMP by phospholipase A2.
 |
| Figure 4. Isomerization of 2,2- to 3,3'-bis(monacylglycero)phosphate under acidic or alkaline conditions. |
Physical and chemical properties: The properties of bis(monacylglycero)phosphate in membranes will be highly dependent on fatty acid composition, but the function in lysosomes is of particular interest and is under investigation. It certainly has a structural role in developing the complex intraluminal membrane system, aided by a tendency not to form a bilayer. Like cardiolipin, it is a cone-shaped molecule with a small but hydrated head group, which is negatively charged, and it encourages fusion of membranes or formation of internal vesicles (invagination) at the pH in the endosomes where it may associate with certain proteins, which carry a positive charge under the acidic conditions, including hydrolases and coactivators.
Its unique stereochemistry means that BMP is resistant to most phospholipases, and this may hinder or prevent self-digestion of the lysosomal membranes. Although the fatty acid constituents may turn over rapidly by transacylation, the glycerophosphate backbone is stable, and it is not touched by the main phospholipases that hydrolyse more common phospholipids such as phosphatidylcholine and phosphatidylethanolamine. However, some phospholipases have been tentatively identified that may be involved in catabolism under acidic conditions and other local environmental factors, although the control mechanisms are not known (see below), and it has been claimed that this has not been rigorously tested with different BMP positional and stereo-isomers. Further, the unique stereochemistry of BMP may help it evade immunorecognition by endogenous antibodies.
Various extracellular membrane-bound vesicles secreted by most living cells and are found in biological fluids that act as mediators of cell-cell communication and have roles in patho-physiological processes. Small vesicles of this kind (<200 nm), usually termed 'exosomes', are of endosomal origin with BMP as an identifying lipid marker and probably a participant in their formation. For example, exosomes shed from reticulocytes are nano-vesicles that carry a cargo of lipids and other materials into the circulation, and they can be distinguished from secreted microvesicles of a different origin by their content of BMP. Studies in vitro suggest that 2,2′-BMP, possibly formed by isomerization, is the preferred isomer for this purpose. The 22:6-22:6 species is reportedly a biomarker for phagocytizing macrophages/microglia cells during cerebral ischaemia and of urinary exosomes as a consequence of drug-induced endolysosomal dysfunction.
Lysosomal metabolism: Bis(monoacylglycero)phosphate is negatively charged at lysosomal pH and can form a stable docking station in the endosomal membranes or membrane vesicles for luminal acid hydrolases that are positively charged at acidic pH and require a water-lipid interface for activation. By binding in this way, BMP stimulates lysosomal lipid-degrading enzymes, including acid sphingomyelinase, acid ceramidase, acid phospholipase A2, and an acid lipase with the capacity to hydrolyse triacylglycerols and cholesterol esters, while by binding to the heat-shock protein Hsp70, which promotes survival of stressed cells by inhibiting lysosomal membrane permeabilization on the inner lysosomal membrane, it activates the acid sphingomyelinase for stabilization of lysosomes. Thus, BMP plays a central role in determining the fate of the lysosomal content by stimulating degradation and sorting of lipids.
 The
endosomal membranes are a continuation of the lysosomal membranes, and they perform the same task in sorting and recycling material back to
the plasma membrane and endoplasmic reticulum, and in particular, they are an important element of
cholesterol homeostasis.
Thus, low-density lipoproteins (LDL) internalized in the liver reach the late endosomes where the constituent cholesterol esters are
hydrolysed by an acidic cholesterol ester hydrolase.
The characteristic network of BMP-rich membranes contained within multivesicular late endosomes regulates cholesterol
transport by acting as a collection and re-distribution point for the free cholesterol generated in this way via intralumenal vesicles
membranes before being redistributed to the endosomal/lysosomal limiting membrane and then to the rest of the cell.
The process is under the control of Alix/AlP1, a cytosolic protein that interacts specifically with this lipid and takes part in sorting
into the multivesicular endosomes.
