Diacylglycerols
Diacylglycerols (or "diglycerides") are esters of the trihydric alcohol glycerol in which two of the hydroxyl groups are esterified with long-chain fatty acids. They can exist as three stereochemical isomers (see our web document on Triacylglycerols (part 1) for a discussion of nomenclature). When the stereochemistry is unknown or when the mixture is racemic, sn-1,2- and 2,3-diacylglycerols are sometimes termed α,β-diacylglycerols while sn-1,3-diacylglycerols may be designated α,α′‑diacylglycerols.
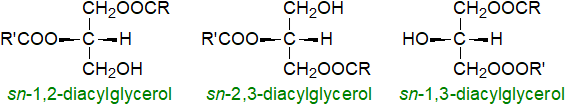 |
| Figure 1. Stereoisomers of diacyl-sn-glycerols. |
Diacylglycerols are formed in animal and plant tissues as intermediates in the biosynthesis of triacylglycerols and other glycerolipids and during the hydrolysis of these by lipases. While their presence is of technological relevance in commercial seed oils as small amounts can have a profound influence on the physical properties, the function of sn-1,2-diacylglycerols derived from phospholipids as signalling mediators in animal tissues is of special importance for human health and wellbeing and is a focus of this web page.
1. Metabolism of sn-1,2-Diacylglycerols in Animals
sn-1,2-Diacylglycerols tend to be minor components of most tissues and membranes in quantitative terms, but in animal cells, they are required as intermediates in the biosynthesis of many glycerolipids, and they take part in signalling processes as second messengers, which respond to external stimuli to modulate vital biochemical mechanisms. They are the only one of the three stereoisomers that act in this way, and they are synthesised and metabolized by innumerable enzymes at spatially different cellular locations, each with distinct enzymatic properties and selectivities.
Biosynthesis and metabolism: sn-1,2-Diacylglycerols are primary intermediates in the formation of phosphatidylcholine, phosphatidylethanolamine and triacyl-sn-glycerols in eukaryotes, and for this purpose, phosphatidate phosphatases (including lipins 1, 2 and 3) are the enzymes that convert phosphatidic acid to sn‑1,2‑diacylglycerols (and are discussed in greater detail in the web pages dealing with these lipids, especially the last). The reverse reaction in which phosphatidic acid is produced by the action of a diacylglycerol kinase is likewise of relevance to tissue concentrations (see below).
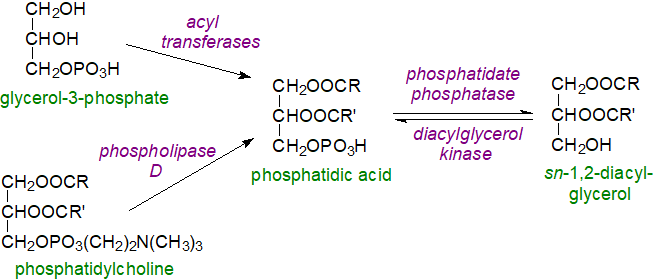 |
| Figure 2. Biosynthesis of sn-1,2-diacylglycerols via phosphatidic acid. |
Most phosphatidic acid is generated de novo via the Kennedy pathway with glycerol-3-phosphate as the precursor, but a second mechanism involves the action of a phospholipase D on phosphatidylcholine. The last can be a direct precursor for diacylglycerols via the action of phospholipase C (see below), and they are also formed as intermediates from monoacylglycerols during the biosynthesis of triacylglycerols by the monoacylglycerol pathway. During the biosynthesis of sphingomyelin, 1,2‑sn‑diacylglycerols are produced from phosphatidylcholine by an exchange reaction with ceramide catalysed by the sphingomyelin synthases SMS1 and SMS2 in the trans-Golgi and plasma membrane; a related pathway may be present in some pathogenic fungi.
sn-1,2-Diacylglycerols can be intermediates in the catabolism of all glycerolipids and during digestion of dietary lipids in the intestines via hydrolysis by lipases. In humans, the lingual lipase in the mouth hydrolyses the ester bond in the sn-3 position of some of the dietary triacylglycerols preferentially, thus generating mainly sn‑1,2‑diacylglycerols, while pancreatic lipase subsequently gives a racemic mixture of the sn‑1,2- and 2,3-isomers on the way to further digestion products such as monoacylglycerols. In contrast, adipose tissue triacylglycerol lipase generates racemic 1,3‑diacylglycerols with 2,3-diacyl-sn-glycerols, and these are hydrolysed further by the hormone-sensitive lipase (see the web page on triacyl-sn-glycerols).
