Lipid of the Month
Each month we highlight a lipid of scientific interest. The LIPID MAPS® Lipid of the Month Archive lists lipids highlighted from 2015 - present.
March 2026
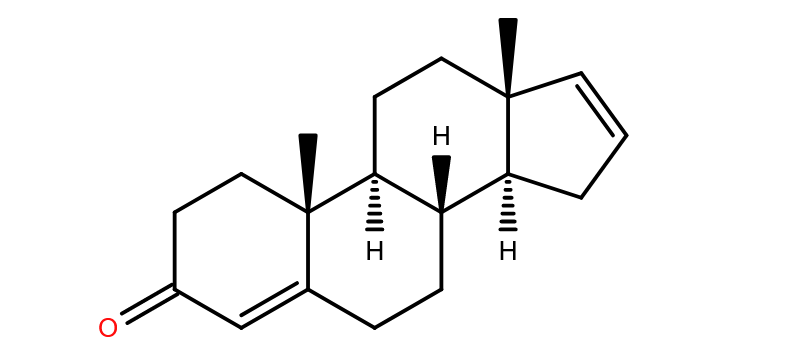
 alpha-mycolic acid
alpha-mycolic acid
It’s nearly 18,000 years ago, Wyoming is in the grip of the last ice age and bison trudge over the snowy ground. Failing to see the edge, one animal, with a chronic respiratory condition, falls into Natural Trap Cave, a sinkhole in the ancient limestone. The 30m fall is likely instantly fatal.
Fast forward to modern times and the bison’s bones are excavated and analysed. DNA from Mycobacterium tuberculosis is discovered in them1 but so also are lipids characteristic of the bacterium2, for example alpha-mycolic acid. These findings show the bison was suffering from TB.
Alpha-mycolic acid forms a family of very long chain fatty acids, typically between 60 and 90 carbons long divided into 2 sidechains and with one or more cyclopropane groups. One of the most abundant found in the bison had 80 carbons. Alpha-mycolic acid is part of a complex array of lipids in the cell wall of M. tuberculosis which makes it resistant to dehydration, many drugs, and allows it to live inside macrophages. The bacterium was discovered by Robert Koch, who announced his finding that it caused TB on 24th March 18823, the reason world TB day occurs on that date.
In spite of many efforts, TB still kills over 1 million people each year. The surface lipids modulate interactions with the host, and so are promising targets for anti-TB drugs4.
References
-
Mycobacterium tuberculosis complex DNA from an extinct bison dated 17,000 years before the present
Clin Infect Dis
2001
DOI 10.1086/321886
-
Mycobacterium tuberculosis complex lipid virulence factors preserved in the 17,000-year-old skeleton of an extinct bison, Bison antiquus
PLoS One
2012
DOI 10.1371/journal.pone.0041923
-
Steps towards the discovery of Mycobacterium tuberculosis by Robert Koch, 1882
Clin Microbiol Infect
2014
DOI 10.1111/1469-0691.12555
-
The lipid language of tuberculosis: Mycobacterium tuberculosis surface molecules in host interaction and drug resistance
mBio
2026
DOI 10.1128/mbio.03959-25
Lipid of the Month Archive
February 2026

In the middle of February comes the beginning of a new year in the Chinese lunar calendar. We enter the year of the horse in the Chinese zodiac.
Horse lipids have been studied for a long time, particularly the steroid hormones. Compounds such as equilin, hippulin1 and estrone sulfate2 were first discovered in horse urine, presumably because it is available in significant quantities. In the days before modern, sensitive equipment, large amounts of samples were needed for any biochemical analysis.
Other lipids are found in the sebum of horses- the oily excretion on the skin. These include a range of long-chain (34-38 carbons) lactones with two double bonds such as 36-methyl-21Z,29Z-heptatriacontadien-37-olide. While a series of similar lactones were characterised in 19843, it seems the function of these molecules remains enigmatic.
Though they are closely related species, horse and zebra sebum lactones differ. Horse lactones have been found to be branched (mostly formed from iso- fatty acids), whereas those from zebra were unbranched4. We understand however that there are easier ways to tell the difference between a horse and a zebra without requiring extensive molecular exploration of their skin secretions!
If you’re celebrating starting the year of the horse this month, Gong hei fat choy- 恭喜发财
References
-
The resolution of isoequilin A and the identification of compound 3.
J Biol Chem
1950
DOI 10.1016/S0021-9258(18)56200-1
-
THE ISOLATION OF ESTRONE SULFATE FROM THE URINE OF PREGNANT MARES,
J Biol Chem
1938
DOI 10.1016/S0021-9258(18)73874-X
-
Structures of the dienoic lactones of horse sebum
Comp Biochem Physiol B
1984
DOI 10.1016/0305-0491(84)90095-6
-
Variation in skin surface lipid composition among the equidae
Comp Biochem Physiol B
1983
DOI 10.1016/0305-0491(83)90353-X
January 2026
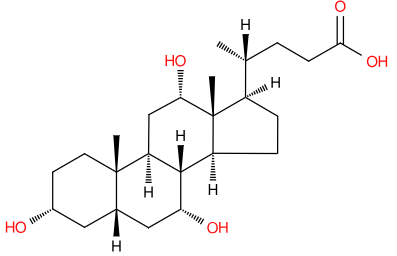
Over the holiday season, it’s likely that many of us have ingested rather more lipids than perhaps we intended. In fact, there’s research showing cholesterol levels spike in January making hypercholesterolemia diagnoses unreliable1. While digesting those holiday lipids, the body will have made use of a further one- cholic acid, which helps solubilise fats in the gut.
Cholic acid, together with chenodeoxycholic acid, are the main bile acids produced in humans, but they can be modified in many ways, not least by gut bacteria2. These bile acids act as detergents to help solubilise dietary fat and aid their absorption into the body. They’re produced from cholesterol by the liver and excreted into the intestinal tract via the gall bladder.
But bile acids don’t just help digest food, they act as signalling molecules, regulating their own synthesis and binding to nuclear hormone receptors, which control gene transcription. They are implicated in a range of diseases3 including metabolic and immune conditions. There is even a suggestion that bile acids could have a role in mental health4.
References
-
The Christmas holidays are immediately followed by a period of hypercholesterolemia
Atherosclerosis
2019
DOI 10.1016/j.atherosclerosis.2018.12.011
-
Review: microbial transformations of human bile acids
Microbiome
2021
DOI 10.1186/s40168-021-01101-1
-
Bile Acids Activated Receptors Regulate Innate Immunity
Front Immunol
2018
DOI 10.3389/fimmu.2018.01853
-
Bile acid signalling and its role in anxiety disorders
Front Endocrinol
2023
DOI 10.3389/fendo.2023.1268865
December 2025

Following a tradition that's nearly 80 years old, the Christmas tree in Trafalgar Square in London comes from Norway. It's a spruce gifted to the people of London from the people of Oslo which even has its own social media accounts.
Within that tree is a lot of December’s Lipid of the Month, alpha-pinene, one of the compounds in conifers which contribute to the distinctive pine forest smell. Synthesised from geranylpyrophosphate1, a common precursor to prenol and sterol lipids, it is arguably the most abundant terpenoid in nature. Together with its isomers, alpha-pinene is found in conifers, and other plants including herbs and cannabis, though it isn’t psychoactive.
Pinene has many therapeutic properties and has been used medicinally for centuries2 by humans. In the tree it’s a chemical used as an anti-freeze and for defence against predation, though conversely, it also acts as an attractant to some insect pests3.
Of course, once cut down from the forests of Norway, no amount of pinene can stop the tree from dying, though conifers take a long time to wilt, go brown and look like they’re dead. When the tree is taken down at the end of the Christmas season, it may seem to be dead, but as Monty Python might say, it is pinene from the fjords.
References
-
Microbial Synthesis of Pinene
ACS Synth Biol
2014
DOI 10.1021/sb4001382
-
α-Pinene: A never-ending story
Phytochemistry
2021
DOI 10.1016/j.phytochem.2021.112857
-
Volatiles as Attractants of Mahogany Shoot Borer, Hypsipyla grandella Zeller (Lepidoptera: Pyralidae)
J Chem Ecol
2023
DOI 10.1007/s10886-022-01398-8
November 2025
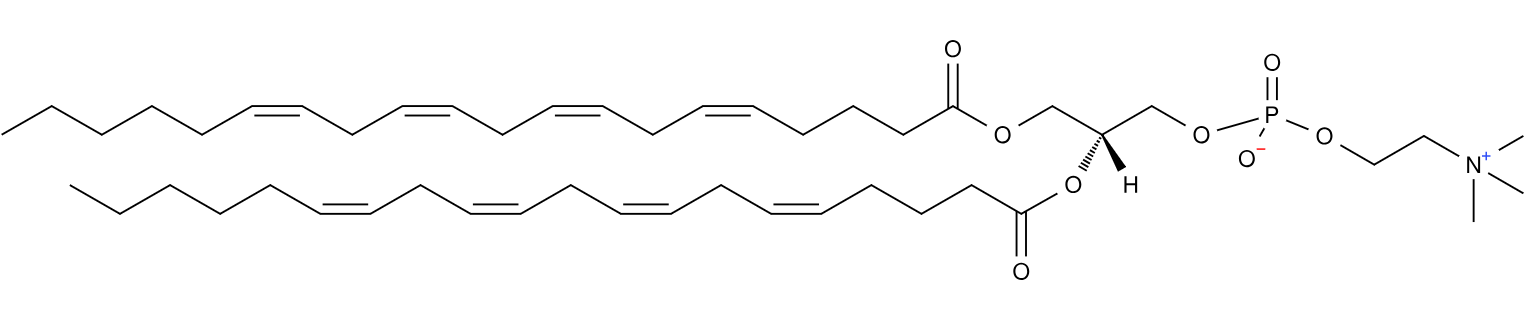
In general, phospholipids in the mammalian cell membrane are asymmetric. The sn1 fatty acid tends to be much more saturated than that at the sn2 position. Indeed, often the sn1 acyl is totally saturated, i.e. it has no double bonds.
There are of course exceptions and one such is the rarely found diarachidonyl phosphatidylcholine (PC 20:4/20:4) first described in 1982 from rat neutrophils supplemented with arachidonic acid and after inducing an inflammatory response1.
This lipid is formed by esterification of arachidonic acid to a lyso-pC, and in the presence of a large amount of arachidonic acid, it is perhaps no surprise that it will get incorporated. But the role of this lipid, besides storing arachidonic acid, was previously unclear. The inflammation factor was of course intriguing.
Last year, this lipid was shown to have a key role in ferroptosis2- the complex process by which a cell dies after excessive peroxidation of its lipids induced by iron and reactive oxygen species, which can be a runaway process. Phospholipids containing polyunsaturated fatty acids are particularly susceptible to ferroptosis mechanisms but also induce them. Diarachidonyl PC can promote the formation of reactive oxygen species in mitochondria.
Diarachidonyl PC, and no doubt other species with two polyunsaturated acyl chains add to the complicated, and not yet fully understood, story of ferroptosis.
References
-
The formation of diarachidonyl diglyceride by rat neutrophils.
Mol Pharm
1982
DOI 10.1016/S0026-895X(25)14930-4
-
Phospholipids with two polyunsaturated fatty acyl tails promote ferroptosis
Cell
2024
DOI 10.1016/j.cell.2024.01.030
October 2025
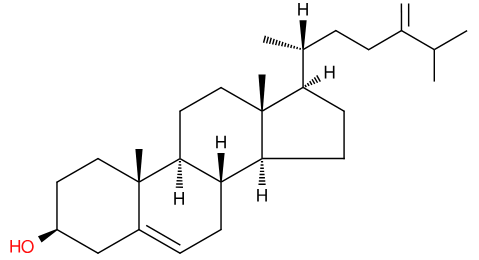
There is much literature on the benefits of plant sterols. To humans, they may be beneficial, but they aren’t essential. To insects, however, they’re vital because insects can’t synthesise sterols de novo1.
24-methylene-cholesterol (24-MC) is one such plant sterol. Also called chalinasterol or ostreasterol, It’s the main sterol in the pollen of many flowers2 and a molecule essential for the growth and development of honeybees. Larvae are fed jelly produced by the workers that is rich in 24-MC3. Without these sterols, bee larvae cannot be reared.
In times when pollen and nectar are not abundant, bee-keepers provide supplemental food for their hives, but this is lacking the sterols that bees require. These molecules are not available in quantities that would make it commercially viable.
A recent paper in Nature describes how a yeast, Yarrowia lipolytica, has been engineered to produce 24-MC and other essential sterols to feed the bees4. To do this, the genes to make ergosterol (the fungal equivalent of cholesterol) had to be removed and other genes to make 24-MC added. That strain was further modified to produce other sterols essential for bees, including desmosterol, campesterol, and cholesterol.
Bee colonies fed with the yeast product were able to rear brood for much longer than those without, so in a time when bees are under threat, there’s a glimmer of hope. This paper should produce quite a buzz!
References
-
The utilization of sterols by insects
J Lipid Res
1964
DOI 10.1016/S0022-2275(20)40254-8
-
Pollen sterols—a mass spectrographic survey
Phytochemistry
1968
DOI 10.1016/S0031-9422(00)85638-1
-
Mandibular glands secrete 24-methylenecholesterol into honey bee (Apis mellifera) food jelly
Mandibular glands secrete 24-methylenecholesterol into honey bee (Apis mellifera) food jelly
2023
DOI 10.1016/j.ibmb.2023.104011
-
Engineered yeast provides rare but essential pollen sterols for honeybees
Nature
2025
DOI 10.1038/s41586-025-09431-y
September 2025

Identifying a biomarker for a particular disease can be hugely useful in establishing a diagnosis or monitoring its progression. Eicosane, a biomarker for Parkinson’s disease (PD), was discovered in an unusual way.
Joy Milne, a nurse from Scotland, has hyperosmia- an unusually acute sense of smell- and noticed that her husband’s scent had changed. Six years later, he was diagnosed with PD and the couple started attending meetings for Parkinson’s sufferers. Milne realised that all those with the disease had the same scent as her husband1.
The odour was present in the sebum- the oily secretion on the skin which can be produced in excess in PD, a condition known as seborrhoea. Samples of sebum were fractionated using gas chromatography and then split into two, one fraction going on to mass spectrometry, in a conventional GC-MS manner, the other fraction diverted to Milne’s nose2.
One of the molecules which Milne identified as having the distinctive smell was eicosane, a twenty-carbon hydrocarbon which is has long been known to function as a pheromone in insects3, but has no clear function in humans. It may not be produced by the patient but by the yeast Malassezia, known to be a part of the skin microbiome and which is linked to seborrhoea. Its role in PD is enigmatic4 but it could be more than simply an opportunistic degrader of excess sebum causing a recognisable odour. There is much more work to be done to understand the roles of eicosane and Malassezia in PD.
References
-
Joy of super smeller: sebum clues for PD diagnostics
Lancet Neurol
2016
DOI 10.1016/S1474-4422(15)00396-8
-
Discovery of Volatile Biomarkers of Parkinson's Disease from Sebum
ACS Cent Sci
2019
DOI 10.1021/acscentsci.8b00879
-
Chemistry of the cephalic and Dufour's gland secretions of Melissodes bees
Ann Entomol
1979
DOI 10.1093/aesa/72.4.514
-
From Skin and Gut to the Brain: The Infectious Journey of the Human Commensal Fungus Malassezia and Its Neurological Consequences
Mol Neurobiol
2025
DOI 10.1007/s12035-024-04270-w
August 2025

Some sentences in the scientific literature just jump out and ask far more questions than they answer. Such was the case, digging through papers characterising disparlure, the sex pheromone of the Gypsy (or Spongy) Moth, Lymantria dispar. It’s produced by flightless females to attract males of this species which is invasive in North America.
The larvae feed on commercially important tree species and can cause major ecological and economic damage. To monitor the presence of the moth, pheromone traps are used, but to use the pheromone in traps, it first has to be identified.
The sentence which jumped out illustrated how crucial this identification was. “The sex attractant was extracted from 78,000 tips (last two abdominal segments of female moths) collected in Spain”1
Scientists in 1970 had collected 78,000 female moths, and cut off the end of the abdomen of each to extract the pheromone. How were they collected? Was each dissection done individually? What do 78,000 moths look like? So many questions! An earlier paper2 describes the extraction process (which previously had been done using 100,000 moths!) which involved over 70 litres of benzene. Health and safety was a different thing in the past.
After describing various chemical, chromatographic and mass spectrometric analyses indicating several properties of the molecule, the paper continues “Unfortunately the amount of pure attractant in the 78,000 tips was considered insufficient for adequate characterization of the attractant.” One can only imagine the despondency after all that work, and so many moths!
There was however sufficient material to narrow down the possibilities, and several potential molecules were synthesised and tested to see if they attracted male moths in the same way as the natural pheromone. The identity of disparlure as cis-7,8-epoxy-2-methyloctadecane was finally determined.
References
-
Potent sex attractant of the gypsy moth: its isolation, identification, and synthesis
Science
1970
DOI 10.1126/science.170.3953.87
-
The Stability of Hydrogenated Gypsy Moth Sex Attractant
J Econ Entomol
1959
DOI 10.1093/jee/52.1.82
July 2025
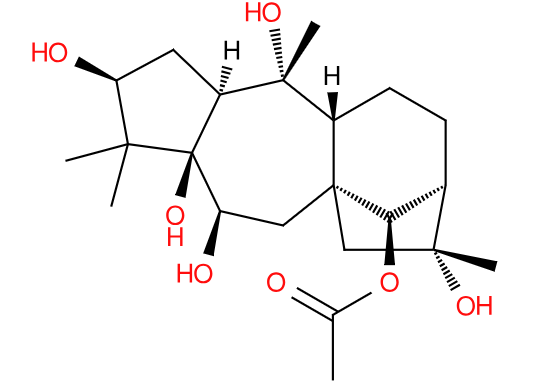
Plants seem to have a vast array of chemical defenses to avoid being eaten. Humans also seem to have a vast array of ways to exploit those chemicals for various different ends.
Such is the case with July’s Lipid of the Month, Grayanotoxin I. Produced by rhododendrons and related plants, it, and related grayanotoxins, disrupt the action of ion channels in nerve membranes causing changes in cardiac function among other symptoms1. Grayanotoxins are 20-carbon prenol lipids containing four rings, two with five carbons, one with six and one with seven. Those rings are variously decorated with methyl, methylene or hydroxyl groups2. Grayanotoxin I is one of the most prevalent.
Bees are not affected by the toxins and happily gather pollen and nectar from rhododendrons to produce what humans term ‘mad honey’. It’s mainly found in the Himalayas and in Turkey- places where there are high concentrations of grayanotoxin producing plants.
Mad honey has been used since antiquity for medicine, recreation and as a weapon. Reports from Greek and Roman literature tell of feeding mad honey to invading forces to incapacitate them3. In modern times, it’s sold as an (alleged) aphrodisiac, relaxant and treatment for peptic ulcers. Some of the websites selling it give the impression it is a veritable cure-all. There are many reports in the literature of hospital admission due to grayanotoxin poisoning, predominantly in middle-aged men, so if you’re thinking of ordering some mad honey, you do so entirely at your own risk.
References
-
Crystal and molecular structure of grayanotoxin-I, C22H36O7
Tetrahedron Lett
1970
DOI 10.1016/s0040-4039(01)98631-9
-
Mad honey: uses, intoxicating/poisoning effects, diagnosis, and treatment
RSC Adv
2018
DOI 10.1039/c8ra01924j
-
Mad Honey and the Poisoner King: A Case of Mass Grayanotoxin Poisoning in the Roman Military
Cureus
2023
DOI 10.7759/cureus.38289
June 2025