In macrophages such as those in foam cells, BMP is likewise involved in the regulation of intracellular cholesterol
traffic where it is reported to have a protective effect by inhibiting the production of pro-apoptotic oxysterols, while in alveolar
macrophages, it has a dynamic role in the provision of arachidonate for eicosanoid production.
The
endosomal membranes are a continuation of the lysosomal membranes, and they perform the same task in sorting and recycling material back to
the plasma membrane and endoplasmic reticulum, and in particular, they are an important element of
cholesterol homeostasis.
Thus, low-density lipoproteins (LDL) internalized in the liver reach the late endosomes where the constituent cholesterol esters are
hydrolysed by an acidic cholesterol ester hydrolase.
The characteristic network of BMP-rich membranes contained within multivesicular late endosomes regulates cholesterol
transport by acting as a collection and re-distribution point for the free cholesterol generated in this way via intralumenal vesicles
membranes before being redistributed to the endosomal/lysosomal limiting membrane and then to the rest of the cell.
The process is under the control of Alix/AlP1, a cytosolic protein that interacts specifically with this lipid and takes part in sorting
into the multivesicular endosomes.
In macrophages such as those in foam cells, BMP is likewise involved in the regulation of intracellular cholesterol
traffic where it is reported to have a protective effect by inhibiting the production of pro-apoptotic oxysterols, while in alveolar
macrophages, it has a dynamic role in the provision of arachidonate for eicosanoid production.
In animal models, it has been demonstrated that different tissues have characteristic BMP profiles that adapt to the nutritional and metabolic state, especially in hepatocytes, brown adipocytes and pancreatic cells, suggesting that this lipid has a role in how these adapt to nutrient availability and ambient temperatures. During cold exposure, there is increased synthesis of BMP in liver lysosomes, by a mechanism regulated by the lysosomal transcriptional regulator transcription factor EB (TFEB) and phospholipase A2 group XV.
Disease: In consequence of its role in lysosomes, it has become evident that bis(monacylglycero)phosphate is involved in the pathology of lysosomal storage diseases such as Niemann-Pick C disease, where cholesterol accumulation is a distinctive feature. Similarly, high levels of BMP enriched in docosahexaenoic acid are found in the retinal pigment epithelium in Stargardt disease, which is characterized by juvenile onset retinal degeneration, and they are presumed to be a consequence of late endosomal/lysosomal dysfunction. It also accumulates as a secondary storage material in the brain of a broad range of mammals with gangliosidoses. In these circumstances, its concentration can increase substantially, probably as a secondary event, and its composition may change to favour molecular species that contain less of the polyunsaturated components. Mutations of CLN5 or of the lysophosphatidylglycerol acyltransferase CLN8, which take part in the biosynthesis of BMP, cause the fatal neurodegenerative Batten disease. Reduced levels of BMP are associated with depression of the lysosomal enzyme GCase and cause an accumulation of glucosylsphingosine, a factor in frontotemporal dementia and other neurological diseases. Dysregulation of BMP metabolism and thence of cholesterol homeostasis may be relevant to atherosclerosis.
Some BMP isomers are antigens recognized by autoimmune sera from patients with a rare and poorly understood disease known as antiphospholipid syndrome (phospholipidosis) so may be a factor in the pathological basis of this illness, which is exacerbated by cationic amphiphilic drugs. In general in such diseases, BMP levels are elevated in the circulation and its fatty acid composition changes, so that this can be used as a diagnostic biomarker that enables a clear distinction between lipid overload and drug-induced lysosomal storage diseases. An elevated concentration of di-docosahexaenoyl BMP in urine is a biomarker of drug-induced phospholipidosis, and it may be a marker for metastatic cancers of macrophage origin. It has been suggested that high levels of this molecule in breast cancer scavenge reactive oxygen species in lysosomes to protect them from oxidant-induced lysosomal membrane permeabilization.