 Physical properties in membranes: sn-1,2-Diacylglycerols accumulate transiently in membranes, where they
bind via strong hydrophobic interactions to particular proteins and then cause changes in the physical properties of the bilayer.
It has been established that subtle differences in diacylglycerol structure influence lipid-protein interactions and the
kinetics of trans-bilayer movement and lipid turnover to result in preferential recruitment of proteins with signalling functions.
As their polar head group is small, they tend to form inverted micellar structures.
In practice, this means that diacylglycerols introduce small areas of unstable negative curvature in membranes that influence the general
morphology of organelles and facilitate membrane fission or fusion.
Membrane fusion is often necessary for efficient cellular metabolism, and diacylglycerols in membranes can assist this process partly via
their physical properties and partly through activation of certain proteins.
For example, diacylglycerols in the cis-Golgi are necessary for the assembly of the nuclear envelope following mitosis.
While rapid trans-bilayer movement (or flip-flop) of diacylglycerols can occur and might suggest an even distribution
between the two leaflets of the plasma membrane, some studies indicate that they flip more slowly across an ordered
raft-like bilayer, i.e., one enriched in sphingomyelin and cholesterol, than across a more
fluid bilayer composed of unsaturated glycerophospholipids.
Physical properties in membranes: sn-1,2-Diacylglycerols accumulate transiently in membranes, where they
bind via strong hydrophobic interactions to particular proteins and then cause changes in the physical properties of the bilayer.
It has been established that subtle differences in diacylglycerol structure influence lipid-protein interactions and the
kinetics of trans-bilayer movement and lipid turnover to result in preferential recruitment of proteins with signalling functions.
As their polar head group is small, they tend to form inverted micellar structures.
In practice, this means that diacylglycerols introduce small areas of unstable negative curvature in membranes that influence the general
morphology of organelles and facilitate membrane fission or fusion.
Membrane fusion is often necessary for efficient cellular metabolism, and diacylglycerols in membranes can assist this process partly via
their physical properties and partly through activation of certain proteins.
For example, diacylglycerols in the cis-Golgi are necessary for the assembly of the nuclear envelope following mitosis.
While rapid trans-bilayer movement (or flip-flop) of diacylglycerols can occur and might suggest an even distribution
between the two leaflets of the plasma membrane, some studies indicate that they flip more slowly across an ordered
raft-like bilayer, i.e., one enriched in sphingomyelin and cholesterol, than across a more
fluid bilayer composed of unsaturated glycerophospholipids.
Generation of sn-1,2-diacylglycerols for signalling purposes: sn-1,2-Diacylglycerols are synthesised at the plasma membrane together with another class of signalling molecules, water-soluble inositol phosphates, by the action of the enzyme phospholipase C on phosphatidylinositol and the polyphosphoinositides with production of the 1-stearoyl-2-arachidonoyl-sn-glycerol species mainly (see the web page on these lipids for a more detailed discussion). While phosphatidylinositol-4,5-bisphosphate has long been considered the main source of diacylglycerols for signalling purposes, it now seems that phosphatidylinositol 4‑phosphate may be more important after stimulation of G protein–coupled receptors. A family of at least sixteen phospholipase C enzymes (three atypical) in seven sub-families exist with differing subcellular locations and substrate specificities that are activated by agonists at receptors on membranes. Of these, a calcium-dependent phosphoinositide-specific phospholipase C (or 'phosphoinositidase C') is especially significant in this context. A key feature of the reaction is that the resulting diacylglycerols are retained in the membrane in which they are formed. While the response is immediate, it is short-lived, and in comparison, the other routes to diacylglycerols occur more slowly but are of longer duration. Under basal conditions before stimulation by hormones or neurotransmitters, 1‑stearoyl,2‑arachidonoyl-sn-glycerol is present at relatively low levels in cells in comparison to other molecular species.