In many cities across the western world, and certainly here in Cardiff, there’s a quiet epidemic of drug use that is depleting a crucial lipid.
Sphingomyelin makes up a large proportion of the myelin sheath of nerve axons. This is (loosely) analogous to the plastic insulation around an electrical wire- without it, nerve impulse conduction is severely impaired. Sphingomyelin was shown to be a ceramide phosphocholine in 19271 and is formed by the addition of phosphocholine to ceramide. The phosphocholine is donated by phosphatidylcholine, and it is the formation of this which is inhibited by the drug nitrous oxide.
Termed ‘laughing gas’ by Humphry Davy, it has long been used as an analgesic due to the euphoric effects inhalation produces. Recently it has become readily available as a catering commodity used for whipping cream due to its solubility in fats. This availability has led to a good deal of recreational use with empty canisters and balloons (through which it is inhaled) often seen littering the city streets and carparks. Increasingly there are reports of users suffering ill-effects of nitrous oxide abuse2, including paralysis.
Nitrous oxide oxidises the cobalt in vitamin B12, rendering it inactive as a co-factor in the methytransferase enzymes that generate S-adenosyl methionine, the methyl donor in methylating phosphatidylethanolamine to form phosphatidylcholine. Absence of phosphatidylcholine means that sphingomyelin cannot be produced, leading to improperly sheathed neurons and ultimately, paralysis. The effects of laughing gas are no laughing matter.
References
-
Verhandlungen Ärztlicher Gesellschaften
Klin Wochenschr
1927
DOI 10.1007/BF01716144
-
Increasing recreational nitrous oxide use: Should we worry? A narrative review
J Psychopharmacol
2022
DOI 10.1177/02698811221082442
May 2025
May 2nd is, apparently, World Tuna Day. While it might not have the same scale as other international observations, it’s an oppor-tuna-ty (sorry!) to look at how we manage fish stocks to ensure sustainability.
Famously, fish are a source of omega-3 fatty acids, such as docosahexaenoic acid (DHA) which they accumulate via their diet, but they harbour many other lipids too. Several carotenoids, collectively termed tunaxanthins, were first isolated from tuna1. These are also present in many other fish species and marine creatures and, like DHA, are likely derived from algae in their diet.Experiments suggest tunaxanthins may be formed from dietary astaxanthin2 which is a commonly occurring pigment in algae.
In photosynthetic algae, carotenoids act as accessory pigments in photosynthesis, but higher up the food chain, tunaxanthin takes on different roles. It is responsible for the bright yellow colouration of parts of the fish3 and is even found in the red throats of male frigate birds, which they use to attract a mate.
So maintaining sustainable fish stocks isn't just about making sure your tuna mayo sandwich is always available, it might also affect lots of other creatures too.
References
-
Animal Carotenoids. 16. Tunaxanthin.
Acta Chemica Scandinavica
1978
DOI 10.3891/acta.chem.scand.32b-0621
-
Origin of tunaxanthin in the integument of yellowtail (Seriola quinqueradiata)
Comp Biochem Physiol B
1985
DOI 10.1016/0305-0491(85)90195-6
-
Carotenoids in Marine Animals
Mar. Drugs
2011
DOI 10.3390/md9020278
April 2025
Curiosity, it is said, killed the cat. Obviously, not Curiosity, the rover exploring the surface of Mars, as that would be incontrovertible proof that there is (or was!) life on our neighbouring planet in a feline form. The Curiosity rover has however found more tantalising evidence that might point to life, either existant or, more likely, now extinct1.
The “Sample Analysis at Mars” instrument on board Curiosity- a gas chromatography-mass spectrometer (GC-MS) has detected long chain alkanes, including dodecane, when analysing chemicals from a mudstone rock. This rock is in the Gale Crater near the Martian equator. While there are non-biological means of producing this molecule it could also be formed from degradation of fatty acids. Further, the researchers could not determine if dodecane itself was present in the rock sample, or if it was formed in the instrument as the sample was heated, decarboxylating a fatty acid present on Mars.
Of course dodecane is abundant on Earth too, as it is a component of oil (itself a biological residue!). Its found in kerosene, (also termed paraffin) which forms aviation fuel among many other fuel oils2.
References
-
Long-chain alkanes preserved in a Martian mudstone
Proc Natl Acad Sci USA
2025
DOI 10.1073/pnas.2420580122
-
An experimental study of n-dodecane and the development of an improved kinetic model
Combustion and Flame
2020
DOI 10.1016/j.combustflame.2019.11.014
March 2025

For many researchers working in human lipidomics, odd-chain fatty acids have been exactly that- odd! The fifteen carbon saturated fatty acid, pentadecanoic, or pentadecylic, acid has however, been the focus of several papers in the literature recently suggesting it may have a hitherto overlooked role.
Most commonly found in dairy products and the fat of ruminants it’s primarily formed by the gut bacteria of these animals. Bacteria in the rumen ferment vegetable matter and produce propionic acid, which forms the starting point for elongation1. Beginning with a three carbon starter (propionyl-CoA) forms odd-chain fatty acids, whereas mammals typically starting from acetyl-CoA which as a 2-carbon starting block, forms even chain lengths.
For a long time, its presence in humans has been used as a marker for dairy ingestion but other than that it hasn’t been considered of any importance. However, five years ago a paper proposed it could be a new essential fatty acid with roles in inflammation and metabolic conditions2. An essential fatty acid is one which is required for health and has to be provided through the diet.
The current evidence for the role of pentadecanoic acid was reviewed in Biochimie last year3. No doubt it is only a matter of time until those supplement adverts start adding C15 to their ‘high-in-omega-three’ products.
References
-
Microbial production of odd-chain fatty acids
Biotechnol Bioeng
2023
DOI 10.1002/bit.28308
-
Efficacy of dietary odd-chain saturated fatty acid pentadecanoic acid parallels broad associated health benefits in humans: could it be essential?
Sci Rep
2020
DOI 10.1038/s41598-020-64960-y
-
New insights on pentadecanoic acid with special focus on its controversial essentiality: A mini-review
Biochimie
2024
DOI 10.1016/j.biochi.2024.10.008
February 2025

Talk of snake oil and you’re selling something that’s a fraud, but talk of snake lipids is oddly rare! So as we start the Chinese year of the snake, let's explore a few serpentine sterols!
Back in 1987, the lab of Donald Downing looked at lipids in the shed skin of some bull snakes1. They found several acylglucosylcholesterol species in which a fatty acid is esterified to the 6-position of a glucose which is itself glycosidically linked to cholesterol. A palmitoyl form was one of the most common found as well as stearoyl and oleoyl.
Similar molecules had been found previously by the same lab in bird skin [ref] and in both families of organisms, it's been proposed that they have a function in maintaining the skin integrity and its waterproof character.
These sterols aren’t the only unusual lipids in snakes, much more recently, novel fatty acids were discovered in the scent glands of an endangered snake from China, the Mangshan pit viper, Protobothrops mangshanensis. Among others, was 4,6-dimethyldeca-5E-enoic acid LMFA01020453. Its function remains unknown, but a role as a pheromone has been speculated3.
As we begin the year of the snake, Gong hei fat choy 恭喜发财!
References
-
Glucosylsterol and acylglucosylsterol of snake epidermis: structure determination
J Lipid Res
1987
DOI 10.1016/S0022-2275(20)38695-8
-
Lipids of chicken epidermis
J Lipid Res
1986
DOI 10.1016/S0022-2275(20)38823-4
-
The scent gland composition of the Mangshan pit viper, Protobothrops mangshanensis
Beilstein J Org Chem
2024
DOI 10.3762/bjoc.20.222
January 2025
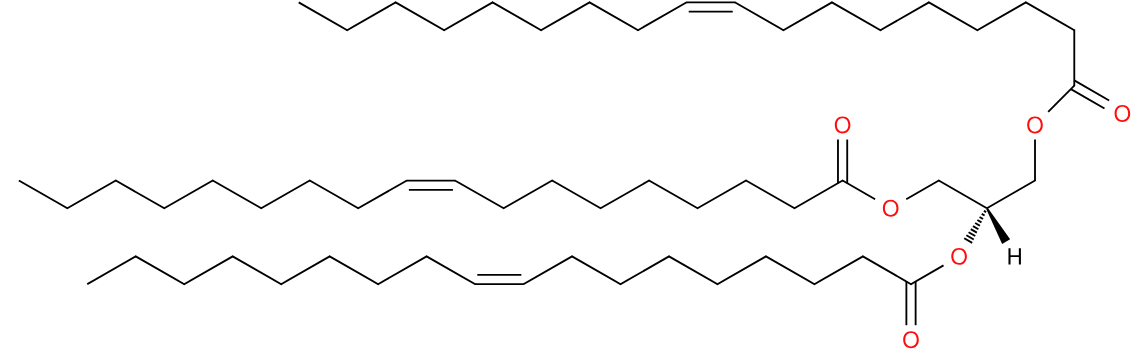
The feast of Hanukkah straddles the new year in the Gregorian calendar, its eight days ending on 2nd January. It is a celebration connected with lipids in the form of fresh olive oil. Triolein, a glyceride in which all three positions of the glycerol are esterified with oleic acid makes up a substantial proportion of olive oil, up to half of it1. This fuelled the menorah, the 7-branched lamp which burned in the temple in Jerusalem. Following the rededication of the temple by the Maccabees in the year 164 BCE, only one day’s supply was available. It miraculously lasted eight days.
Triolein is also one of the two ingredients in Lorenzo’s oil, a putative treatment for adrenoleukodystrophy2 and made famous by the eponymous film. The other ingredient is glyceryl trierucate. Adrenoleukodystrophy is caused by a failure in beta-oxidation resulting in the body being unable to remove long-chain fatty acids.
Triolein is not only a lamp fuel, foodstuff and potential therapy, it might be effective in making drill lubricant too3. An all round useful molecule!
References
-
A straightforward quantification of triacylglycerols (and fatty acids) in monovarietal extra virgin olive oils by high-temperature GC
Anal Methods
2012
DOI 10.1039/C2AY05574K
-
"Lorenzo's oil" therapy for X-linked adrenoleukodystrophy: rationale and current assessment of efficacy
J Mol Neurosci
2007
DOI 10.1007/s12031-007-0041-4
-
A novel graphene/triolein complex-based lubricant for improving high temperature water-based drilling fluid
RSC Adv
2023
DOI 10.1039/d3ra04850k
December 2024

It is that time of year when angels are much in evidence - on Christmas cards and decorations, atop trees and sung about in carols. They even have an organic acid named after them, at least indirectly as angelic acid is named after the plant from which it was first isolated, Angelica archangelica1.
Angelic acid is a five carbon, branched chain acid with a single unsaturation, 2-methyl-2Z-butenoic acid. It's the isomer of tiglic acid, also named after a plant, Croton tiglium. Angelic acid is found especially in the roots of umbelliferous plants, so if you leave a carrot out for Santa’s reindeer, or decorate a dish with candied angelica, both will contain angelic acid.
It’s described as having a spicy smell and acidic taste, so possibly far from angelic. However it is often found esterified to other compounds, typically terpenoids, such as the previous lipid of the month (beta-escin), or ingenol mebutate which is a treatment for various skin conditions2.
References
-
Ueber eine eigenthümliche flüchtige Säure aus der Angelicawurzel: Eine briefliche Mittheilung,
Justus Liebigs Ann Chem
1842
DOI 10.1002/jlac.18420420211
-
Ingenol Mebutate: Expanded Utility J Drugs Dermatol
J Drugs Dermatol
2020
DOI 10.36849/JDD.2020.4731
November 2024

Aesculus hippocastanum is not, as you might think, a spell uttered by Harry Potter, but the Latin name of the horse chestnut tree, several of which nestle between the LIPID MAPS office and the concrete brutalism of the hospital buildings beyond.
The leaves and seeds, used to play conkers in many a school yard, are so rich in soap-like molecules called saponins that they can be used to make a detergent preparation. Prominent among the saponins are the escins (or aescins, depending on your spelling) - oleanane triterpenoids substituted with sugars and short-chain fatty acids. The most abundant is beta-escin, which has a trisaccharide group as well as angelic acid attached1.
Escins are not completely angelic however! Although, the escins can do an arguably angelic job - as well as making a useful soap-substitute2, they can be medicinal and are reported to have various beneficial properties including antitumor, antiviral and anti-inflammatory actions3. However they are toxic and make all parts of the horse chestnut poisonous, unlike the sweet chestnut which is edible.
References
-
Bioactive saponins and glycosides. III. Horse chestnut. (1): The structures, inhibitory effects on ethanol absorption, and hypoglycemic activity of escins Ia, Ib, IIa, IIb, and IIIa from the seeds of Aesculus hippocastanum L.
Chem Pharm Bull
1996
DOI 10.1248/cpb.44.1454
-
Aescin - a natural soap for the formation of lipid nanodiscs with tunable size
Soft Matter
2021
DOI 10.1039/d0sm02043e
-
β-Escin: An Updated Review of Its Analysis, Pharmacology, Pharmacokinetics, and Toxicity
Am J Chin Med
2023
DOI 10.1142/S0192415X23500908
October 2024