Non-enveloped viruses of the family Reoviridae, which include mammalian pathogens, enter cells without the aid of a limiting membrane and thus cannot fuse with host cell membranes. The bluetongue virus was the first to be shown to use BMP in late endosomes and endolysosomes for membrane penetration and entry into host cells, and it has since been established that this mechanism may influence the infectivity of many other viruses, including COVID-19, influenza and Lassa virus, by enabling them to hijack the endosomal machinery leading to fusion of viral and endosomal membranes and release of viral RNA into the cytosol. Modulation of BMP levels in lysosome-associated diseases by pharmacological means may have therapeutic potential.
Catabolism: Although bis(monoacylglycero)phosphate is resistant to hydrolysis by many of the common phospholipases because of its unique stereochemistry. it is now known to be hydrolysed with high specificity in liver by a hydrolase designated α/β-hydrolase domain-containing 6 or ABHD6, once thought to be mainly a monoacylglycerol lipase capable of degrading the endocannabinoid 2‑arachidonoylglycerol. There is a related enzyme designated ABHD12 in brain, while some hydrolysis has been observed in vitro at least by the lysosomal acid sphingomyelinase and by lysosomal phospholipase A2, especially following isomerization of BMP as it reacts more rapidly with 3,3′-diacyl-BMP than with 2,2′-diacyl-BMP. The latter was originally characterized a 1-O-acylceramide synthase and is a lysosomal and late endosomal enzyme that maintains general glycerophospholipid homeostasis in cells.
3. Related Lipids
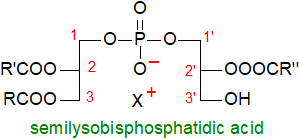 'Semilysobisphosphatidic acid' (hemi-BMP), i.e., with three moles
of fatty acid per mole of lipid, is occasionally found in tissues, including plasma, but its stereochemistry has not been determined.
It is sometimes found in the Golgi membranes, where the relative amount varies in different regions,
but can attain as much as 15% of the total phospholipids in those compartments that are most active metabolically.
It would not be at all surprising if this lipid were found to have a distinctive role in the Golgi complex,
but this is a matter of speculation.
'Semilysobisphosphatidic acid' (hemi-BMP), i.e., with three moles
of fatty acid per mole of lipid, is occasionally found in tissues, including plasma, but its stereochemistry has not been determined.
It is sometimes found in the Golgi membranes, where the relative amount varies in different regions,
but can attain as much as 15% of the total phospholipids in those compartments that are most active metabolically.
It would not be at all surprising if this lipid were found to have a distinctive role in the Golgi complex,
but this is a matter of speculation.
The fully acylated lipid, bis(diacylglycero)phosphate or 'bisphosphatidic acid', has been found in lysosomes from cultured hamster fibroblasts (BHK21 cells). In addition, it has been detected in bacteria, where it presumably has a different stereochemistry because of the mechanism of its synthesis from phosphatidylglycerol.
Although Archaeal glycerolipids have the phosphate moiety linked to position sn-1 of the glycerol moiety, the biosynthesis and metabolism of these lipids are entirely different from those of bis(monoacylglycerol)phosphate.
4. Analysis
Bis(monoacylglycero)phosphate is easily misidentified as phosphatidic acid in many chromatographic systems. Modern mass spectrometric methods can be used for analysis by using differential fragmentation of sodiated ions for identification and quantification, but many prefer to use liquid chromatography/mass spectrometry to ensure separation from phosphatidylglycerol with which it is isobaric. Positional isomers have been separated by a thin-layer chromatography procedure. If required, the two lipids can be differentiate by vigorous acetolysis; phosphatidylglycerol yields monoacetyldiacylglycerol and triacetylglycerol, while BMP yields diacetylmonoacylglycerol.
Recommended Reading
- Abe, A., Hinkovska-Galcheva, V., Bouchev, P., Bouley, R. and Shayman, J.A. The role of lysosomal phospholipase A2 in the catabolism of bis(monoacylglycerol)phosphate and association with phospholipidosis. J. Lipid Res., 65, 100574 (2024); DOI.