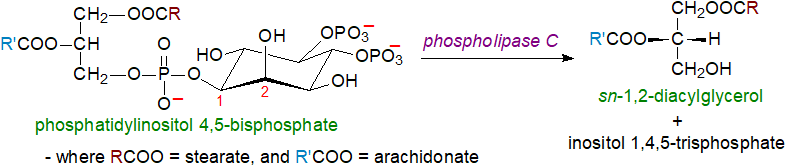 |
| Figure 3. Hydrolysis of phosphatidylinositol to sn-1,2-diacylglycerols by phospholipase C. |
Other phospholipids can by hydrolysed to sn-1,2-diacylglycerols in the same way to generate various molecular species of diacylglycerols, but while their relevance from a signalling standpoint is less well established, they may be important for lipid remodelling. Perhaps surprisingly, the cytosolic phospholipase C that hydrolyses phosphatidylcholine has not been characterized and the relevant gene is not known, although sphingomyelin synthases 1 and 2 are candidates and can generate saturated- and/or monounsaturated fatty acid-containing diacylglycerols from several phospholipids, and phosphatase orphan 1 (PHOSPHO1) is a further possibility. Two isoforms of phospholipase C activity that are specific for phosphatidylcholine have been identified in natural killer cells at the outer leaflet of the plasma membrane and within the cytoplasm.
In the cell nucleus, it appears that there are two distinct pools of diacylglycerols with very different compositions that are produced from phosphatidylinositides (polyunsaturated) and phosphatidylcholine (saturated and monoenoic) by appropriate stimuli, and these may be needed for different purposes. The fatty acid compositions of the diacylglycerols derived from most routes reflect the compositions of the parent phospholipids, although there is evidence that in general, the diacylglycerols must contain polyunsaturated fatty acids to act optimally as messengers.
Protein kinase C: An important function of sn-1,2-diacylglycerols, such as those derived from phosphatidylinositol, is that they affect vital processes in cell physiology by binding to and activating members of the protein kinase C (PKC) family of enzymes, often acting in concert with the soluble phosphoinositides. These enzymes regulate other proteins through the phosphorylation of hydroxyl groups of serine and threonine amino acid residues, and they can sense diacylglycerols generated in the different cellular compartments in various physiological processes. The sn-1,2-configuration is crucial, and while 1‑stearoyl,2-arachidonoyl-sn-glycerol has received most study, it has become evident that other molecular species are differentially recruited to characteristic diacylglycerol binding sites, and these may each result in different downstream phosphorylation patterns.
Diacylglycerols act to increase the concentration of calcium ions in the cell, which stimulates the translocation of the various iso-enzymes of protein kinase C to the inner face of the plasma membrane, followed by intracellular relocation to organelles such as the mitochondria, endoplasmic reticulum, Golgi apparatus and nucleus. The main PCKs (α, β and γ) bind in a 1:1 ratio to the sn‑1,2‑diacylglycerols at a highly conserved cysteine-rich ‘C1’ domain, which consists of a sequence of 50 amino acids in a characteristic motif. Other conserved regions such as the ‘C2’ domain assist in membrane recruitment of the kinase by interaction with anionic phospholipids, often by a mechanism triggered by Ca2+. Phosphatidylserine can serve this purpose, but there is a strong preference for phosphatidylinositol 4,5-bisphosphate, mediated by a basic patch distal to the Ca2+ binding site that targets these PKC isoenzymes selectively to the plasma membrane. A second novel group of PCKs (δ, ε, η, θ) contain tandem C1 domains, which bind diacylglycerols. At the same time, by exposing small areas of the apolar regions of neighbouring lipids, they improve the hydrophobic interactions with other proteins within membranes thereby affecting their activities.
Thus, protein kinase C isoenzymes regulated by lipid second messengers contain one or more membrane-targeting modules, which result in protein kinase activation, typically by relieving autoinhibitory constraints. They are involved in both short- and long-term modifications of normal cellular physiology with more than a hundred substrates identified to date, and in consequence, diacylglycerols have a role in the pathophysiology of many disease states. As they accumulate in many different organs of obese patients, for example, this can lead to the disruption of metabolic homeostasis and the development of diabetes and associated metabolic diseases.
They are a factor in cancer, and disruptions to the balance between diacylglycerol generation by phospholipase C and termination by diacylglycerol kinases are known to be associated with tumour initiation, progression and metastasis. By means of mass spectrometric imaging of lumpectomy specimens from breast cancer patients, diacylglycerols derived from phosphatidylcholine were found to be substantially elevated relative to those in normal tissue and are therefore biomarkers for the diagnosis of the disease. Diacylglycerols are regulators of T cells and are intimately involved in tumour immuno-surveillance with an influence upon the interaction between cancer cells and their cellular environment. As an aid to study of these processes, an invaluable finding has been that tumour-promoting phorbol esters mimic diacylglycerols to stimulate the same enzymes (phorbol is a tetracylic diterpene produced by certain plant families that is not metabolized in tissues so is persistent in its activity), and so they are valuable biomedical research tools in models of carcinogenesis.