Here in the UK, October is National Cholesterol Month, a chance to raise awareness of the importance of maintaining a healthy level of cholesterol. It’s a molecule which is vital to human health, but like much in life, it’s possible to have too much of a good thing. High levels of cholesterol can lead to heart and circulatory problems, but it is essential in cell membranes, and a precursor to the steroid hormones and vitamin D.
Cholesterol is a molecule with a long history of research, peppered with Nobel Prizes. Its molecular formula was discovered in 1888 by Reinitzer1 who was investigating the properties of what would later be called liquid crystals. While a lot of work had been done on the chemical nature of sterols2, cholesterol’s full 3D structure was not determined until 1945 when Dorothy Crowfoot Hodgkin published the crystal structure of cholesteryl iodide3. The iodine was needed as a ‘heavy atom’ to solve the complex maths of crystallography.
Almost all vertebrates synthesise cholesterol, but also absorb it in the diet. Statins, used to combat high cholesterol, are the most commonly prescribed drugs here in the UK. They work by blocking one of the enzymes at the start of the cholesterol biosynthesis pathway. A better way to control cholesterol levels is through a healthier diet containing less cholesterol.
References
-
Beiträge zur Kenntniss des Cholesterins.
Monatshefte für Chemie
1888
DOI 10.1007/BF01516710
-
Carbon skeleton of the sterols
Chem & Ind
1932
DOI 10.1002/jctb.5000512203
-
The crystal structure of cholesteryl iodide
Proc Royal Soc Chem A
1945
DOI 10.1098/rspa.1943.0040
September 2024
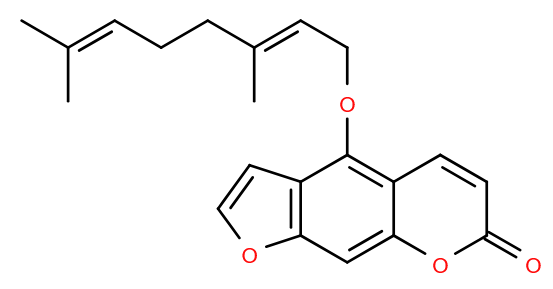
For reasons which are unimportant, your Lipid of the Month author has quite a lot of sparkling grapefruit drinks at home. Fortunately, your author is not on any medication, as certain drugs and grapefruit can be a bad combination.
The cause, possibly among others, is a molecule found in grapefruit called bergamottin, which was identified nearly 90 years ago1. It’s a furanocoumarin flavonoid linked to a geranyl chain, so really both a polyketide and a prenol combined. Along with related compounds, such as dihydroxybergamottin, it affects metabolism of many drugs, including some very common ones, for instance some used to treat high blood pressure or high cholesterol2.
It isn’t that bergamottin reacts with drugs directly, but rather it inhibits cytochrome P450 enzymes which metabolise those drugs. Drugs are administered at a dose which takes this into account, so inhibiting the enzyme can effectively lead to overdosing3. Alternatively, if the drug is administered in a pre- form, relying on enzymes to convert it to the active form, under dosing can occur as the active drug isn’t produced in the expected amount.
That’s not to say that grapefruit is bad for you, it’s a source of vitamin C and there’s even a suggestion that bergamottin itself has potential as an anticancer agent4. Your author will continue enjoying the beverage- in moderation, obviously. Cheers!
References
-
Uber Bergamottin und über die Auffindung von Limettin im Bergamottol (XXXIV. Mitteil. uber naturliche Cumarine)
Berichte der Deutschen chemischen Gesellschaft
1937
DOI 10.1002/cber.19370701115
-
The effects of fruit juices on drug disposition: a new model for drug interactions
Eur J Clin Invest
2003
DOI 10.1046/j.1365-2362.33.s2.2.x
-
Grapefruit–medication interactions: Forbidden fruit or avoidable consequences?
CMAJ
2013
DOI 10.1503/cmaj.120951
-
Pharmacological Utilization of Bergamottin, Derived from Grapefruits, in Cancer Prevention and Therapy
Int J Mol Sci
2018
DOI 10.3390/ijms19124048
August 2024
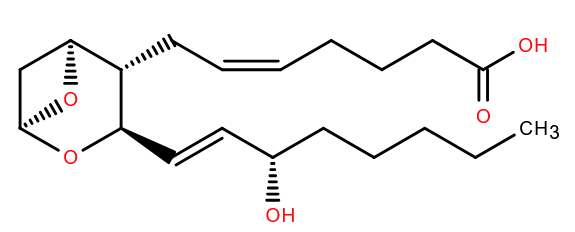
Our Lipid of the Month article in April 2021 revealed what a dangerous fruit the avocado is, and if you cut yourself while preparing one you trigger a process that involves many many molecules. These include protein clotting factors but also lipids, among them Thromboxane A2 (TXA2), characterized by Bengt Samuelsson[1], who died recently at the age of 90. It was named thromboxane as it was formed in thrombocytes (platelets) and contains an oxane ring. Samuelsson was awarded the 1982 Nobel Prize for his work on prostaglandins and other molecules derived from arachidonic acid, including TXA2. He continued to publish throughout his life and his most recent paper in PubMed was only last year[2].
TXA2 is formed from Prostaglandin H2 (PGH2) by the enzyme thromboxane synthase but is unstable- having a half life in vitro of only around 30 seconds before becoming Thromboxane B2. In that time however it is able to cause aggregation of platelets and constrict blood vessels around the wound by activation of the thromboxane receptor.
Not only can PGH2 form TXA2, but is also the precursor for several other eicosanoids including prostaglandins, leukotrienes and prostacyclin, several of which were discovered or studied by Samuelsson. His work has led to drugs not only to affect the clotting cascade, but in other diverse areas of medicine too[3]..
References
-
Thromboxanes: a new group of biologically active compounds derived from prostaglandin endoperoxides
Proc Natl Acad Sci USA
1975
DOI 10.1073/pnas.72.8.2994
-
Modulation of the 5-Lipoxygenase Pathway by Chalcogen-Containing Inhibitors of Leukotriene A4 Hydrolase
Int J Mol Sci
2023
DOI 10.3390/ijms24087539
-
Role of basic science in the development of new medicines: examples from the eicosanoid field
J Biol Chem
2012
DOI 10.1074/jbc.X112.351437
July 2024
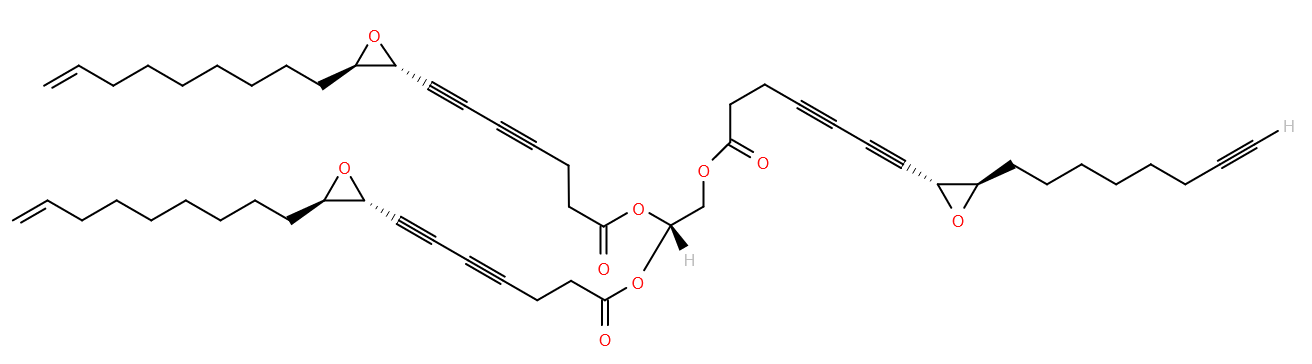
Among the more unusual molecules in our structure database is undoubtedly Lycogaride C1, a triglyceride found in wolf’s milk. That’s not, we should point out, the substance which suckled Romulus, the mythical founder of Rome, but rather it’s the slime mold Lycogala epidendrum, commonly called wolf’s milk.
Lycogaride C, or 1,2-(8R,9R-epoxy-17-octadecen-4,6-diynoyl)-3-(8R,9R-epoxy-heptadec-4,6,16-triynoyl)-sn-glycerol, has a number of unusual features, at least from a human perspective, but perhaps not usual for a slime mold lipid! It contains seven triple bonds distributed between the three fatty acids- the most triple bonds of any molecule in LMSD to date. Each fatty acid also contains an epoxy group within the chain.
It’s not the only ‘unusual’ lipid in L.epidendrum- several other lycogarides with similar structures have been identified2. But what role do these strange lipids play in the physiology of the organism? Little research seems to have been done, but it seems they inhibit germination of rice. Whether that is its purpose remains to be seen.
References
-
Three novel polyacetylene triglycerides, Lycogarides A-C, from the Myxomycetes Lycogala epidendrum
Chem Pharm Bull
1994
DOI 10.1248/cpb.42.1531
-
Acylglycerols from the slime mould, Lycogala epidendrum
Phytochemistry
1996
DOI 10.1016/0031-9422(95)00664-8
June 2024
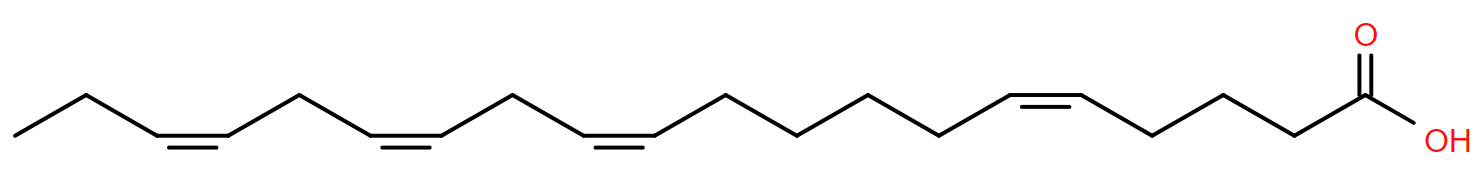
The month of June and the juniper tree can both trace their etymology to youth, though the poet Ovid suggests maybe June’s naming is ambiguous1. Juniper means youth producing, presumably because of its evergreen nature rather than any elixir of life it may contain.
One compound juniper does contain is named after the plant. Juniperonic acid, found in the seeds of juniper and other plants2, is a twenty carbon fatty acid with four double bonds, making it isomeric with arachidonic acid, perhaps more familiar to those studying human lipids. Both have a species level shorthand of FA 20:4, but while arachidonic acid has the double bonds each three carbons apart, at carbons 5,8,11 and 14, juniperonic acid has them at 5,11,14 and 17. It is ‘non-methylene interrupted’.
Juniperonic acid is not confined to the plant world, it has been found in humans too3 though whether endogenously formed or from the diet is unknown. It may even be a substrate for mammalian production of an essential fatty acid4.
Given the several forms of 20:4 fatty acid existing in nature, assuming such a molecule in a human sample will be arachidonic acid may not be valid. June might be named after Juno or Juventus, but FA 20:4 could have an even more elusive identity on which Ovid is understandably silent.
References
-
Ovid’s Fasti
Loeb Classical Library
8
DOI 10.4159/DLCL.ovid-fasti.1931
-
Positional Distribution of delta5-Olefinic Acids in Triacylglycerols from Conifer Seed Oils: General and Specific Enrichment in the sn-3 Position
J AOCS
1997
DOI 10.1007/s11746-997-0174-1
-
Ozone-enabled fatty acid discovery reveals unexpected diversity in the human lipidome
Nat Commun
2023
DOI 10.1038/s41467-023-39617-9
-
Metabolic Conversion of C20 Polymethylene-Interrupted Polyunsaturated Fatty Acids to Essential Fatty Acids
Lipids
2014
DOI 10.1007/s11745-014-3896-5
May 2024
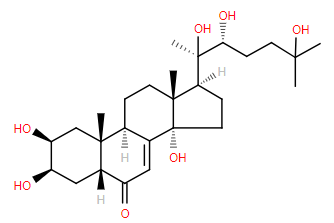
This spring, as the soil warms up across the US, a sterol hormone will play a part in a truly amazing sight. Hundreds of thousands of periodical cicada nymphs will emerge from the ground, and, influenced by 20-hydroxyecdysone, moult out of their skin to become adults.
20-hydroxyecdysone is a sterol molecule first characterised in 19661. It is formed from cholesterol and controls moulting (ecdysis) in arthropods, including lobsters and insects like the periodical cicada. It binds to the ecdysone receptor, a transcription factor in the nucleus of cells, to regulate gene expression2.
Insects generate a range of ecdysteroiods similar to 20-hydroxyecdysone, and many plants also produce the same, or similar molecules [ref]. These must be as a deterrent against arthropods eating the plants, as a significant amount of moulting hormones in the insect’s diet will undoubtedly alter their physiology.
Some periodical cicadas spend 13 years underground as nymphs, others spend 17 years. These prime numbers mean they only appear together every 221 years. As the last time this happened was in 1803, 2024 is expected to be a huge year for cicadas and the hormones that drive their metamorphosis.
References
-
20-hydroxy-ecdyson, isoliert aus insekten
Tetrahedron
1966
DOI 10.1016/S0040-4039(01)99901-0
-
The Evolution of Insect Metamorphosis
Curr Biol
2019
DOI 10.3390/ijms23158664
-
Phytoecdysteroids: Distribution, Structural Diversity, Biosynthesis, Activity, and Crosstalk with Phytohormones
Int J Mol Sci
2022
DOI 10.3390/ijms23158664
April 2024

For those celebrating Easter at the moment, even in the most non-religious way, there’s likely to be quite a lot of egg-shaped chocolate consumed, and with it, April’s Lipid of the Month, oleoylethanolamine (oleoyl-EA).
Oleoyl-EA is a fatty acid derivative, related to the endocannabinoid anandamide, and produced endogenously in mammals and other vertebrates. In rats, administration of oleoyl-EA led to appetite suppression and thus a decreased food intake of the animal1.
It’s known that oleoyl-EA binds to a transcription factor called peroxisome proliferator-activated receptor-alpha (PPARalpha) which is involved in aspects of lipid metabolism. Exactly how this results in a lessened appetite though remains to be seen.
Oleoyl-EA is also found in the chocolate2, but will eating chocolate keep you thin? Well, one study suggests that there isn’t a sufficient amount present to have any effect3, and our own real-world experience gives us the answer too!
References
-
An anorexic lipid mediator regulated by feeding
Nature
2001
DOI 10.1038/35102582
-
Brain cannabinoids in chocolate
Nature
1996
DOI 10.1038/382677a0
-
Trick or treat from food endocannabinoids? Nature 1998 10.1038/25267
Nature
1998
DOI 10.1038/25267
March 2024
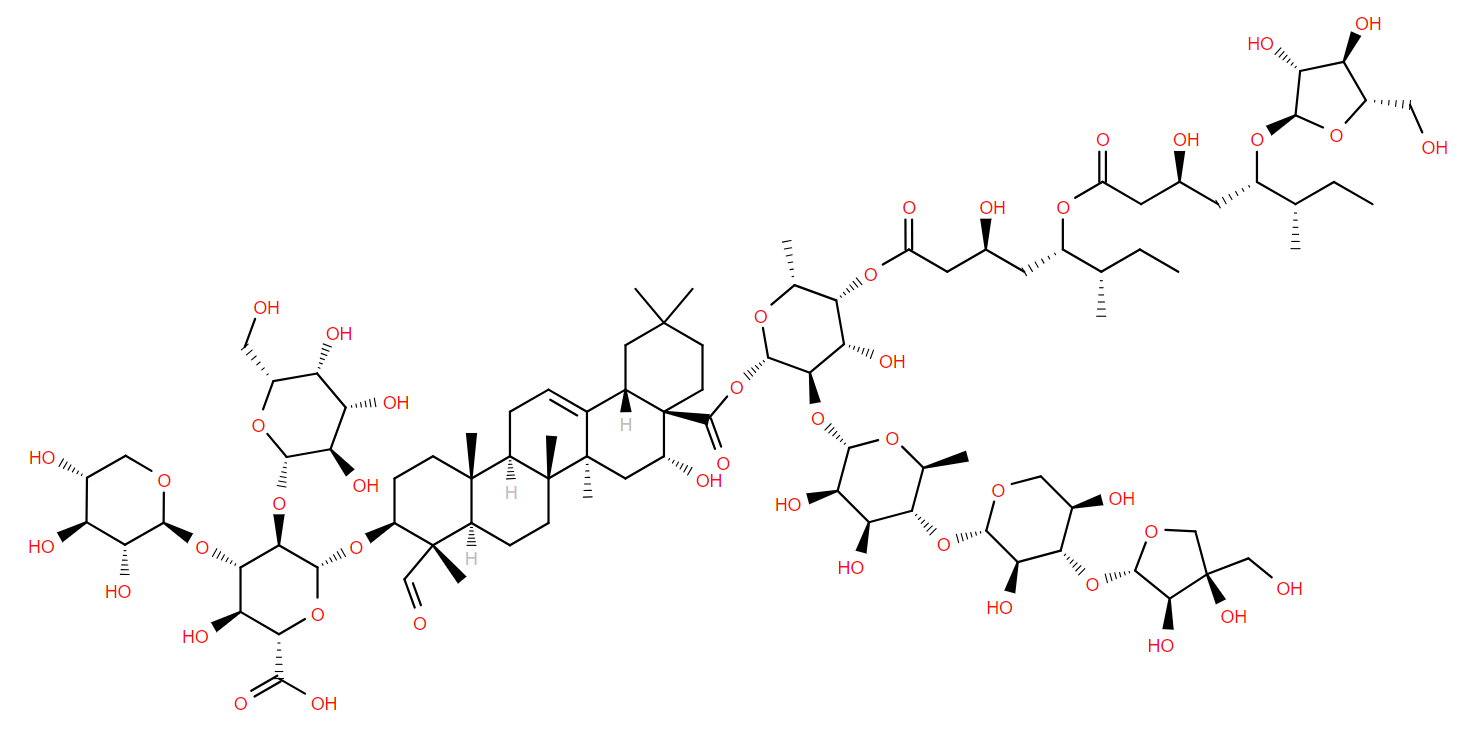
Among the more complex molecules in our main database, LMSD, are two that go by the enigmatic name of QS-21. Found in the soap bark tree (Quillaja saponaria), native to Chile, they are heavily glycosylated derivatives of quillaic acid. They differ only in one of the sugar groups, apiose or xylose. In addition to the sugars, QS-21 also contains two hydroxy anteiso-octanoic acids, linked as in an estolide1.
QS-21 is an effective vaccine adjuvant- a substance added to a vaccine that triggers a more effective immune response. It is used in, or being evaluated for, vaccines for shingles, HIV, COVID and some cancers.
But the demand for QS-21 causes a problem; chemical synthesis of such a complex molecule is a challenge and the process of harvesting the natural source unfortunately kills the tree. This is neither environmentally sound, nor a sustainable process for QS-21 production long term.
Recently, two groups have reported producing QS-21 without harm to native trees2,3. One has successfully cultured Q. saponaria cells, which produce QS-21 naturally. The other cloned genes for a 20-step biosynthetic process from into tobacco and produced QS-21 transgenically.
Both these works show that the future of producing QS-21 sustainably, to aid the effectiveness of vaccines, is looking bright.
References
-
Structural and Immunological Characterization of the Vaccine Adjuvant QS-21
Vaccine Design
1995
DOI 10.1007/978-1-4615-1823-5_22
-
Complete biosynthesis of the potent vaccine adjuvant QS-21
Nat Chem Biol
2024
DOI 10.1038/s41589-023-01538-5
-
Chemical and biological characterization of vaccine adjuvant QS-21 produced via plant cell culture
iScience
2024
DOI 10.1016/j.isci.2024.109006
February 2024
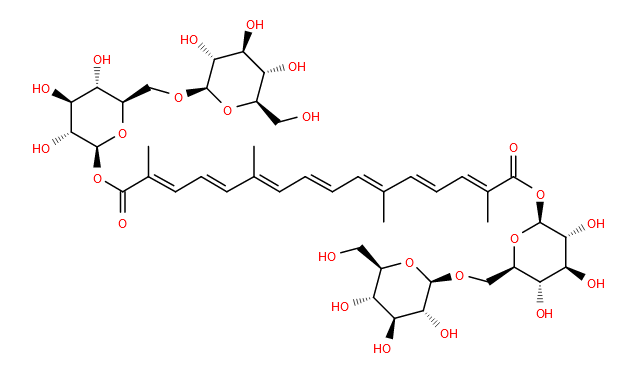
The crocus starting to flower in the gardens around LIPID MAPS HQ are a sure sign that, at least here in Cardiff, spring is around the corner.
Within the flowers is the molecule crocin. This is a yellow-red pigment responsible for the colour of the spice saffron, made from the flowers of certain crocus species.
The structure of crocin was determined nearly 100 years ago1 as a glycosylated form of the apocarotenoid crocetin. Crocetin is termed an apocarotenoid as its synthesized from a 40 carbon tetraterpene carotenoid which is then cleaved to make it smaller2. In the case of crocetin, to a 20 carbon molecule, which subsequently has 2 glucose units added at each end to make crocin. This of course poses a challenge for classification- is crocetin a tetraterpene, because it started out that way, or a diterpene because it ends up with only 20 carbons? Further, if one did not know the biosynthetic route, how could one tell?
In addition to its colourant and taste properties, many pharmacological functions have been attributed to crocin and saffron more generally. These include anti-hypertensive, anti-dementia, anti-cancer and even aphrodisiac properties3, 4! Saffron is harvested from a specific type of crocus however, and most of the crocus plant is poisonous. If you see these plants flowering in the early springtime, please don’t eat them!
References
-
Pflanzenfarbstoffe XV. Der Zucker des α-Crocins,
Helv Chim Acta
1929
DOI 10.1002/hlca.192901201103
-
Oxidative remodeling of chromoplast carotenoids: identification of the carotenoid dioxygenase CsCCD and CsZCD genes involved in Crocus secondary metabolite biogenesis
Plant Cell
2003
DOI 10.1105/tpc.006536
-
A review of therapeutic impacts of saffron (Crocus sativus L.) and its constituents
Physiol Rep
2023
DOI 10.14814/phy2.15785
-
The effect of saffron, Crocus sativus stigma, extract and its constituents, safranal and crocin on sexual behaviors in normal male rats
Phytomedicine
2007
DOI 10.1016/j.phymed.2007.09.020
January 2024
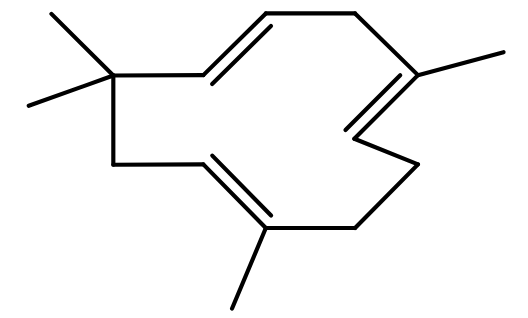
It is the time of year when many make resolutions, often to lose weight, certainly that which may have been gained over the festive period. Your favourite search engine will find no shortage of sites extolling the benefits of various drugs or diets to achieve this. One of them is humulene, a 15-carbon sesquiterpene claimed to be an appetite suppressant. A search of Pubmed for the same however does not suggest that much research has been done to substantiate this claim.
Humulene, also called alpha-caryophyllene, is formed from farnesyl diphosphate. It is found in many plants, notably hops, Humulus lupulus, after which it is named. Humulene and its metabolites are responsible for the ‘hoppy’ flavour of beers in which hops are used as an ingredient1. Consuming humulene in beer will certainly not be an effective weight-loss strategy!
Due to the apparent paucity of scientific studies on the alleged weight-loss properties of humulene, if your New Year resolution is to get trimmer so people remark “Hmm, you lean!” perhaps humulene, especially in beer, is not the way forward.
References
-
Chemical transformations of characteristic hop secondary metabolites in relation to beer properties and the brewing process: a review
Food Chem
2015
DOI 10.1016/j.foodchem.2014.09.139
December 2023