- Abe, A., Hinkovska-Galcheva, V., Verma, R. and Shayman, J.A. Isomerization of bis(monoacylglycero)phosphate by acyl migration. J. Lipid Res., 66, 100789 (2025); DOI.
- Akgoc, Z., Sena-Esteves, M., Martin, D.R., Han, X., d'Azzo, A. and Seyfried, T.N. Bis(monoacylglycero)phosphate: a secondary storage lipid in the gangliosidoses. J. Lipid Res., 56, 1006-1013 (2015); DOI.
- Bulfon, D. and others. Functionally overlapping intra- and extralysosomal pathways promote bis(monoacylglycero)phosphate synthesis in mammalian cells. Nature Commun., 15, 9937 (2024); DOI.
- Czolkoss, S., Borgert, P., Poppenga, T., Holzl, G., Aktas, M. and Narberhaus, F. Synthesis of the unusual lipid bis(monoacylglycero)phosphate in environmental bacteria. Environm. Microbiol., 23, 6993-7008 (2021); DOI.
- Goursot, A. Mineva, T., Bissig, C., Gruenberg, J. and Salahub, D.R. Structure, dynamics, and energetics of lysobisphosphatidic acid (LBPA) isomers. J. Phys. Chem. B, 114, 15712-15720 (2010); DOI.
- Grabner, G.F, and 12 others. Metabolic regulation of the lysosomal cofactor bis(monoacylglycero)phosphate in mice. J. Lipid Res., 61, 995-1003 (2020); DOI.
- Gruenberg, J. Life in the lumen: The multivesicular endosome. Traffic., 21, 76-93 (2020); DOI.
- Hullin-Matsuda, F., Colosetti, P., Rabia, M., Luquain-Costaz, C. and Delton, I. Exosomal lipids from membrane organization to biomarkers: Focus on an endolysosomal-specific lipid. Biochimie, 203, 77-92 (2022); DOI.
- Janssens, G.E and others. A conserved complex lipid signature marks human muscle aging and responds to short-term exercise. Nature Aging, 4, 681-693 (2024); DOI.
- Luquain, C., Dolmazon, R., Enderlin, J.M., Laugier, G., Lagarde, M. and Pageaux, J.F. Bis(monoacylglycerol) phosphate in rat uterine stromal cells: structural characterization and specific esterification of docosahexaenoic acid. Biochem. J., 351, 795-804 (2000); DOI.
- Medoh, U.N. and Abu-Remaileh, M. The bis(monoacylglycero)phosphate hypothesis: from lysosomal function to therapeutic avenues. Annu. Rev. Biochem., 93, 447-469 (2024); DOI.
- Sheokand, P.K. and others. RAM-LAG1-CLN8 family proteins are acyltransferases regulating phospholipid composition. Sci. Adv., 11, eadr3723 (2025); DOI.
- Showalter, M.R., Berg, A.L., Nagourney, A., Heil, H., Carraway, K.L. and Fiehn, O. The emerging and diverse roles of bis(monoacylglycero) phosphate lipids in cellular physiology and disease. Int. J. Mol. Sci., 21, 8067 (2020); DOI.
- Singh, S., Dransfeld, U.E., Ambaw, Y.A., Lopez-Scarim, J., Farese, R.V. and Walther, T.C. PLD3 and PLD4 synthesize S,S-BMP, a key phospholipid enabling lipid degradation in lysosomes. Cell, 187, 6820-6834.e24 (2024); DOI.
- Wang, X.Y., Schmitt, M.V., Xu, L.N., Jiao, Y.P., Guo, L.J., Lienau, P., Reichel, A. and Liu, X.H. Quantitative molecular tissue atlas of bis(monoacylglycero)phosphate and phosphatidylglycerol membrane lipids in rodent organs generated by methylation assisted high resolution mass spectrometry. Anal. Chim. Acta, 1084, 60-70 (2019); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: December 2025 | ||
© The LipidWeb is open access and fair use is encouraged but not text and data mining, AI training, and similar technologies.