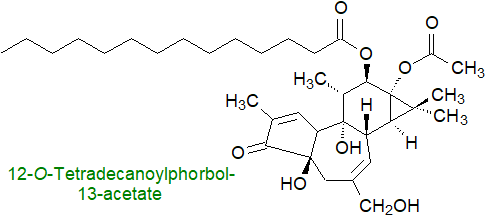
Other functions: Diacylglycerols bind to protein kinase D, a cytosolic serine-threonine kinase that in turn binds to the trans-Golgi membrane network and regulates transport of proteins to the cell surface; in the absence of diacylglycerols, protein transport is blocked. In this context, those diacylglycerols generated by the action of sphingomyelin synthase may be important. The identification of non-kinase receptors of sn-1,2-diacylglycerols, many but not all of which have the conserved C1 domain, has revealed new and strategic functions in regulating cellular responses and in cytoskeletal remodelling. In migrating endothelial leader cells, phospholipase C signalling is restricted to the front to generate a diacylglycerol gradient that by interacting with the integrated Ca2+ control system promotes persistent forward migration. Diacylglycerols interact with the insulin signalling cascade and thence with glucose uptake in skeletal muscle. They are one of the precursors of the endocannabinoid 2‑arachidonoylglycerol with its own distinctive signalling properties.
In insects, although lipids are stored as triacylglycerols in fat bodies, they are transported in haemolymph (the insect equivalent of plasma) as sn‑1,2‑diacylglycerols bound to the lipoprotein lipophorin to those tissues where they are required for synthesis of membrane phospholipids and as a source of energy (see our web page on lipoproteins).
Diacylglycerol kinases: A significant proportion of the sn-1,2-diacylglycerols are removed from tissues by serving as precursors for the biosynthesis of phosphatidic acid via the action of diacylglycerol kinases with ATP as the phosphate donor (as illustrated in Figure 2 above). This is in turn an intermediate in the biosynthesis of other glycerolipids as well as being a signalling mediator in its own right. In mammals, there is a family of at least ten diacylglycerol kinase isoenzymes (in five subfamilies), which are structurally related to the sphingosine kinase, and each of these has slightly different properties and functions; all contain a shared lipid kinase catalytic domain. These may be segregated in distinct cellular organelles and activated by different means, and while some are cytosolic, some are associated with membranes, and others are located within the nucleus. The various isoenzymes use saturated-monosaturated fatty acid and docosahexaenoic acid (22:6)-containing diacylglycerol species, but with one exception, not those derived from phosphatidylinositol (18:0/20:4-species). Diacylglycerol kinases can contribute to cellular asymmetry and control the polarity of cells by regulating the gradients in diacylglycerol and phosphatidic acid concentrations. They have a negative effect on signalling by diacylglycerols by reducing their concentrations in cells, but they do generate phosphatidic acid for signalling purposes.
Diacylglycerol kinase kappa (DGKκ) has been termed a master regulator that controls the switch between the diacylglycerol and phosphatidic acid signalling pathways. In the brain, different isoenzymes are expressed in different types of neurons, but a deficiency of DGKκ in neurons is a critical factor in Fragile X syndrome, a common cause of inherited mental retardation and autism. A further isoform of the enzyme DGKζ controls diacylglycerol metabolism at the immunological synapse, where it interacts with PKCα. The expression of DGKα and DGKζ isoforms is increased in T cells in comparison with other cell types. PHOSPHO1 may supply diacylglycerols to DGKδ
DGKε is the only DGK isoform with specificity for one particular species of diacylglycerol, i.e., 1-stearoyl-2-arachidonoyl-sn-glycerol. It is unique in that it is the smallest of the DGK isoforms, it is the only one devoid of a regulatory domain, and it is the only one to have a hydrophobic segment that is predicted to exist as a transmembrane helix. As DGKε utilizes diacylglycerols containing the same acyl chains as those found in phosphatidylinositol, it is seems likely that it is responsible for catalysing one step in the phosphatidylinositol cycle. It is also reported to promote adipose tissue remodelling in mice with effects upon obesity, insulin resistance and beige adipogenesis. In so doing, it alters the expression of lipid metabolizing enzymes, such as lipases and diacylglycerol acyltransferase (DGAT), suggesting an involvement in lipid energy homeostasis.