It’s the time of year when theories regarding the famously red nose of Santa’s lead reindeer, Rudolf, come to the fore. Some suggest a dense network of blood vessels1, but a more lipid-centric cause seems to have been overlooked.
The carotenoid lycopene is a deep red colour and found in many vegetables, including carrots which children in some countries traditionally leave for Rudolf on Christmas eve.
Lycopene is a 40 carbon tetraterpenoid made up of eight isoprene units with 11 conjugated double bonds that absorb light to give the molecule its colour. In plants it is an accessory pigment to chlorophyll in photosynthesis, and in fruits such as tomatoes it signals the fruit’s ripeness to animals which eat the fruit and disperse the seeds. Lycopene is a precursor to other carotenoids such as carotene, for which carrots are more well known. Carotene is metabolised to make the visual pigment retinal, but the saying that eating carrots will help one see in the dark has little evidence to support it. Night-vision would undoubtedly be important for a nocturnally aviating cervid however.
Perhaps the many children leaving carrots for Rudolf don’t realise that carrots aren’t part of the natural diet of the reindeer. The carrot, Daucus carota, was domesticated in Asia2, but Rangifer tarandus the reindeer, is only partially domesticated3 and native to the Arctic region. The species may be unable to handle such an annual abundance of lycopene and one must consider that Rudolf may have a genetic condition whereupon the excess accumulates in his muzzle. Presumably this does not cause any health issues as he and his companions are over 80 years old and remain able to tour the globe in one evening. Indeed a navigational nose has clearly been advantageous to this particular individual!
References
-
Why Rudolph's nose is red: observational study
BMJ
2012
DOI 10.1136/bmj.e8311
-
Population genomics identifies genetic signatures of carrot domestication and improvement and uncovers the origin of high-carotenoid orange carrots
Nat Plants
2023
DOI 10.1038/s41477-023-01526-6
-
Investigating the domestication and early management of reindeer (Rangifer tarandus) in the Sámi archaeological context from teeth geometric morphometrics Sci Rep 2023 10.1038/s41598-023-33422-6.
Sci Rep
2023
DOI 10.1038/s41598-023-33422-6
November 2023
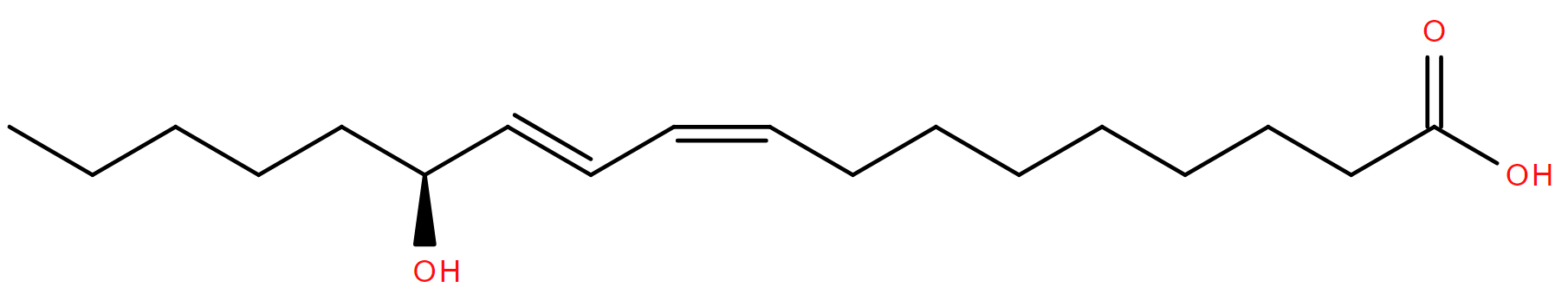
Throughout this month, men worldwide will be seen with facial hair they usually don’t have. It will be displayed with varying levels of sartorial elegance in support of Movember- a campaign to raise awareness of men’s health issues including diseases such as prostate cancer.
As with other cancers, lipids play a role in prostate cancer biochemistry with tumours having a disregulated lipid metabolism in general1. One of the lipids which is produced in larger amounts than normal is 13S-hydroxy-9Z,11E-octadecadienoic acid, or 13S-HODE.
13S-HODE is elevated due to increased expression of 15-LOX which converts linoleic acid to 13S-HpODE. This is then reduced to 13S-HODE. The amount of 15-LOX correlates with the proliferation of the cancer2.
13S-HODE is a signaling molecule, which binds to peroxisome proliferator-activated receptors (PPAR) and alters their activity in controlling gene expression. The genes under PPAR control include, among others, some of those related to tumorigenesis and lipid metabolism.
A major source of linoleic acid in our diet is vegetable oil, so it’s unsurprising that 13-HODE is present in these oils too. In fact it is also named coriolic acid after the coraria seed oil from which it was isolated3. In plants 13-HODE is an intermediate on the pathway to jasmonic acids- plant signalling hormones which are structurally similar to the prostaglandins.
This month, if you see a man with a moustache that really doesn’t suit him, perhaps he is sporting it to highlight men’s health and, very indirectly, the lipids involved in it.
References
-
Genetics of lipid metabolism in prostate cancer
Nat Genet
2018
DOI 10.1038/s41588-017-0037-0
-
Concordant induction of 15-lipoxygenase-1 and mutant p53 expression in human prostate adenocarcinoma: correlation with Gleason staging
Carcinogenesis
2000
DOI 10.1093/carcin/21.10.1777
-
Structure and intraglyceride distribution of coriolic acid
Lipids
1968
DOI 10.1007/BF02531282
October 2023

The prostaglandins are generally formed from arachidonic acid, liberated from phospholipids in the membrane. In contrast, the prostaglandin ethanolamides, or prostamides, are formed from a different starting point, but via the same enzymatic pathway. Rather than adding an ethanolamine to the acidic part of the prostaglandin (or precursor), the cyclooxygenase enzymes act on anandamide.
Prostaglandin F2α ethanolamide, otherwise known as prostamide F2α, or PGF2alpha-EA
is perhaps the most well studied of the prostamides. It has been implicated in the pain response to inflammation in mice1. Synthetic analogues such as bimatoprost are used to treat glaucoma2 and very recently, a paper has suggested a new role for prostamde F2α3- it stimulates preadipocyte proliferation in white adipose tissue.
No doubt the full role of these lipids in human health is yet to be determined.
References
-
Discovery of prostamide F2α and its role in inflammatory pain and dorsal horn nociceptive neuron hyperexcitability
PLoS One
2012
DOI 10.1371/journal.pone.0031111
-
Promising alternative clinical uses of prostaglandin F2α analogs: beyond the eyelashes
J Am Acad Dermatol
2015
DOI 10.1016/j.jaad.2014.10.012
-
New role for the anandamide metabolite prostaglandin F2α ethanolamide: Rolling preadipocyte proliferation
J Lipid Res
2023
DOI 10.1016/j.jlr.2023.100444
September 2023
This summer many places in the northern hemisphere have broken records. Extreme temperatures in excess of 40 degrees C (104 F) have been recorded in parts of Europe, North America and China. A great deal of sweat has been sweated!
In males, one component of that sweat is androstadienone, a steroid secreted particularly in the armpits which acts as a pheromone - a molecule released into the environment which affects the behaviour of others, usually by smell.
Smelling androstadienone has been shown to alter the level of the hormone cortisol in females,1 a stress-response hormone, and cause a range of changes in behaviour. Its scent has been reported to alter the perception of what emotion a face is showing, modulate aggression, make men less cooperative (but also more cooperative!) and women more generous and relaxed.2-6 As with any pheromone, the smell might not be consciously ‘smelled’ for a molecule to influence behaviour. To add to the confusion, not everyone agrees that androstadienone is a pheromone at all!8
But, whether or not your scent alters the behaviour of those around you, in hot weather deodorant is probably a good idea.
References
-
Smelling a Single Component of Male Sweat Alters Levels of Cortisol in Women
J Neurosci
2007
DOI 10.1523/JNEUROSCI.4430-06.2007
-
Olfaction in the Multisensory Processing of Faces: A Narrative Review of the Influence of Human Body Odors
Front Psychol
2021
DOI 10.3389/fpsyg.2021.750944
-
Androstadienone modulates human aggression in a sex-dependent manner
Soc Cogn Affect Neurosci
2023
DOI 10.1093/scan/nsad006
-
Androstadienone, a Chemosignal Found in Human Sweat, Increases Individualistic Behavior and Decreases Cooperative Responses in Men
Chem Senses
2018
DOI 10.1093/chemse/bjy002
-
A putative human pheromone, androstadienone, increases cooperation between men
PLoS One
2013
DOI 10.1371/journal.pone.0062499
-
The Putative Chemosignal Androstadienone Makes Women More Generou
J Neurosci Psychol Econ
2016
DOI 10.1037/npe0000055
-
Behavioral and electrophysiological effects of androstadienone, a human pheromone
Psychoneuroendocrinology
2000
DOI 10.1016/S0306-4530(99)00056-6
-
Reproducible research into human chemical communication by cues and pheromones: learning from psychology's renaissance
Philos Trans R Soc Lond B Biol Sci
2020
DOI 10.1098/rstb.2019.0262
August 2023

Cut open a jagua fruit and in a few seconds blue veins begin to form in the white flesh. An iridoid monoterpene called genipin is responsible for the unusual colour. The fruit, from Genipa americana, a plant related to coffee, is native to tropical forests in South America. It is used by indigenous people as a dye to paint the skin.
Genipin forms the blue colour when it polymerises and reacts with amines in the presence of oxygen1. It is of great interest to the food industry as natural blue pigments are rare and difficult to obtain. In fact more than one patent for this use of genipin has been filed. It also has potential uses as a therapeutic agent with suggestions that it may be active against cancer, diabetes, have antiviral and anti-allergy properties2. Whether all, or any, of these properties translate to the clinic remains to be seen. Of course there are ethical and ecological issues around large-scale genipin production from either farmed, or wild-harvested jaguar fruit3.
The iridoid monoterpene family is named after the first described member Iridomyrmecin, found in Iridomyrmex, or rainbow ants. The iridoid family is found in plants and some insects, though these may be obtained through their diet. While at first glance, iridoids might not seem to be lipids, they are formed from geranial, a common prenol precursor, by cyclisation of the linear monoterpene oxo-geranial4.
References
-
Studies on the Blue Pigments Produced from Genipin and Methylamine. I. Structures of the Brownish-Red Pigments, Intermediates Leading to the Blue Pigments
Chem Pharm Bull
1994
DOI 10.1248/cpb.42.668
-
Novel Findings regarding the Bioactivity of the Natural Blue Pigment Genipin in Human Diseases
Int J Mol Sci
2012
DOI 10.3390/ijms23020902
-
Natural blue
Science
2023
DOI 10.1126/science.adj2001
-
An alternative route to cyclic terpenes by reductive cyclization in iridoid biosynthesis
Nature
2012
DOI 10.1038/nature11692
July 2023
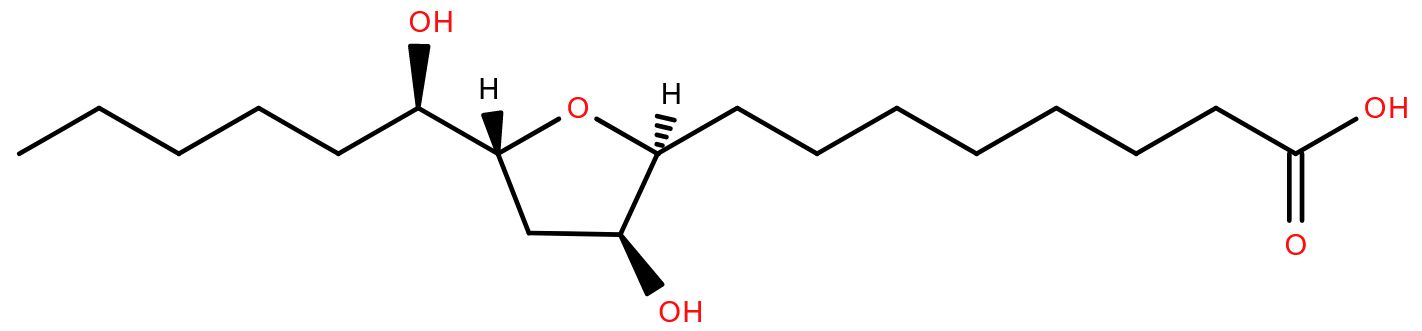
Several species of fish travel from the sea to freshwater rivers in order to spawn. Famously, salmon and eels do so, but the lamprey, a ‘primitive’ jawless fish, is also anadromous (the term for a fish moving from salt to freshwater to complete its life cycle). Lamprey larvae live in rivers for many years before migrating out to sea where they feed by parasitizing fish. Once mature, they return to the rivers to reproduce.
An adult lamprey finds its way to a suitable stream by smell. It detects a lipid-based pheromone called petromyric acid A1which is secreted by larval lampreys and washed downstream in the current. Adults sensing this hydroxy fatty acid containing a tetrahydrofuran ring know that the river is a good breeding ground, because it already has lots of baby lampreys living in it.
Lampreys have been shown to respond to petromyric acid A at concentrations as low as 10-11 M and there is a possibility that it could be used to control lamprey populations in places such as the Great Lakes where they are considered an invasive species.
References
-
Fatty-acid derivative acts as a sea lamprey migratory pheromone
Proc Natl Acad Sci USA
2018
DOI 10.1073/pnas.1803169115
June 2023
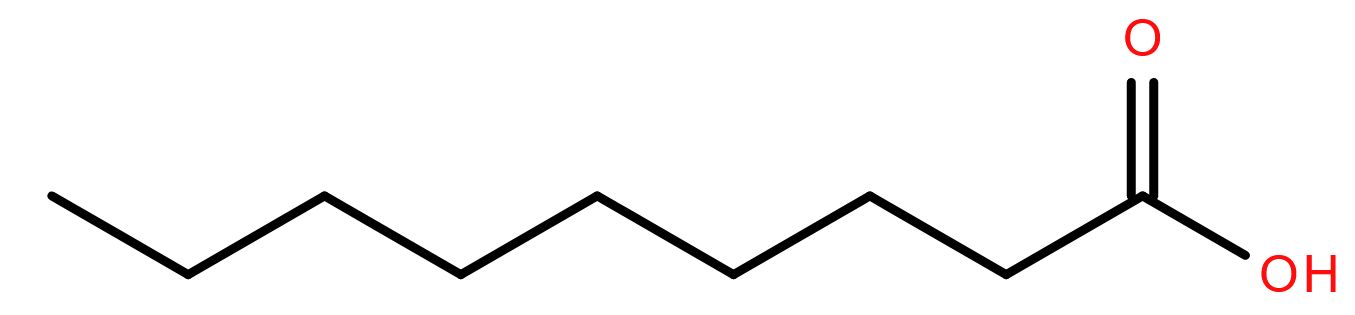
With summer in full swing in the northern hemisphere (a whole week without rain here in Cardiff!) plants are growing prolifically. Some are in the wrong place and need killing off.
In the 1990s a weedkiller was approved with a lipid active ingredient, pelargonic acid. This nine-carbon fatty acid is a ‘safe’ herbicide as it is not toxic to humans and does not persist in the ground. It’s a ‘burn-down’ chemical so called because it only kills the foliage, causing it to look burned. It doesn’t kill the roots of the plant.
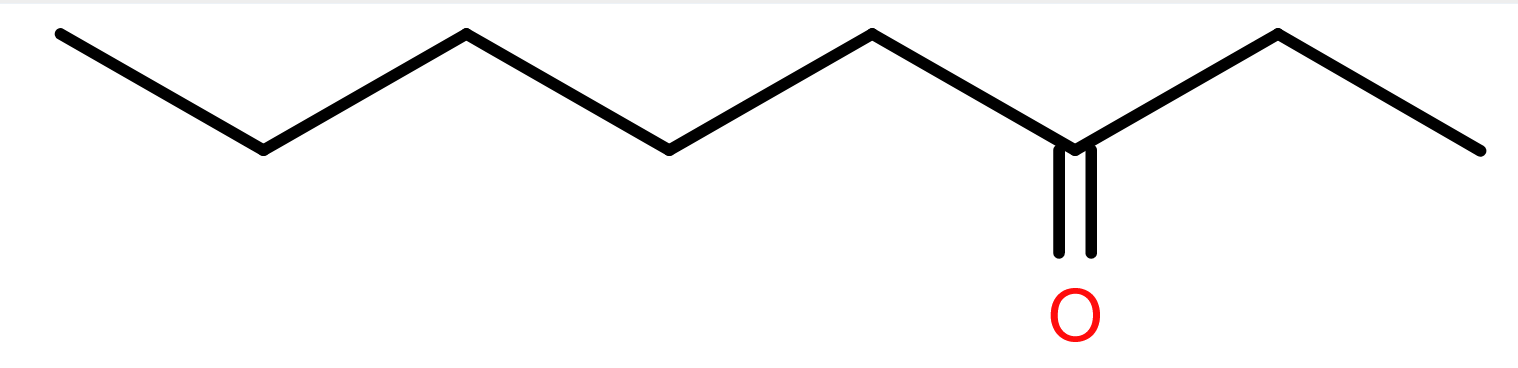
Pelargonic acid (or nonanoic acid to give it its formal name) is so called as it was first isolated in a mixture of its esters from pelargonium plants1. That mixture shared the scent of pelargoniums- non-hardy plants often called geraniums and very popular in flower borders. Pelargonic acid is also found naturally in fruits and dairy produce.
Pelargonic acid is thought to act essentially as a detergent, causing permeability of cell membranes and those of organelles. This liberates chlorophyll which, as a powerful oxidising agent, reacts with many cellular components, including membrane lipids, and damages them2. That would explain why it works best on sunny days and in warm weather. Fortunately, those weather conditions, at least at the moment, are in ready supply.
References
-
On a common origin of the acids (CH)n O4 with a boiling-point under 300° centigrade
Mem. Proc. Chem. Soc
1845
DOI 10.1039/MP8450300235
-
Phytotoxic activity of middle-chain fatty acids II: peroxidation and membrane effects
Pesticide Biochem Physiol
2004
DOI 10.1016/j.pestbp.2004.06.010
May 2023

Undoubtedly one of the most talked-about events this month will be the coronation in London of King Charles III in a ceremony where lipids play a central role. Since before recorded history, kings, queens, and other leaders have been anointed with perfumed oils as part of their investiture rituals.
The recipe for the oil to anoint British monarchs has been unchanged for hundreds of years, however for Charles III, some ingredients will be missing. It will lack civetone (from the glands of the civet cat) and ambergris. The latter is a waxy substance, the origins of which have been the subject of (often wild) speculation for centuries. It’s now known to be formed in the guts of sperm whales1.
The main lipid of ambergris is ambrein, a triterpene which breaks down to give various scented molecules. Ambergris has been prized in the perfume industry for centuries and has traditionally been added to the base olive oil (containing triglycerides like triolein) along with other richly perfumed ingredients for the coronation.
Ambergris is rare, sometimes found floating in the sea, or washed up on beaches having been (presumably) excreted by the whale. Exactly how a sperm whale synthesises ambrein is unknown. It almost certainly starts from squalene and there is evidence that bacterial enzymes (likely via the whale’s gut microbiota) play a part2. The unknown nature of ambrein’s synthesis only adds to the allure of ambergris.
As Herman Melville comments in Moby Dick “Who would think, then, that such fine ladies and gentlemen should regale themselves with an essence found in the inglorious bowels of a sick whale!”. This month, it won’t be Britain’ new monarch!
References
-
Ambergris: the search for its origin
Isis
1982
DOI 10.1086/353040
-
Biosynthesis of ambrein in ambergris: evidence from isotopic data and identification of possible intermediates
Nat Prod Res
2021
DOI 10.1080/14786419.2019.1644630
April 2023
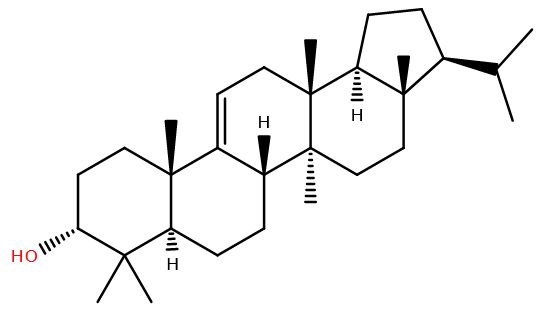
Think of a fossil, and likely you’ll picture an ammonite peeking out of a rock, or maybe that huge T. rex skeleton in a museum. But life leaves chemical fossils behind too.
Arborinol, is a pentacyclic triterpenoid named after the shrub Glycosmis arborea (now called Glycosmis pentaphylla) whose role in vivo seems yet to be deciphered. It’s one of many triterpenoids made by flowering plants, which remain after a plant decomposes. These are often converted to arborane which is detectable in rocks today. Thought to be found only in plants, the presence of arborane compounds was seen as a marker for their existence at the time the rock was soil at the earth’s surface. The isomer of arborinol, isoarborinol has even been found intact in rock which is 50 million years old1.
The discovery that bacteria can also synthesise arborane molecules2 means that arborane cannot be used exclusively as a marker for flowering plants, and solves a mystery of arborane in rocks which pre-date the origin of plants.
The structure of arborinol was solved using x-ray crystallography in 1965 by Olga Kennard3. Kennard had the vision that bringing together molecular data in a central database would be vital for science and the whole would be greater than the sum of the parts. She was director of the Cambridge Crystallographic Data Centre for many years and was also instrumental in founding several other databases, including PDB and ENA. By pioneering the archiving of so much chemical data in central repositories, Dr Kennard, who died last month aged 98, enabled a vast number of scientists to use these data. Her contribution to science is incalculable.
References
-
Triterpene alcohol isolation from oil shale
Science
1969
DOI 10.1126/science.163.3872.119
-
Synthesis of arborane triterpenols by a bacterial oxidosqualene cyclase
Proc Natl Acad Sci USA
2016
DOI 10.1073/pnas.1617231114
-
The complete structure of the triterpene arborinol
Tetrahedron Letts
1965
DOI 10.1016/s0040-4039(01)89324-2
March 2023
With the possible exception of cholesterol, it’s not often for a lipid to be in the news, but octan-3-one
managed this recently thanks to its apparent nematicidal role in the oyster mushroom.
Predictably headlines varied from the measured “The mushroom that is both delicacy and predator” to the somewhat less measured “Killer mushroom releases fungus ‘nerve gas’ to paralyse victims before ‘rapid death’”.
The oyster mushroom, Pleurotus ostreatus is a delicacy in some parts of the world, naturally living on decaying wood which is poor in nitrogen. It’s known to kill nematodes, and ingest them as a source of nitrogen. A recent paper in Sci Adv1 has identified octan-3-one as the compound responsible for dispatching the nematodes. Released from fragile structures called toxocysts, octan-3-one causes paralysis and cell death in the nematode by disrupting cell membrane integrity.
Octan-3-one is not unique to P. ostreatus, it is a volatile, scented molecule used as a pheromone in insects and plants2 and found in other fungi too. A question that remains to be answered though, is how the toxocyst stores a compound which disrupts nematode cell membranes, without causing damage to its own.
References
-
A carnivorous mushroom paralyzes and kills nematodes via a volatile ketone
Sci Adv
2023
DOI 10.1126/sciadv.ade4809
-
Freshly Distilled Oil of the Leaves of Rasmarinus Officianalis L Contained 3-Octanone
Z Naturforsch
1978
DOI 10.1515/znc-1978-1-226
February 2023
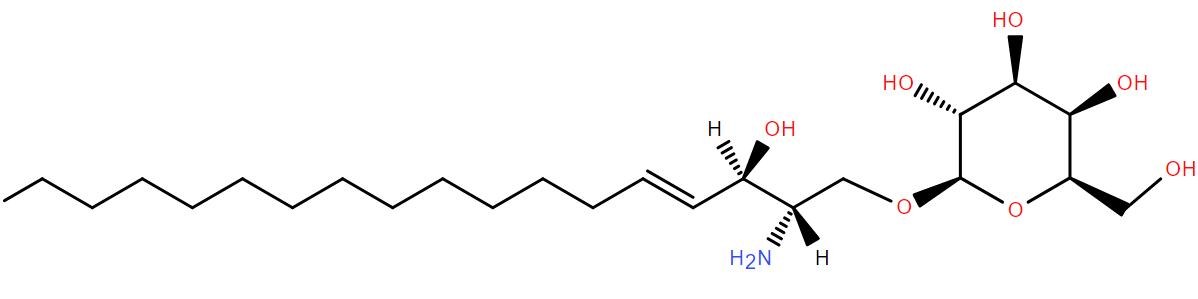
Over 100 years ago, Krabbe and Copenhagen described a rare inherited neurological condition which would become known as globoid cell leukodystrophy, or Krabbe disease1. It’s now known to be characterized by an accumulation of the lipid psychosine in the membranes of cells such as Schwann cells and oligodendrocytes in the nervous system. It leaves these cells unable to provide the myelination around nerve axons which is necessary for nerve function.
Psychosine accumulates due to a deficiency in an enzyme encoded by the GALC gene. The most common mutations in GALC cause a form of Krabbe disease which
is usually fatal in infancy, though some mutations result in an
adult-onset phenotype2. GALC encodes galactocerebrosidase which ordinarily cleaves the galactose headgroup from galactoceramides (for example, from (GalCer d18:1/16:0) in their degradation pathway. In its absence, the acyl chain of the molecule can be removed by acid ceramidase, leaving galactosyl-sphingosine, or psychosine, behind.
Evidence suggests that build up of psychosine, essentially ‘lyso’ galactosylceramide, disrupts the properties of the membrane3 (ref) which may contribute to the disease phenotype.
While Krabbe disease was described in 1916, psychosine was described much earlier. It was mentioned by Thudichum in 1884 in his treatise on the chemical constitution of the brain. Presumably it was named after its isolation from brain extracts (psycho- referring to the mind). The link between psychosine and Krabbe disease was made fifty years ago in 19724.
References
-
A New Familial, Infantile Form Of Diffuse Brain-Sclerosis
Brain
1916
DOI 10.1093/brain/39.1-2.74
-
A novel compound heterozygous mutation in GALC associated with adult-onset Krabbe disease: case report and literature review
Neurogenetics
2022
DOI 10.1007/s10048-021-00682-1
-
Psychosine, the cytotoxic sphingolipid that accumulates in globoid cell leukodystrophy, alters membrane architecture
J Lipid Res
2013
DOI 10.1194/jlr.M039610
-
Globoid cell leukodystrophy: Additional deficiency of psychosine galactosidase
Biochem Biophys Res Commun
1972
DOI 10.1016/0006-291X(72)90381-6
January 2023
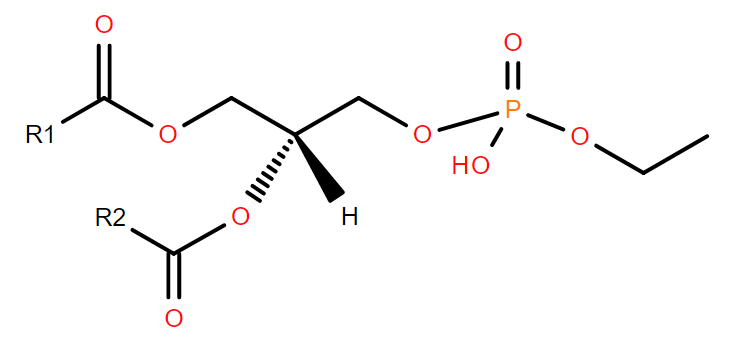
Over the festive season, it’s likely many of our readers might have enjoyed an alcoholic drink or two. In doing so, they will have formed phosphatidylethanol in their blood, especially in red blood cell membranes. It has been suggested that phosphatidylethanol is responsible for some of the effects of alcohol on the body1.
Phosphatidylethanol is formed from phosphatidylcholine by the enzyme phospholipase D2. This enzyme removes the choline headgroup, generally using water to attack the choline-phosphate bond to form phosphatidic acid. However, ethanol can substitute for water in the reaction, resulting in phosphatidylethanol instead. The most common form detected has palmitoyl and oleoyl acyl chains (PEth 16:0/18:1)3, consistent with the most prevalent acyl chains in phosphatidylcholine.
Phosphatidylethanol remains in the body much longer than ethanol itself, potentially for 2-3 weeks. It acts as a clinical and forensic marker for alcohol consumption, and its abuse4, for example in assessing alcohol consumption prior to liver transplants, or in drink-driving cases, or even post-mortem. It’s said to be a more reliable marker than asking people how much alcohol they drink as there are many reasons why people might underestimate.
Given how long phosphatidylethanol remains in your system, and depending how festive your festive season was, it may be clear of your body by the end of ‘Dry January’, if you take part in that.
References
-
A Molecular Target for an Alcohol Chain-Length Cutoff J Mol Biol 2019 https://doi.org/10.1016/j.jmb.2018.11.028
J Mol Biol
2019
DOI 10.1016/j.jmb.2018.11.028
-
Formation of phosphatidylethanol in rat brain by phospholipase D
Biochem Biophys Res Comm
1987
DOI 10.1016/0006-291X(87)91507-5
-
Identification of 48 homologues of phosphatidylethanol in blood by LC-ESI-MS/MS
Anal Bioanal Chem
2010
DOI 10.1007/s00216-010-3458-5
-
Alcohol Biomarkers in Clinical and Forensic Contexts
Dtsch Arztebl Int
2018
DOI 10.3238/arztebl.2018.0309
December 2022
Just before Christmas 1938, a strange fish was landed in the South African port of East London. It would turn out that the fish was well known to science, but only from 400 million year old fossils called coelacanths. Far from being extinct as previously thought, the coelacanth line was alive (though maybe not well). Today, there are two known living species of coelacanth, in the genus Latimera, named after Majorie Courtenay-Latimer, the woman who preserved and studied that first specimen.
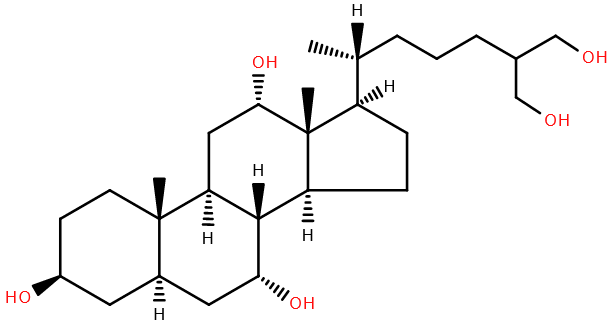
In 1964, the components of coelacanth bile were analysed1, identifying a novel compound, a 27-carbon pentahydroxy sterol, as the major component. This bile alcohol, principally found as a sulphate conjugate, was termed latimerol.
Latimerol is described as primitive, being chemically more similar to cholesterol (from which it is derived) than bile alcohols and acids of other organisms, for instance cholic acid in humans.
Bile, with its component acids and alcohols, assists in solubilising dietary fats, which would be plentiful in the carnivorous coelacanth diet. In addition, bile alcohols and salts are known to act as pheromones and semiochemicals in fish2. Whether laterimol has such a role is unknown. As the coelacanth habitat is relatively remote, and it is a critically endangered species3, further study is challenging. Indeed, we may never know the full role of latimerol should the coelacanth go extinct.
References
-
Comparative studies of ‘bile salts’. 20. Bile salts of the coelacanth, Latimeria chalumnae Smith
Biochem J
1964
DOI 10.1042/bj0930034
-
Bile Salts as Semiochemicals in Fish
Chemical Senses
2014
DOI 10.1093/chemse/bju039
-
The IUCN Red List of Threatened Species
Latimeria chalumnae. The IUCN Red List of Threatened Species
2000
DOI 10.2305/IUCN.UK.2000.RLTS.T11375A3274618.en
November 2022

Nematodes have the option of a ‘time out’ period in their life cycle, partially due to a lipid. If food is in short supply, the temperature is not optimal, or there are too many nematodes in a small area, a nematode larva can moult into a ‘Dauer’ form. In this form (German for duration or enduring) the nematode enters a kind of suspended animation until conditions improve.
The molecule which signals population density, named Daumone, was identified in 20051 as a heptanoic acid linked to an unusual sugar, ascarylose, via an omega-1 hydroxy group. Ascarylose is named after the Ascaridoidea nematode family.
Ascarosides with far longer acyl chains make up a protective layer around within nematode eggs shells2. In fact, nematodes produce a variety of hormones which share a common ‘core’ of ascarylose linked to varying length fatty acids and possibly ‘decorated’ with other chemical groups. These form a whole modular toolkit of signalling molecules in the nematode3 and have their own subclass within the LIPID MAPS structure database termed ascarosides.
If only we humans could hibernate while food and heat are in short supply!
References
-
Chemical structure and biological activity of the Caenorhabditis elegans dauer-inducing pheromone.
Nature
2005
DOI 10.1038/nature03201
-
Structure of the Ascarosides from Ascaris suum
J Nat Prod
1996
DOI 10.1021/np960236+
-
Comparative metabolomics reveals biogenesis of ascarosides, a modular library of small-molecule signals in C. elegans
J Am Chem Soc
2012
DOI 10.1021/ja210202y
October 2022
In the northern hemisphere, as the trees display their autumn colours, in a process of ‘de-greening’ there is an abundance of phytol, a 20-carbon prenol primary alcohol containing four methyl branches. Phytol is part of chlorophyll and is liberated as chlorophyll is degraded, cleaving the phytyl ‘tail’ from yields phytol and chlorophyllide1. Chlorophyll is also broken down in the guts of ruminants by the gut microbiota, and the phytol is converted to phytanic acid. This molecule is absorbed by the animal into its tissues. Because the human gut lacks organisms which degrade chlorophyll, dairy and meat products are the main source of phytanic acid in the human diet, and not any green vegetables. Inability to metabolise phytanic acid causes problems for humans2. The presence of a methyl on the third carbon prevents phytanic acid being degraded by beta-oxidation, as is the case for many fatty acids. It needs to be broken down by alpha-oxidation, and deficiencies in enzymes of this pathway lead to conditions such as Refsum disease, where phytanic acid builds up in the body. Treatment for Refsum disease is to remove pytanic acid from the diet, by avoiding meat and dairy products. Ironically, the solution to problems caused by a molecule coming from chlorophyll is to eat more greens!
References
-
Degradation pathway(s) of chlorophyll: what has gene cloning revealed?
Trends Plant Sci
2000
DOI 10.1016/s1360-1385(00)01735-0
-
Phytanic acid: production from phytol, its breakdown and role in human disease
Cell. Mol. Life Sci.
2006
DOI 10.1007/s00018-005-5463-y
September 2022