Diacylglycerols in nutrition: Mixed diacylglycerols are manufactured commercially by enzymic hydrolysis for use as emulsifiers, and edible oils consisting of 80% 1,3‑diacylglycerols are marketed in Japan, Europe and the USA as cooking oils and vegetable oil spreads. It is suggested that they are metabolized in a different way from triacylglycerols and have beneficial nutritional properties. When they are digested, the resulting 1/3‑monoacylglycerols are absorbed into tissues relatively poorly, apparently limiting the accumulation of fats in body tissues. Further, it has been suggested that they slow the increase in triacylglycerol and cholesterol concentrations in blood after a meal, and that there is increased fat oxidation with an influence on food intake by increasing satiety. Although these commercial 1,3‑diacylglycerol oils are claimed to be useful anti-obesity agents, they have been banned from sale in some countries as they have been found to contain glycidol fatty acid esters, which are potentially carcinogenic.
Catabolism: Diacylglycerols are hydrolysed efficiently by hormone-sensitive lipase in adipose tissue and by diacylglycerol lipases both at the plasma membrane and intracellularly in many tissues. Of course, they are removed from cells when they are utilized as intermediates in the synthesis of other lipids, for example by the action of diacylglycerol kinases.
2. Diacylglycerol Metabolism in Plants and Microorganisms
In plants and some microorganisms, sn-1,2-diacylglycerols are intermediates in the biosynthesis of glycerolipids, including many phospholipids and the mono- and digalactosyldiacylglycerols. By perturbing membrane structure, diacylglycerols may affect plant enzymes indirectly, but there is relatively limited evidence for the existence of diacylglycerol signalling pathways in higher plants. Only one enzyme related to the protein kinase C family tends to be found in plants (PKCδ) that might potentially be regulated, but no relevant receptor has been detected. Rather, diacylglycerols generated from phosphatidylcholine, phosphatidylethanolamine and other phospholipids by the action of non-specific phospholipases Cs, six forms of which are known in Arabidopsis, are rapidly phosphorylated by diacylglycerol kinases to phosphatidic acid, and this is the main second messenger. Incidentally, as well as their action on phospholipids, two of the isoforms of non-specific phospholipases C hydrolyse the glycosidic bond in monogalactosyldiacylglycerols to yield diacylglycerols. There appears to be little data on the role of individual phospholipase C isoforms in the subsequent synthesis of new lipids.
There is at least one phospholipase C that is specific for phosphatidylinositol and lacks the pleckstrin homology (PH) domain of the mammalian equivalents, and there is evidence that some stress conditions can be alleviated by the action of this enzyme. Phosphatidylinositol 4,5-bisphosphate is a substrate that yields diacylglycerols with inositol 1,4,5-phosphate (IP3), which may have a role in plant growth, development and stress responses, and there is one report that diacylglycerols may be signalling mediators in gametophyte development and root growth. Apart from signalling, diacylglycerols participate in catabolism, remodelling of lipids in membranes and lipid synthesis and metabolism in general. There is a suggestion that the water-soluble products of phospholipase C, e.g., phosphorylcholine from phosphatidylcholine, rather than diacylglycerols may act in signalling in plants.
Fungi contain a phospholipase C subtype, which is related to the mammalian PLCδ and regulates the intracellular calcium concentration, and its activity may be related to pathogenicity, especially in plant infections. Others are known that degrade plant glycosylinositol phosphorylceramides or cellulose during infections, or inhibit the growth of nematodes. Enzymes of the phospholipases C type have been identified in bacteria, and these use a wide range of substrates and are often toxins in animal hosts that can determine the outcome of host–pathogen interactions. However, rather than utilizing diacylglycerol intermediates, phospholipids in prokaryotes are synthesised almost entirely via the CDP-diacylglycerol pathway.
3. Analysis
It is easy to generate diacylglycerols artefactually on extracting or storing tissues if inappropriate methods are used. Often, attempts are made to analyse 1,2-/2,3- and 1,3-diacylglycerols separately, but the data may not be meaningful as acyl migration occurs rapidly until an equilibrium is reached with about 67% of the 1,3-isomer. All diacylglycerols will isomerize slowly on standing in inert solvents or in the dry state even at low temperatures. They can be recovered from tissues with minimal isomerization, when necessary, by extracting the tissues with non-alcoholic solvents such as diethyl ether or chloroform, taking care not to heat extracts at any stage. When pure positional isomers are required, it is necessary to chromatograph the partial glycerides on TLC plates coated with silica gel G impregnated with boric acid at a level of 10% of the adsorbent), using a solvent system of chloroform (alcohol-free)-acetone (96:4, v/v). To differentiate analytically between sn-1,2- and 2,3-diacylglycerols, the stereospecific analysis procedures used for triacyl-sn-glycerols can be adapted to the purpose; methods using chiral phase chromatography are promising.