Glucosyl sphingosine was first detected in mammalian tissue nearly fifty years ago when it was observed in patients with Gaucher's disease1, This condition is characterized by a deficiency in glucocerebrosidase, the enzyme which removes the glucose from glucosyl ceramide to form ceramide. As a result, glucosyl ceramide accumulates in certain tissues.
Glucosyl sphingosine is formed from glucosyl ceramide by removal of the acyl group. This is an unusual reaction which is not observed in healthy subjects. In the skin of patients suffering from atopic dermatitis (AD) however, glucosyl sphingosine is seen in high amounts due to increased levels of an enzyme which specifically removes the acyl chain from glucosyl ceramide2.
Glucosyl sphingosine has been implicated as the source of irritation and itchiness in AD3 with recent papers suggesting a mechanism by which it activates various molecular receptors and ion channels in the skin which seem to interact with each other4,5.Understanding this mechanism more fully could help in treating the symptoms of AD.
References
-
Isolation and characterization of glucosylsphingosine from Gaucher's spleen
J Lipid Res
1974
-
Abnormal Expression of the Novel Epidermal Enzyme, Glucosylceramide Deacylase, and the Accumulation of its Enzymatic Reaction Product, Glucosylsphingosine, in the Skin of Patients with Atopic Dermatitis
Lab Invest
2003
DOI 10.1097/01.lab.0000059931.66821.92
-
Glucosylsphingosine induces itch-scratch responses in mice
Biomol. Ther. (Seoul)
2010
DOI 10.4062/biomolther.2010.18.3.316
-
Glucosylsphingosine evokes pruritus via activation of 5-HT2A receptor and TRPV4 in sensory neurons
Br. J. Pharmacol.
2022
DOI 10.1111/bph.15733
-
Activation of serotonin receptor 2 by glucosylsphingosine can be enhanced by TRPA1 but not TRPV1: Implication of a novel glucosylsphingosine-mediated itch pathway
Biochim Biophys Acta Biomembr
2022
DOI 10.1016/j.bbamem.2022.184014
August 2022
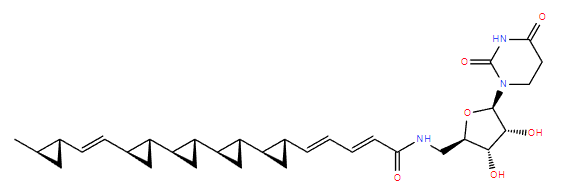
Just when you thought it was safe to go back in the… soil
The microbial world is one huge battle ground as various organisms compete with each other. In 1990 a novel antibiotic was discovered produced by the bacterium Streptoverticillium fervens which had anti-fungal activity1.
This molecule, called FR-900848 initially, is unusual due to the multiple cyclopropyl groups along the acyl chain which is linked to a nucleoside via an amide bond. The three-membered cyclopropyls, looking like shark teeth, led to the colloquial name Jawsamycin.
Jawsamycin stops the growth of fungi, but has no activity against bacteria. It acts by inhibiting the fungal enzyme SPT14 that produces another lipid, glycosylphosphatidylinositol. Specifically, it adds a sugar N-acetyl glucosamine to the phosphatidylinositol2. While jawsamycin stops the fungal enzyme,it has no effect on the equivalent human enzyme.
Though chemically, jawsamycin could be considered a fatty-amide, the biosynthetic pathway leading to the formation of jawsamycin utilities polyketide synthase3. Thus jawsamycin is considered a linear polyketide.
Recently the polyketide synthases involved in creating molecules such as jawsamycin have been repurposed to produce polycyclopropanated fatty acids4. These are energy dense molecules which the authors state have potential to replace fossil fuels. If such fuels catch on,lots of bacteria will need to be grown in culture to produce enough fuel molecules.
We’re gonna need a bigger.. fermenter!
References
-
A novael antifungal antibiotic FR-900848
J Antibiot
1990
DOI 10.7164/antibiotics.43.748
-
Jawsamycin exhibits in vivo antifungal properties by inhibiting Spt14/Gpi3-mediated biosynthesis of glycosylphosphatidylinositol
Nat Commun
2020
DOI 10.1038/s41467-020-17221-5
-
Biosynthesis of the structurally unique polycyclopropanated polyketide-nucleoside hybrid jawsamycin (FR-900848).
Angew Chem Int Ed Engl.
2014
DOI 10.1002/anie.201402623
-
Biosynthesis of polycyclopropanated high energy biofuels
Joule
2022
DOI 10.1016/j.joule.2022.05.011
July 2022
Vitamin K is essential in forming the enzymes of the blood clotting cascade. It was discovered by accident by Henrik Dam, who was studying cholesterol metabolism in chicks. He was feeding the chicks a cholesterol-free diet by extracting cholesterol from their food with chloroform. The chicks however started haemorrhaging. It was known that they could synthesise cholesterol, and adding vitamins A or D to the diet made no difference, so an unknown substance must be at play. The chloroform had not only extracted cholesterol, but also this unknown substance, initially named Koagulationsvitamin, or vitamin K1.
Dam was awarded the Nobel Prize in 1943 for its discovery, along with Edward Doisy who elucidated its structure2. This was vitamin K1, produced in green plants and also called phylloquinone. Bacteria produce a related family of molecules termed vitamin K2 or menaquinone, in which the prenyl chain is unsaturated and can vary in length.
Vitamin K is essential for humans because enzymes in the blood clotting cascade contain modified glutamic acid amino acids. Vitamin K acts as a co-factor in forming this modification3 and so without vitamin K, these enzymes can’t function and the blood doesn’t clot.
In plants of course, vitamin K1 has nothing to do with blood clotting, or even amino acid modification. It acts instead to move electrons in the photosynthetic machinery4. At some point in evolution, animals have repurposed the molecule to do a completely different job.
References
-
The Antihæmorrhagic Vitamin of the Chick.: Occurrence And Chemical Nature
Nature
1935
DOI 10.1038/135652b0
-
On the Constitution of Vitamin K1
J Am Chem Soc
1939
DOI 10.1021/ja01876a510
-
Structural and functional insights into enzymes of the vitamin K cycle
J Thromb Haemost
2016
DOI 10.1111/jth.13217
-
Phylloquinone in photosystem I: are quinones the secondary electron acceptors in all types of photosynthetic reaction centers?
Trends Biochem Sci
1988
DOI 10.1016/0968-0004(88)90206-X
June 2022
7-dehydrocholesterol is the final precursor to cholesterol in the Kandutsch-Russell biosynthesis pathway1. It’s also the molecule on which sunlight acts to form vitamin D, and in insects, cholesterol is converted back to 7-dehydrocholesterol on the pathway to synthesise the developmental ecdysone hormones2.
Too much 7-dehydrocholesterol is, however, not a good thing. People with mutations in the enzyme which converts it to cholesterol, 7-Dehydrocholesterol reductase, show elevated levels of 7-Dehydrocholesterol. They suffer from a condition called Smith–Lemli–Opitz syndrome. This can manifest in many different ways, including physical, cognitive and behavioural symptoms of varying severity.
Some of the behavioural issues which Smith–Lemli–Opitz syndrome can cause include self-harm3, curiously, a process also seen in brooding female octopuses, and possibly for the same reason. Mother octopuses brooding their eggs change their behaviour dramatically. They fast, decline physiologically and ultimately die, often repeatedly and deliberately injuring limbs during this time. A recent paper4 has shown that the optic gland of the California two-spot octopus is key to this behaviour, and removal of the gland, which produces 7-dehydrocholesterol during brooding, stops the self-harm and death of the creature.
References
-
Preputial gland tumor sterols. 3. A metabolic pathway from lanosterol to cholesterol
J Biol Chem
1960
DOI 10.1016/S0021-9258(18)64608-3
-
Neverland is an evolutionarily conserved Rieske-domain protein that is essential for ecdysone synthesis and insect growth
Development
2006
DOI 10.1242/dev.02428
-
Behavioral phenotype of RSH/Smith-Lemli-Opitz syndrome
Ment Retard Dev Disabil Res Rev
2000
DOI 10.1002/1098-2779(2000)6:2<131::AID-MRDD7>3.0.CO;2-R
-
Steroid hormones of the octopus self-destruct system
Curr Biol
2022
DOI 10.1016/j.cub.2022.04.043
May 2022
Your skin has a tough job protecting you from the world. One of the many abuses it suffers is exposure to ultraviolet wavelengths of sunlight. This generates, via singlet oxygen, hydroperoxysqualene from squalene, one of the most abundant lipids on the skin surface.
As any of the double bonds in squalene can be attacked by singlet oxygen, there are six potential forms of singly hydroperoxidated squalene produced1 which have been shown to accumulate on the skin. Shown here is 2-hydroperoxy-squalene. The presence of hydroperoxysqualene triggers inflammatory reactions, maybe contributing to the body’s response to sunburn2 and even to acne3.
Hydroperoxysqualene induces expression of genes linked to inflammation but its effect has been shown to be mitigated by gamma-tocotrienol, a form of vitamin E4.
So, if you’re going to be enjoying a lot of sunshine, it might be wise, along with other precautions, to top up your vitamin E.
References
-
Mass Spectrometric Discrimination of Squalene Monohydroperoxide Isomers
J Oleo Science
2017
DOI 10.5650/jos.ess16159
-
The possible role of squalene and its peroxide of the sebum in the occurrence of sunburn and protection from the damage caused by U.V. irradiation
J Toxicol Sci
1984
DOI 10.2131/jts.9.151
-
A possible role for squalene in the pathogenesis of acne. I. In vitro study of squalene oxidation
Br J Dermatol
1986
DOI 10.1111/j.1365-2133.1986.tb04060.x
-
Gamma-Tocotrienol Reduces Squalene Hydroperoxide-Induced Inflammatory Responses in HaCaT Keratinocytes
Lipids
2010
DOI 10.1007/s11745-010-3458-4
April 2022
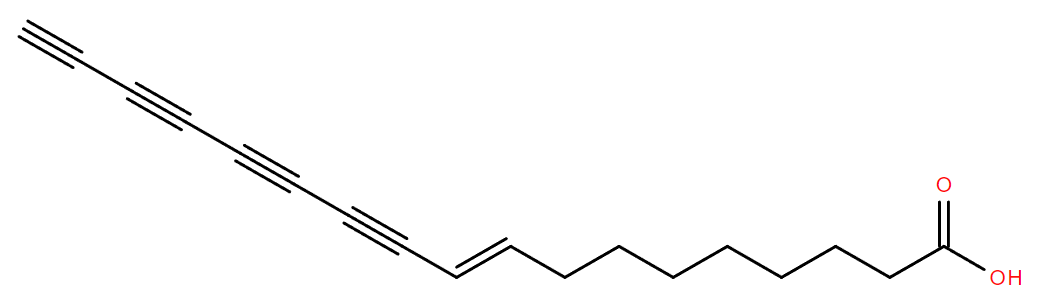
Some lipids appear so bizarre that one checks whether the paper describing them was published on April 1st! That was certainly the case with Protegenin A and its sibling protegenins1. With up to four conjugated carbon-carbon triple bonds in an 18-carbon chain, at first glance one might think these fatty acids must be synthetic, They are however, completely natural and not April Fools!
Protegenins are fatty acids produced by a strain of Pseudomonas protegens as weapons in the continual competition between microbes. The presence of this bacterium in the soil is known to protect plants from fungal infection2. Similar families of polyyne lipids have been discovered in other bacteria, for example the Caryoynencins from Pseudomonas (now Trinickia) caryophylli3.
The likely production of protegenins in this bacterium was inferred from the presence of a cluster of genes predicted to have functions in the biosynthesis of polyyne fatty acids. Purification and analysis of the compounds secreted into the growth medium confirmed the existence of the protegenin family of compounds and their chemical structures..
Knocking out a gene essential for protegenin production resulted in significantly more cucumber seedlings rotting when grown in the presence of the knock-out than when grown with the ‘wild type’. While the molecular mechanism of how protegenins act seems as yet unknown, this experiment indicated that they have anti-fungal activity.
As spring is here in the northern hemisphere, and seeds are being sown, there are all kinds of seemingly odd compounds silently aiding their growth.
References
-
Protegenins A-D, Bacterial Polyynes Involved in the Antioomycete and Biocontrol Activities of Pseudomonas protegens.
ACS Chem Biol
2021
DOI 10.1021/acschembio.1c00276
-
Pseudomonas protegens sp. nov., widespread plant-protecting bacteria producing the biocontrol compounds 2,4-diacetylphloroglucinol and pyoluteorin
Nat Rev Microbiol
2005
DOI 10.1016/j.syapm.2010.10.005
-
Caryoynencins, potent antibiotics from a plant pathogen Pseudomonas caryophylli
Tetrahedron Letts
1987
DOI 10.1016/S0040-4039(00)96437-2
March 2022
Those with a classical education might conclude sapienic acid is somehow connected to wisdom. In fact that’s only partly true. It is named after Homo sapiens, ‘wise man’ (allegedly), in whom it is believed to be unique among animals1. Sapienic acid forms a significant part of sebum- the oily substance secreted by sebaceous glands.
These glands express a delta-six desaturase enzyme which converts palmitic acid to sapienic acid. While delta-six desaturases are common, they usually add a double bond at this position in fatty acids which are already desaturated at the delta-9 position. The skin desaturase is identical to the form found elsewhere, suggesting unique conditions within the sebaceous gland favouring desaturation of palmitic acid, rather than palmitoleic acid.
Sapienic acid acts as an anti-microbial agent, forming part of the skin’s defenses against infection2. Why sapienic acid is absent from other animals appears to remain a mystery.
While sapienic acid is unique to humans in the animal kingdom, it is not unique in life. It is the major fatty acid of certain plant seeds, such as Thunbergia alata3. In this species, is it formed by the same route as in humans, by desaturation of palmitic acid. Had sapienic acid been discovered here first, who knows what it might have been called; maybe alatic acid or thunbergic acid?
References
-
Identification of the Δ-6 Desaturase of Human Sebaceous Glands: Expression and Enzyme Activity
J Invest Dermatol
2003
DOI 10.1046/j.1523-1747.2003.12123.x
-
Thematic Review Series: Skin Lipids. Antimicrobial lipids at the skin surface
J Lipid Res
2008
DOI 10.1194/jlr.R700016-JLR200
-
Occurrence of Cis-6-hexadecenoic acid as the major component ofThunbergia alata seed oil
Lipids
1971
DOI 10.1007/BF02531295
February 2022
Eat a deadly carrot, and you’ll know about it. Plants of the Thapsia genus, collectively termed ‘deadly carrots’, cause serious digestive problems and even death in any mammal unfortunate enough to ingest them. Simply touching some can cause skin irritation. The toxicity, and medicinal properties, of deadly carrots has been known since antiquity in the Mediterranean regions where they grow1. The Romans used its emetic and purgative properties for slimming. Doubtless an unpleasant, and possibly lethal strategy.
Like many umbelliferous species, Thapsia plants contain compounds to deter animals from eating them. The main toxin isolated from Thapsia is a sesquiterpene called thapsigargin. It has a guaianolide skeleton, which is likely derived from farnesyl pyrophosphate, though the biosynthesis has yet to be elucidated2. This core is adorned with several fatty acyl groups.
Thapsigargin is a specific inhibitor of the sarcoplasmic/endoplasmic reticulum calcium ATPase (SERCA) pump, essential for regulating calcium homeostasis within cells. Because it traps the pump in one conformation, thapsigargin has been essential in solving crystal structures of this protein.
As SERCA is a target for chemotherapeutics, thapsigargin and its analogues are being investigated as potential anti-cancer drugs3 and it has even been suggested as a lead compound for anti-virals against coronavirus4. In both cases, its low abundance naturally, and difficulty of chemical synthesis remain a challenge.
Thapsic acid, a 16-carbon dicarboxylic fatty acid, is also said to be named after Thapsia, from which it was first isolated, but the original work describing that seems to be lost, at least to this author.
References
-
Sesquiterpenoids from Thapsia Species and Medicinal Chemistry of the Thapsigargins
Prog Chem Org Nat Prod
DOI 10.1007/978-3-7091-6529-4_2
-
Localization and in-Vivo Characterization of Thapsia garganica CYP76AE2 Indicates a Role in Thapsigargin Biosynthesis
Plant Physiol
2017
DOI 10.1104/pp.16.00055
-
Thapsigargin—From Traditional Medicine to Anticancer Drug
Int J Mol Sci
2021
DOI 10.3390/ijms22010004
-
Multi-level inhibition of coronavirus replication by chemical ER stress
Nat Commun
2021
DOI 10.1038/s41467-021-25551-1
January 2022
References
-
Pigments of Staphylococcus aureus, a series of triterpenoid carotenoids
J Bact
1981
DOI 10.1128/jb.147.3.900-913.1981
-
Structure and Biosynthesis of Staphyloxanthin from Staphylococcus aureus
J Biol Chem
2005
DOI 10.1074/jbc.M505070200
-
Staphyloxanthin plays a role in the fitness of Staphylococcus aureus and its ability to cope with oxidative stres
2020
Infect Immun.
DOI 10.1128/IAI.00204-06
-
MSI-1 combats drug-resistant S. aureus by affecting bacterial viability and inhibiting carotenoid pigment production
Micro Res
2021
DOI 10.1016/j.micres.2021.126909
December 2021
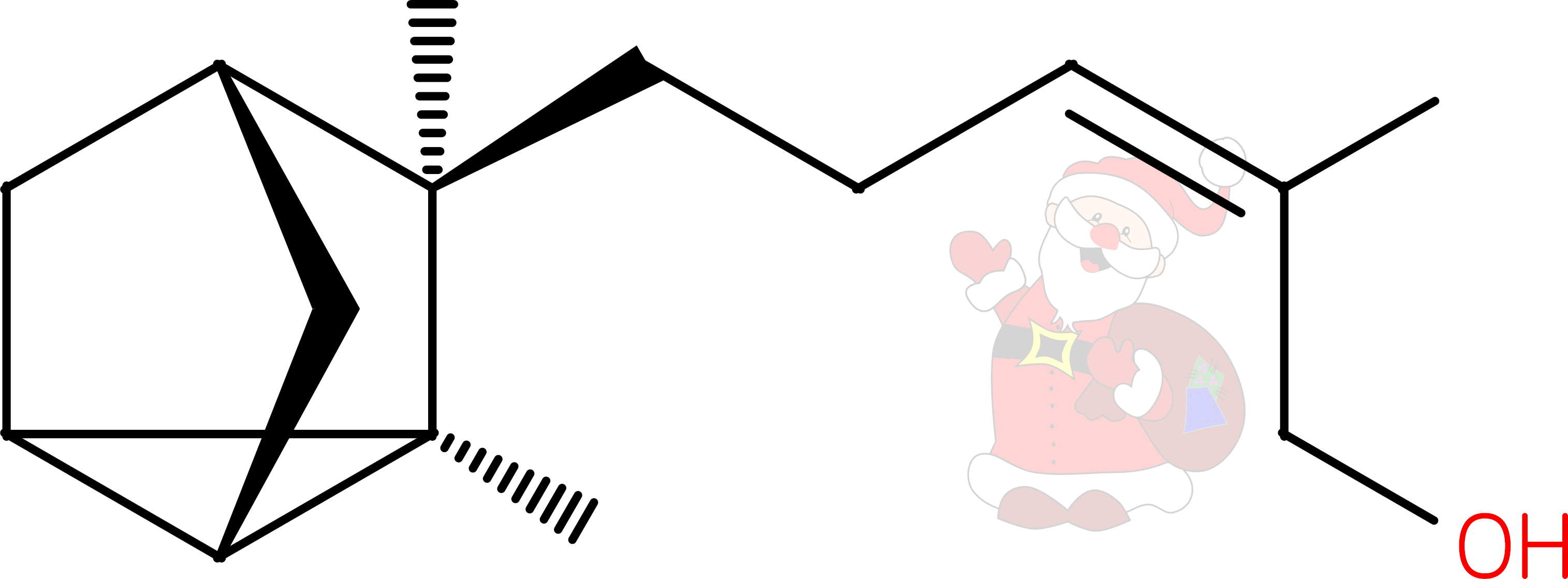
At this time of year, one might get a message from a friend or family member asking
“What do you want from Santa? LOL”
But santalol isn’t Father Christmas laughing out loud, but rather a terpene from the heartwood of the sandalwood tree; Santalum album. If you’re giving, or receiving, perfume this Christmas, santalol might well be in the gift, as it’s valued in the perfume industry.
In fact there are two related santalols, alpha and beta. Alpha-santalol makes up over half of the oil distilled from the wood, beta-santalol being the other major component. Sandalwood oil has been used medicinally and religiously for thousands of years, with various modern medicinal properties ascribed to alpha-santalol1.
The structure of the tricyclic alpha-santalol was established in 19632, and a chemical synthesis achieved shortly after3
So much sandalwood oil is now used that Santalum album is considered vulnerable to extinction. Recently, santalol has been produced biosynthetically in commercial quantities by cloning santalol-producing genes into Rhodobacter sphaeroides, and even bakers’ yeast5
References
-
Medicinal properties of alpha-santalol, a naturally occurring constituent of sandalwood oil: review
Nat Prod Res
2019
DOI 10.1080/14786419.2017.1399387
-
The stereochemistry of α- and β-santalol
Tetrahedron Letts
1963
DOI 10.1016/S0040-4039(01)90982-7
-
Total synthesis of α-santalol
Tetrahedron Letts
1967
DOI 10.1016/S0040-4039(00)90958-4
-
Biosynthesis of Sandalwood Oil: Santalum album CYP76F cytochromes P450 produce santalols and bergamotol
PLoS One
2013
DOI 10.1371/journal.pone.0075053
-
Optimized biosynthesis of santalenes and santalols in Saccharomyces cerevisiae
Appl Microbiol Biotechnol
2021
DOI 10.1007/s00253-021-11661-9
November 2021
Undoubtedly the hottest lipid of the moment is capsaicin- the molecule which imparts the hot sensation we experience when eating chilli peppers and which helped win David Julius this year’s Nobel Prize in Physiology or Medicine. The concentration, and efficacy of these compounds relate to the ‘hotness’ which is measured on the Scoville scale1. Capsaicin acts by stimulating heat receptors and Julius’ team used if to identify which particular protein receptor in sensory neurons it triggered2. The receptor turned out to be TrpV1, a transmembrane protein located in nerve endings. It is also responsible for detecting high temperature. Put your hand in hot water, or a chilli in your mouth and TrpV1 allows cations to flood into the nerve cell, initiating a signal your brain interprets as heat.
Capsaicin, a vanillylamide of 8-methyl-6-nonenoic acid, is very lipophilic, explaining why milk or yoghurt, containing fats, can ease the spiciness of a curry, but water will not. Its presence in peppers deters mammals from eating the fruit and destroying the seeds by chewing them. This strategy might appear to have failed for humans, but serendipitously, our love of chilli means we now cultivate the plants, actively ensuring its genes live on. The avian version of TrpV1 does not react to capsaicin, so birds can eat peppers without the hot taste. They do not digest the seeds but excrete them, with a lump of fertilizer, to grow a new plant.
While capsaicin causes a sometimes painful burning sensation, and may be responsible for some of the more well-known effects of spicy food3, it can actually be used in the treatment of pain as prolonged exposure desensitizes the receptor such that it can no longer transmit a signal4.
References
-
Note on Capsicums
J. Am. Pharm. Assoc.
1912
DOI 10.1002/jps.3080010520
-
The capsaicin receptor: a heat-activated ion channel in the pain pathway
Nature
1997
DOI 10.1038/39807
-
Capsaicin—the spicy ingredient of chili peppers: A review of the gastrointestinal effects and mechanisms
Trends in Food Science & Technology
2021
DOI 10.1016/j.tifs.2021.08.034
-
Capsaicin: Current Understanding of Its Mechanisms and Therapy of Pain and Other Pre-Clinical and Clinical Uses
Molecules
2016
DOI 10.3390/molecules21070844
October 2021
October’s Lipid of the Month is N-Retinylidene-N-retinylethanolamine or A2E. It forms rarely, but accumulates in the eye and is associated with age-related eye diseases such as macular degeneration. It’s chemical identity was determined in 19961 .
Light falling on receptors in the retina isomerises a double bond in retinal, converting it from cis to trans. This triggers the process of signalling to the brain that you have seen something. There follows a series of reactions to recycle the retinal back to the cis conformation to detect another photon.
Sometimes, the retinal escapes this cycle and reacts with the ethanolamine headgroup of PE lipids. More rarely, but associated with exposure to bright light, this species can react with a second retinal to form A2E. While A2E formation is rare, there is no enzymatic mechanism to degrade it. Over time, this orange-coloured and fluorescent molecule accumulates in retinal pigment epithelial cells.
Clearly, coloured molecules in the eye are going to affect vision, but A2E can also disrupt membrane integrity as it behaves like a detergent. In addition, any (or all) of the double bonds in the retinal can be modified by reactive oxygen species to form epoxides which have been shown to cause DNA damage2.
References
-
Ocular Age Pigment “A2-E”: An Unprecedented Pyridinium Bisretinoid
J. Am. Chem. Soc.
1996
DOI 10.1021/ja953480g
-
A2E, a byproduct of the visual cycle
Vision Res.
2003
DOI 10.1016/S0042-6989(03)00475-9
September 2021
The most abundant fatty acid in the hair of most mammals, including humans, is 18-methyl-eicosanoic acid (18-MEA). It makes up as much as half of all the fatty acids in hair and is found exclusively on the surface of the cuticle1. It is covalently linked to the exterior surface of the hair protein via thioester bonds where it functions to maintain a waterproof barrier to the hair surface. It has also been ascribed roles in keeping hair untangled2.
Why this particular fatty acid, unusual for its anteiso methyl branch, is found on the hair surface is unclear, but studies have shown that the methyl group affects the way fatty acids are packed on a surface3
The thioester bond linking 18-MEA to the hair is broken by alkaline conditions, often found in hair colourants and perms, which means the surface lipid layer is damaged by these treatments. There are hair products on the market which contain 18-MEA, but as the linkage is created enzymatically as the hair grows from its follicle, it is hard to see how these would replace 18-MEA to the natural state.
References
-
Integral lipids of mammalian hair
Comparative Biochemistry and Physiology Part B: Comparative Biochemistry
1989
DOI 10.1016/0305-0491(89)90264-2
-
18-MEA and hair appearance
Journal of Cosmetic Science
2010
DOI 10.1111/j.1468-2494.2010.00609_6.x
-
Self-assembly of long chain fatty acids: effect of a methyl branch
Phys. Chem. Chem. Phys.
2014
DOI 10.1039/C4CP00512K
-
Hairs from Patients with Maple Syrup Urine Disease Show a Structural Defect in the Fiber Cuticle
Journal of Investigative Dermatology
1996
DOI 10.1111/1523-1747.ep12343618
August 2021
The old joke says that the one thing worse than finding a caterpillar in your apple, is finding half a caterpillar. Of course, finding no caterpillar is even better! One of the chemicals fruit farmers use to keep apples, and other fruit, caterpillar-free is codlemone, 8E,10E-dodecadien-1-ol.
The codling moth (Cydia pomonella) is a major pest of fruit trees, its larvae feed on apples, pears, and also walnuts, causing serious economic damage. Female codling moths emit codlemone as a pheromone to attract a mate, so a trap baited with codlemone lures males in as the Sirens of myth lured sailors to their deaths with song. Alternatively, releasing codlemone throughout an orchard means males cannot locate a female. In either case, mating is disrupted and no larvae are produced to eat the fruit.
Codlemone acts synergistically with other volatile chemicals, some released by the moths and others, such as pear ester (ethyl 2E,4Z-decadienoate), by fruit trees themselves. This enables males to better locate females sitting on a good source of larval food. Mixtures of such pheromones in commercial products lead to more efficient mating disruption and fewer ruined apples1.
A recent paper2 suggests that moths began to produce codlemone via the duplication of fatty acid desaturase genes, allowing different functionalities to evolve which introduce specific double bonds to a lipid. First, a desaturase forms a double bond at carbon nine of lauryl-CoA. There follows a further desaturation resulting in double bonds at carbons eight and ten, before a reductase releases codlemone from the CoA.
Disrupting the mating behaviour of the codling moth with pheromones means less non-specific insecticide needs to be used, but still fewer caterpillars, whole or fractional, in our fruit.
References
-
Codling Moth Management and Chemical Ecology
Ann. Rev. Entomol.
2008
DOI 10.1146/annurev.ento.53.103106.093323
-
Evolution of the codling moth pheromone via an ancient gene duplication
BMC Biol.
2021
DOI 10.1186/s12915-021-01001-8
July 2021
On and in the human body, microbes out-number our own cells by about ten to one. Increasingly, we are coming to understand that these commensal bacteria are actively interacting with us, influencing our immune systems, and also our behaviour1. One way in which they do this is via signalling molecules which mimic those of our own bodies. Bacteroides vulgatus is a bacterium which is found in the human intestinal tract and produces a lipid called commendamide2, N-acyl-3-hydroxypalmitoyl-glycine. Commendamide is an abbreviation of ‘commensal mimicking endogenous amide’ and, as the name suggests, it likely mimics amide lipids, such as the endocannabinoids which interact with G-protein coupled receptors (GPCR) to modulate various aspects of physiology as diverse as mood, memory, hormone and and immune system regulation. Endocannabinoids bind to the same GPCRs which are stimulated by compounds in cannabis, the most abundant being tetrahydrocannabinol.
It's unlikely however that commendamide induces the same feeling of being chilled and hungry as THC, as it interacts with a different GPCR to that which the endocannabinoids bind. Commendamide interacts with GPCR132/G2A, which is involved in signalling in the immune system. So B. vulgatus might be using it’s lipid signaling molecule to modulate our immune response to it being around.
There may be multiple roles for commendamide, it has been shown to solubilise cholesterol, lyse red blood cells, and possibly play a role in quorum sensing3. Undoubtedly the relationship between humans and our bacteria is a complex one, with much still to be discovered.
References
-
Gut Microbiota as a Mediator of Host Neuro-Immune Interactions: Implications in Neuroinflammatory Disorders
Mod Trends Psychiatry
2021
DOI 10.1159/000510416
-
Functional metagenomic discovery of bacterial effectors in the human microbiome and isolation of commendamide, a GPCR G2A/132 agonist
Proc Natl Acad Sci U S A.
2015
DOI 10.1073/pnas.1508737112
-
The Bacteroidales produce an N-acylated derivative of glycine with both cholesterol-solubilising and hemolytic activity
Sci Rep
2017
DOI 10.1038/s41598-017-13774-6
June 2021
On a warm summer evening at Old Warden airfield, England, the faint smell of June’s Lipid of the Month lingers in the still air. It comes from the engines of several antique, but still air-worthy, aeroplanes.
In the early years of the 20th century, castor oil, pressed from seeds of the castor oil plant Ricinus communis, was found to be a good lubricant for rotary aircraft engines1. Ricinolein is the main component, forming up to 90% of the oil. Ricinolein is a triglyceride of ricinoleic acid, a fatty acid chemically similar to oleic acid but with the addition of a hydroxyl group at the 12 position. It’s likely these hydroxyls, together with the homogeneity of castor oil, confer the properties that make it a much better lubricant than other vegetable oils.
Over time, hot ricinolein will polymerise and could ‘gum up’ an engine, but as rotary engines either burn or eject all of their oil, it is never in the engine long enough for gumming up to be an issue. So successful were the castor-containing lubricants developed and sold by the Wakefield Oil Company, it changed its name to an abbreviation of castor oil, and Castrol sells motor oils to this day.
Ricinolein has many more uses; it’s found in paints, cosmetics, inks and more widely in the chemical industry. It’s even suggested to be a renewable resource for other chemicals which can be derived from it2.
Castor oil is also probably most notorious for its laxative properties. Ricinoleic acid mimics prostaglandin E2 to stimulate smooth muscle contraction in the gut3. It is widely said that the continuous ejection of castor oil from aircraft engines had a laxative effect on the First World War pilots sitting behind those engines, but solid evidence for this is surprisingly difficult to find.
References
-
A History of Aircraft Piston Engine Lubricants
West Coast International Meeting and Exposition
1981
DOI 10.4271/810849
-
Castor oil as a renewable resource for the chemical industry
Eur. J. Lipid Sci. Technol. 2010
2010
DOI 10.1002/ejlt.200900138
-
Castor oil induces laxation and uterus contraction via ricinoleic acid activating prostaglandin EP3 receptors
Proc Natl Acad Sci U S A.
2012
DOI 10.1073/pnas.1201627109
May 2021
May’s Lipid of the Month is generated by one of the immune system’s many mechanisms used to protect the body from pathogens. 2-chlorohexadecanal is formed when the vinyl ether bond of plasmalogen lipids containing a 1Z-hexadecenyl chain (for example 1-(1Z-hexadecenyl)-2-(9Z-octadecenoyl)-sn-glycero-3-phosphocholine) is attacked by endogenous hypochlorous acid (HOCl)1. The resulting 2-chloroaldehyde can subsequently oxidise to form a 2-chloro fatty acid.
HOCl is produced by neutrophils from hydrogen peroxide and chloride ions with the aid of the enzyme myeloperoxidase, located in the lysosome. This relatively strong oxidising agent will react with many compounds, essentially destroying anything within the lysosome. HOCl also dissociates to hypochlorite, the active constituent of bleach, but it should not be necessary to stress that attempting to augment the immune system by adding bleach is a very bad idea!!
2-chlorohexadecanal is not simply a lipid breakdown product but acts as a signalling molecule of infection itself. Its presence indicates a site of infection or inflammation and attracts other neutrophils to the site2. It can also induce expression of cyclooxygenase-23, the enzyme which forms prostaglandin H2, another part of the inflammation pathway.
References
-
Reactive chlorinating species produced by myeloperoxidase target the vinyl ether bond of plasmalogens: identification of 2-chlorohexadecanal
J. Biol. Chem
2001
DOI 10.1074/jbc.M101447200
-
Reactive Chlorinating Species Produced during Neutrophil Activation Target Tissue Plasmalogens
J. Biol. Chem
2002
DOI 10.1074/jbc.M109489200
-
2-Chlorohexadecanal and 2-chlorohexadecanoic acid induce COX-2 expression in human coronary artery endothelial cells
Lipids
2008
DOI 10.1007/s11745-008-3189-y
April 2021
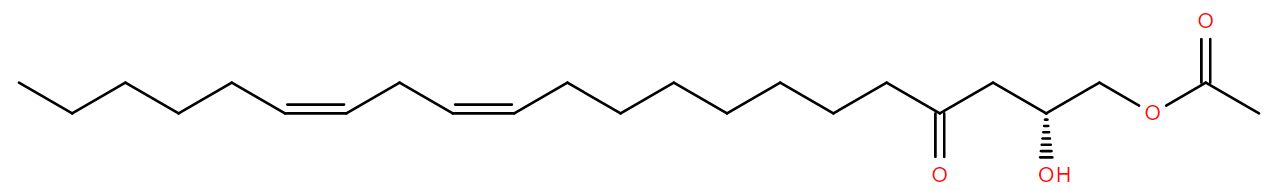
Avocados are undoubtedly a dangerous fruit, with around 50,000 avocado-related knife injuries reported over a 10-year period in the US alone1. But that’s not the only way the avocado plant can harm you. It has been known for a long time that the leaves are toxic to animals, especially to lactating livestock. Necrosis of the mammary gland tissues has been observed in goats and mice and cardiac problems follow eating avocado leaves in a variety of species.
The compound responsible for these effects was identified in 1995 as 1-acetoxy-2R-hydroxy-12Z,15Z-heneicosadien-4-one2, more conveniently termed ‘Persin’ (from the Latin name of the Avocado Persea americana). Persin is a 21-carbon, acetylated fatty alcohol, with two double bonds, a keto and a hydroxyl group along the acyl chain. When chemically synthesised, only the R-hydroxyl form was shown to replicate the toxic effects of avocado leaves.
Many toxins can form the basis of drugs and the effects of persin on mammary gland tissue has attracted attention as a potential therapeutic against breast cancer3, though the absence of literature on this subject in very recent years may suggest such research is no longer active.
Evidently, persin is absent in any significant amount from avocado fruit, otherwise they would be inedible, but next time you cut an avocado, don’t hold it in your hand!
References
-
Avocado-related knife injuries: Describing an epidemic of hand injury
Am J Emerg Med
2020
DOI 10.1016/j.ajem.2019.06.051
-
Isolation and identification of a compound from avocado (Persea americana) leaves which causes necrosis of the acinar epithelium of the lactating mammary gland and the myocardium
Nat Toxins
1995
DOI 10.1002/nt.2620030504
-
A novel plant toxin, persin, with in vivo activity in the mammary gland, induces Bim-dependent apoptosis in human breast cancer cells
Mol Cancer Ther
2006
DOI 10.1158/1535-7163.MCT-06-0170
March 2021
Last month, across the world, people watched the stunning video of NASA’s Perseverance rover as it was lowered to the surface of Mars via the hydrazine-powered rockets of its ‘Skycrane’. It’s main mission: to search for signs of ancient life on the Red Planet. Here on Earth, some of our many forms of life actually produce the rocket fuel hydrazine.
Anammox bacteria, found in the ocean, anaerobic bogs and lakes, and in waste-water treatment plants, oxidise ammonia and nitrate to nitrogen and water, via a hydrazine intermediate. They do so in a lipid-bound compartment called the anammoxosome, the membrane of which contains unique lipids found nowhere else in nature. These molecules, including March’s Lipid of the Month 8-[5]-ladderane-octanoic acid, were first identified in 20021 and contain linearly fused cyclobutane groups called ‘ladderanes’ in their fatty acyl chains.
Ladderane lipids form a very tightly-packed membrane which is much less permeable than ‘normal’ membranes, and probably serves to prevent the highly reactive and toxic hydrazine escaping the anammoxosome, as well as efficiently maintaining a proton gradient for the production of ATP.
Hydrazine powers cranes on Mars, but it is contained by ladders on Earth, showing that biology is incredibly resourceful and the diversity of lipids, seemingly endless. Whether Perseverance will find signs of ancient Martian life remains to be seen, but if it does, what sort of lipids might the Martians have contained?
References
-
Linearly concatenated cyclobutane lipids form a dense bacterial membrane
Nature
2002
DOI 10.1038/nature01128
February 2021
Quite a lot of February’s Lipid of the Month is being injected into people’s arms at the moment. It’s found in both the Pfizer-BioNTech and Moderna vaccines for COVID-19. 1,2-Distearoyl-sn-glycero-3-phosphocholine (DSPC) is a component of the liposome in these vaccines, the vehicle which protects the mRNA encoding the viral spike protein and delivers it into cells for translation.
DSPC is a commonly occurring phospholipid in eukaryotic cell membranes, it has two saturated fatty acyl chains of 18 carbons each and a charged phosphocholine head group. It has been used to study model membranes and liposomes for over forty years, so its physical properties in membranes are very well characterised. Commercially, DSPC is readily available and relatively inexpensive.
In the liposome, DSPC is a structural component, forming a stable lipid bilayer1 which prevents leakage of the nucleic acid cargo. But the liposome is a complex object, the lipids don’t simply form a ‘bubble’ enclosing the mRNA. The structure is much more lipid-filled; lipids within the liposome interact with each other and with the mRNA cargo in addition to forming the outer ‘shell’.2
While these COVID vaccines are novel in that they are the first examples of mRNA vaccines delivered via liposomes, it is not the first case of drug delivery via liposomes in the clinic, and not even the first example of RNA delivery in this way.3 So while these vaccines are a triumph of science, they build on work, including lipid research, that has taken place over decades.
References
-
Nanomaterial Delivery Systems for mRNA Vaccines
Vaccines
2021
DOI 10.3390/vaccines9010065
-
Structure of Lipid Nanoparticles Containing siRNA or mRNA by Dynamic Nuclear Polarization-Enhanced NMR Spectroscopy
J. Phys. Chem. B
2018
DOI 10.1021/acs.jpcb.7b10795
-
The Onpattro story and the clinical translation of nanomedicines containing nucleic acid-based drugs.
Nat. Nanotechnol.
2019
DOI 10.1038/s41565-019-0591-y
January 2021