Routine determination of molecular species of diacylglycerols in lipid extracts can be accomplished by various chromatographic methods of which high-temperature gas chromatography was considered the most appropriate until recently, as information on the composition as well as the absolute amount is obtained in this way. Modern mass spectrometric methodology linked to HPLC is now favoured by those with access to the technology.
References
- Ali, U., Lu, S.P., Fadlalla, T., Iqbal, S., Yue, H., Yang, B., Hong, Y.Y., Wang, X.M. and Guo, L. The functions of phospholipases and their hydrolysis products in plant growth, development and stress responses. Prog. Lipid Res., 86, 101158 (2022); DOI.
- Almena, M. and Mérida, I. Shaping up the membrane: diacylglycerol coordinates spatial orientation of signalling. Trends Biochem. Sci., 36, 593-603 (2011); DOI.
- Cooke, M. and Kazanietz, M.G. Overarching roles of diacylglycerol signaling in cancer development and antitumor immunity. Science Signal., 15, eabo0264 (2022); DOI.
- Eichmann, T.O. and Lass, A. DAG tales: the multiple faces of diacylglycerol-stereochemistry, metabolism, and signaling. Cell. Mol. Life Sci., 72, 3931-3952 (2015); DOI.
- González-Mendoza, V.M., Sánchez-Sandoval, E., Castro-Concha, L.A. and Hernández-Sotomayor, S.M.T. Phospholipases C and D and their role in biotic and abiotic stresses. Plants-Basel, 10, 921 (2021); DOI.
- Ianni, F., Carotti, A., Protti, M., Favilli, A., Gerli, S., Furlanetto, S., Mercolini, L. and Sardella, R. Chiral high-performance liquid chromatography analysis of mono-, di-, and triacylglycerols with amylose- and cellulose-phenylcarbamate-based stationary phases. J. Pharm. Biomed. Anal., 236, 115720 (2023); DOI.
- Katan, M. and Cockcroft, S. Phospholipase C families: Common themes and versatility in physiology and pathology. Prog. Lipid Res., 49, 101065 (2020); DOI.
- Laxalt, A.M., van Hooren, M. and Munnik, T. Plant PI-PLC signaling in stress and development. Plant Physiol., 197, kiae534 (2025); DOI.
- Lee, W.J., Zhang, Z., Lai, O.M., Tan, C.P. and Wang, Y. Diacylglycerol in food industry: Synthesis methods, functionalities, health benefits, potential risks and drawbacks. Trends Food Sci. Technol., 97, 114-125 (2020); DOI.
- Mondal, S., Pal, B. and Sankaranarayanan, R. Diacylglycerol metabolism and homeostasis in fungal physiology. FEMS Yeast Res., 24, foae036 (2024); DOI.
- Racca, L., Baldanzi, G. and Massarotti, A. Modulators of diacylglycerol kinase activity: A review of advances and challenges. Med. Res. Rev., 46, 149-175 (2026); DOI.
- Schuhmacher, M., Grasskamp, A.T., Barahtjan, P., Wagner, N., Lombardot, B., Schuhmacher, J.S., Sala, P., Lohmann, A., Henry, I., Shevchenko, A., Coskun, U., Walter, A.M. and Nadler, A. Live-cell lipid biochemistry reveals a role of diacylglycerol side-chain composition for cellular lipid dynamics and protein affinities. Proc. Natl. Acad. Sci. USA, 117, 7729-7738 (2020); DOI).
- Ware, T.B. and Hsu, K.L. Advances in chemical proteomic evaluation of lipid kinases-DAG kinases as a case study. Curr. Opinion Chem. Biol., 65, 101-108 (2021); DOI.
- Yang, B., Fan, R.Y., Yao, S.B., Lou, H.X., Li, J.W., Guo, L. and Wang, X.M. Non-specific phospholipase Cs and their potential for crop improvement. J. Exp. Botany, in press (2025); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: February 2026 | ||
© The LipidWeb is open access and fair use is encouraged - but not text and data mining, AI training, and similar technologies (text scraping).