In more northerly locations, January’s lipid of the month is in relatively short supply. 1α,25-dihydroxyvitamin D3, or calcitriol, is the active form of vitamin D, which binds to the vitamin D receptor to regulate transcription of potentially thousands of genes. While classically known to be involved in controlling calcium and phosphate levels in the body, it also plays a role in many other processes, including inflammation, infection, immunity, and some cancers1.
Vitamin D is a secosteroid (the prefix seco- from the latin ‘to cut’ indicating one of the sterol rings is broken) formed from 7-dehydrocholesterol. It’s in short supply in winter because the body needs sunlight to make that cut. Ultraviolet light falling on the skin cleaves a bond (between carbons 9 and 10 in the B ring) to form pre-vitamin D3, which isomerises spontaneously to vitamin D3, also known as cholecalciferol.
Cholecalciferol is the form which is found in vitamin supplements, and in foods such as fatty fish and eggs but it is not yet active. It must first undergo two hydroxylation reactions, one at each end of the molecule. The first reaction takes place in the liver to make 25-hydroxyvitamin D3, or calcidiol. Later, in the kidneys and other tissues, calcidiol has another hydroxyl group added, at the opposite end of the molecule to produce calcitriol.
There has been a great deal of research published over the past year on the link between vitamin D and COVID-19. At the time of writing there were over 300 such articles indexed on PubMed. Whether any effect is specific to this virus, or indicative of general immune health, we may never know. Given the many genes influenced by vitamin D, it’s definitely a complicated question to answer.
References
-
Primary Vitamin D Target Genes of Human Monocytes
Front. Physiol.
2019
DOI 10.3389/fphys.2019.00194
December 2020
In A Christmas Carol, Charles Dickens describes Marley’s ghost as having “a dismal light about it, like a bad lobster in a dark cellar.” Written before electric lighting and refrigeration, luminescent lobsters might have been a more commonly observed phenomenon than they are today.
The dismal light of Dickens’ decaying decapod would have been triggered by an acyl homoserine lactone (AHL), December’s lipid of the month.
Several species of bacteria, including Vibrio harveyi, an opportunistic pathogen of shellfish, produce light. Given it’s ecological niche, there would likely be lots of V. harveyi on the surface of a rotting lobster, and it’s only when there are lots in one place that they make light. AHL is used to sense how many bacteria are around using a system known as quorum sensing. Different species make different AHLs, each with specific acyl groups. In the case of V. harveyi, 3-hydroxybutyryl homoserine lactone is it’s calling-card1.
Each bacterium secretes AHL and is able to sense its concentration in the environment. If the bacterium is swimming about in the sea, the AHL quickly gets diluted and as the concentration is very low, no light is produced. As a single bacterium could not make enough light to be detected, doing so would be futile. However, if there are lots of bacteria in one place, the concentration of AHL is increased because they’re all secreting it, and it acts to switch on transcription of the genes needed to generate light. Many bacteria in one place make enough light for the blue-green glow to be visible.
While it may be hard to see the evolutionary advantage in making rotting lobsters glow, species related to V. harveyi form symbiotic relationships with squid and fish, and live in their light-emitting organs where producing light is critical for the symbiosis.
Quorum sensing, far from just prompting bacteria to make the ghostly glow Dickens spoke of, is also critical for other bacterial properties, including many which have practical implications for humans. For example, the pathogen Pseudomonas aeruginosa uses N-(3-oxododecanoyl)homoserine lactone to sense how many of its fellow bacteria are around, which contributes to its virulence2>.
References
-
Biosynthesis and stereochemistry of the autoinducer controlling luminescence in <em>Vibrio harveyi<em>
J. Bacteriol.
1993
DOI 10.1128/jb.175.12.3856-3862.1993
-
The <em>Pseudomonas aeruginosa<em> Quorum-Sensing Molecule N-(3-Oxododecanoyl)Homoserine Lactone Contributes to Virulence and Induces Inflammation <em>In Vivo<em>
J. Bacteriol.
2002
DOI 10.1128/jb.184.4.1132-1139.2002
November 2020
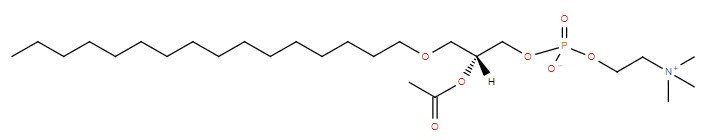
Turn the clock back 50 years, and phospholipids were considered solely structural components of cell membranes. However, in the 1970s a compound which caused platelet aggregation, blood clotting, and promoted inflammation was identified. It was named platelet-activating factor (PAF); and in 1979 shown to be a phospholipid 1
PAF is a phospholipid in which the glycerol has an alkyl group, typically of 16 carbons (though this varies) attached via an ether linkage at the sn-1 position. The sn-2 position is occupied by an acetate group while sn-3 position carries the phosphocholine headgroup. It interacts with a specific G-coupled protein receptor to cause some, if not all, of its effects and is active at picomolar concentrations.
PAF can be synthesised in two ways, either the ‘remodelling pathway’ by which it is synthesised from membrane alkyl/acyl phospholipids via the enzyme phoshpolipase A2 which removes the acyl chain at the sn-2 position, forming a lyso-phosphocholine. The sn-2 position is then enzymatically esterified with an acetyl group to form PAF. The other route to its synthesis is the ‘de novo’ route in which the alkylglycerol part is first formed, then the phosophocholine head group is added. Finally the acetyl group is added via the same enzyme as in the remodelling pathway.
Not only is this lipid involved in inflammatory and allergic processes, but it has also been implicated in cancers, and neurodegenerative diseases such as Alzheimers’2. Platelet activating factor does much more than activate platelets and there is doubtless much still to be learnt about this molecule.
References
-
Platelet-activating factor. Evidence for 1-O-alkyl-2-acetyl-sn-glyceryl-3-phosphorylcholine as the active component (a new class of lipid chemical mediators)
J. Biol. Chem
1979
-
Forty Years Since the Structural Elucidation of Platelet-Activating Factor (PAF): Historical, Current, and Future Research Perspectives
Molecules
2019
DOI 10.3390/molecules24234414
October 2020
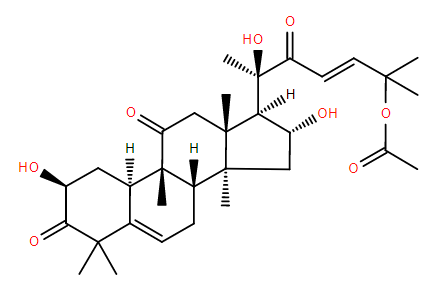
It’s October already and here in the northern hemisphere it is what the poet Keats described as ‘the season of mists and mellow fruitfulness’, which, he said, conspires with the sun to ‘swell the gourd’. October’s lipid of the month is found in, and named after, the gourd (Cucurbitaceae) family of plants, including Halloween and Thanksgiving’s iconic gourd, the pumpkin.
Cucurbitacin B is a particularly widespread cucurbitacin, present in many different plant species. Like other cucurbitacins it is a triterpenoid, a huge family of molecules formed from squalene which also encompasses cholesterol and the steroid hormones. Cucurbitacins often occur modified by sugars. Cucurbitacin B glucoside (LMST01010408), where a glucose is linked at the 2-position (the hydroxyl group towards the bottom left of the image) is also called arvenin I as it was first discovered in Anagallis arvensis- not a gourd but a primrose1.
Many cucurbitacins have a bitter taste, indeed arvenin I is described as ‘strongly bitter’, and are likely produced as a defense to try and prevent the plant (and the pumpkin) being eaten. Ironically, some of these taste properties appeal to humans, so the very things designed to protect, actually encourage us to eat them. Of course this is still beneficial for the plant, as human cultivation ensures success for a species.
Various pharmacological properties have been ascribed to Cucurbitacin B, including anti-inflammatory and anti-cancer properties against several different tumour types, such as those in the liver 2. It has even been reported to protect memory in a mouse-model of Alzheimers’ disease3.
So while LIPID MAPS is in no way giving you medical advice, remember if you’re making a pumpkin pie, carving a pumpkin lantern, or maybe both, there’s more to a pumpkin than a wholesome meal and seasonal fun.
References
-
Isolation and structures of arvenins from Anagallis arvensis L. (Primulaceae). New cucurbitacin glucosides
Chem Pharm Bull (Tokyo)
1978
DOI 10.1248/cpb.26.3107
-
Cucurbitacin B induces apoptosis and S phase cell cycle arrest in BEL-7402 human hepatocellular carcinoma cells and is effective via oral administration
Cancer Letters
2010
DOI 10.1016/j.canlet.2010.01.029
-
Cucurbitacin B induces neurogenesis in PC12 cells and protects memory in APP/PS1 mice
Cell Mol Med
2019
DOI 10.1111/jcmm.14514
September 2020
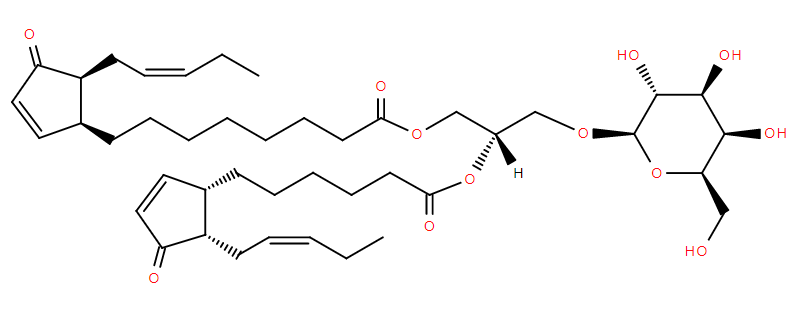
Absentmindedly crush a leaf while out on a country walk and you are likely to set up a whole cascade of reactions within the plant. One of them results in the formation of Arabidopside A1, a member of the arabidopside family of plant oxylipins discovered in the model plant, Arabidopsis thaliana.
The major lipids in plant membranes, particularly membranes of the chloroplast, are galactolipids, a glycerol esterified by two acyl chains and with a galactose sugar as the polar group. The arabidopsides are galactolipids produced in response to plant wounding or infection2. They are characterised by the presence of oxophytodienoic acid and/or dinor-oxophytodienoic acid as the acyl chains. Arabidopside A contains both of these species, the former at the sn1 position, and the latter at sn2. Phytodienoic acids are known plant hormones in their own right and precursors of the well known plant hormone jasmonic acid. They are chemically similar to the mammalian inflammatory signalling molecules, the prostaglandins.
Exactly how the arabidopsides function isn’t really known, it’s been suggested that they are reservoirs of phytodienoic acids which can be released when needed, but that doesn’t explain why their concentration increases when a plant is wounded. Just imagine how much arabidopside you might cause a plant to make when you mow the lawn!
References
-
Arabidopsides A and B, two new oxylipins from Arabidopsis thaliana
Tetrahedron Letters
1993
DOI 10.1016/S0040-4039(03)01148-1
-
Wounding Stimulates the Accumulation of Glycerolipids Containing Oxophytodienoic Acid and Dinor-Oxophytodienoic Acid in Arabidopsis Leaves
Plant Physiology
2006
DOI 10.1104/pp.106.082115
August 2020
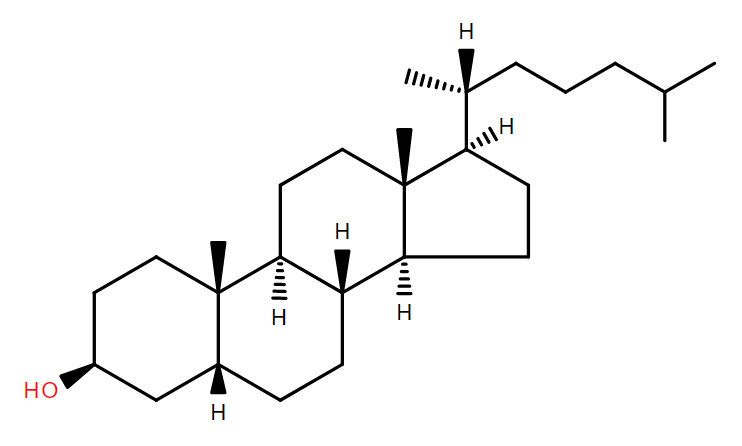
Coprostanol, August’s lipid of the month, is used as a biomarker indicating the presence of sewage in the environment. It is formed by gut bacteria which reduce dietary cholesterol to produce coprostanol, a highly water-insoluble molecule which, in anaerobic conditions, can be stable for thousands of years. This means that coprostanol is not only useful to monitor modern cases of sewage contamination, but can also be used as an indicator of the presence of sewage in archaeological sites. Its very poor water solubility means that any coprostanol in a sample is likely to have originated within the sample, rather than have been deposited from contaminated water flowing through the area over time.
Just as cholesterol is reduced to coprostanol, other dietary sterols are reduced to their respective stanols by gut bacteria. The plant-based diet of ruminant animals results in more faecal stigmastanol, produced from sitosterol. Stanol biomarker ratios therefore can be diagnostic of the species of the faeces[1].
A recent study in Science Advances[2] analysed stanol biomarkers in 12400 year old coprolites (fossilised excreta) found in an Oregon cave. DNA evidence had previously suggested these were human in origin, but a question of contamination with modern DNA remained. Because coprostanol is not able to seep over time into a sample through contaminated water, and contamination during analysis is extremely unlikely (unless you’re doing science really badly!), the ratio of coprostanol to other sterols in the sample confirmed them to be “unequivocally” human in origin. This research adds to the body of knowledge and the debate surrounding the first human settlement of the Americas.
References
-
The origin of faeces by means of biomarker detection
Environment International
2002
DOI 10.1016/s0160-4120(01)00124-6
-
Pre-Clovis occupation of the Americas identified by human fecal biomarkers in coprolites from Paisley Caves, Oregon
Science Advances
2020
DOI 10.1126/sciadv.aba6404
July 2020
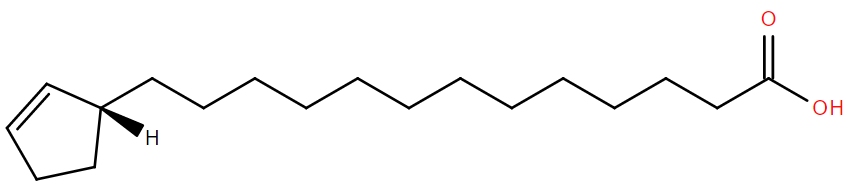
July’s lipid of the month, chaulmoogric acid, is an ingredient in the first effective leprosy treatment. Found in the seeds of the chaulmoogra tree (Hydnocarpus wightianus), it’s an unusual fatty acid containing a cyclopentenyl group which is thought to act as a biotin mimic, though its mode of action isn’t really known.
Chaulmoogra oil had been used for centuries as an ointment to treat leprosy, but the viscous, insoluble and noxious substance was unsuitable for internal treatment. Alice Ball, a pharmaceutical chemist at the University of Hawaii solved that by developing a method of solubilising the (presumed) active ingredients of chaulmoogra oil, chaulmoogric acid and the related hydnocarpic acid, in useful quantities. Ball’s method was essentially saponification to release the fatty acids from triglycerides and then esterification.
Born in July 1892, Ball died aged only 24, before her method could be published and before she could know how effective it was. The work was continued, with no reference to Ball, by Arthur Dean, the preparation of Chaulmoogra oil becoming known as ‘Dean derivatives’. A review by Harry T Hollmann in 1922[1] attempted to set the record straight. Hollmann, a doctor at Kalhihi hospital in Hawaii where leprosy patients were sent, had invited Ball to look into producing a soluble chaulmoogra oil derivative. In the review he explains how Ball, the first woman, and first African American to receive a Masters degree from the College of Hawaii, developed the process of producing ethyl esters of chaulmoogra oil. Under the name ‘Dean derivatives’, these were credited with curing 78 patients at the Kalhihi hospital, who were discharged “no longer a menace to society”[2]. Alice Ball’s preparation remained the treatment for leprosy until the advent of antibiotics in the 1940s.
You can read more about Alice Ball in Brianna Bibel’s excellent blog, ‘The Bumbling Biochemist’
References
-
The Fatty Acids Of Chaulmoogra Oil In The Treatment Of Leprosy And Other Diseases
Arch Derm Syphilol.
1922
DOI 10.1001/archderm.1922.02350260097010
-
Treatment Of Leprosy With The Dean Derivatives Of Chaulmoogra Oil
J.Am.Med.Assoc.
1920
DOI 10.1001/jama.1920.02620480021007
June 2020
Fatty acid esters of hydroxyl fatty acids (FAHFAs, or estolides) are a fairly recent addition to the known mammalian lipidome. They were described in 2016 as a class of lipids with both anti-diabetic and anti-inflammatory effects [1]. Given the combinatorial possibilities of linking a fatty acid with a further hydroxylated fatty acid, where the hydroxyl could be anywhere along the chain, the FAHFA family has potential to be vast.
One example, the linoleic acid ester of 13-hydroxylinoleic acid (13-LAHLA)[2] has been shown to have anti-inflammatory effects. It suppresses expression of genes involved in inflammation such as interleukins 6 and 1beta. It also suppresses expression of cyclooxygenase 2, an enzyme in the prostaglandin synthesis pathway and the target of the commonly used drug ibuprofen.
While it is known that some FAHFAs are ligands for G-protein coupled receptors, their precise mechanism of action, and indeed the full extent of the family, remains to be worked out. There is still much work to be done.
References
-
Discovery of a class of endogenous mammalian lipids with anti-diabetic and anti-inflammatory effects
Cell
October 2014
DOI 10.1016/j.cell.2014.09.035
-
Linoleic acid esters of hydroxy linoleic acids are anti-inflammatory lipids found in plants and mammals
J.Biol.Chem
May 2019
DOI 10.1074/jbc.RA118.006956
May 2020
Sterol lipids function as hormones across a wide range of species and can have a dramatic effect on the morphology of an individual. 20-Hydroxyecdysone is one such sterol hormone which controls skin-shedding and body pattern in arthropods such as crabs and insects. Its effects on an individual can be dramatic.
A recent paper [1] from Antonia Monteiro’s lab showed 20-Hydroxyecdysone to have a pivotal role in controlling the size of ‘eye spots’ on the wings of the African butterfly Bicyclus anynana. Adults which emerge from their pupae in the wet season have large eyespots on their wings but those which reach adulthood in the dry season, lack eye spots and are a more dull brown colour.
This difference is determined by the temperature experienced by the caterpillar over two specific days of its life, just before it pupates. While many species show a difference in levels of 20-Hydroxyecdysone depending on temperature, Monteiro showed that in B. anynana, cells in the centre of what would become the eyespot contained the 20-Hydroxyecdysone receptor, making them very sensitive to the hormone.
20-Hydroxyecdysone is not just made by arthropods. Some plants also produce this hormone, presumably as a defence mechanism, disrupting the development of insects grazing on them.
References
-
Origin of the mechanism of phenotypic plasticity in satyrid butterfly eyespots
eLife
February 2020
DOI 10.7554/eLife.49544
April 2020
Humans are unable to synthesise linoleic acid, and so we must obtain it through the food we eat. Linoleic acid was discovered to be essential in the diet by Mildred and George Burr, working at the University of Minnesota in 1930[1]. They showed that fats were not simply of calorific value but some, including linoleic acid, were absolutely required for health.
Linoleic acid is an 18-carbon fatty acid with two double bonds and present in large amounts in many vegetable oils. It’s one of those polyunsaturated fats which the adverts tell us are good for us.
It’s a precursor to arachidonic acid, which in turn is a precursor to a great many lipidic signalling molecules in the body. A study published in January 2019 looked at the effect of coronavirus infection on cellular lipids and showed that linoleic to arachidonic acid metabolism was “the most perturbed pathway” on infection of cells in their model system.[2]
References
-
On the nature and role of the fatty acids essential in nutrition
J. Biol. Chem.
January 1930
-
Characterization of the Lipidomic Profile of Human Coronavirus-Infected Cells: Implications for Lipid Metabolism Remodeling upon Coronavirus Replication.
Viruses
2019, 11(1), 73
DOI 10.3390/v11010073
March 2020
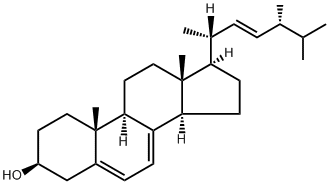
Ergosta-5,7,22E-trien-3β-ol (Ergosterol) is a sterol found in cell membranes of fungi and protozoa, but not animals. The common name, "ergosterol", of this lipid is derived from the group of fungi from which it was first isolated — "ergot" fungi, of genus Claviceps.
The role of ergosterol in fungi is similar to that of cholesterol in animal cells and ergosterol is essential to their survival. Ergosterol is synthesized from lanosterol and several anti-fungal drugs work by targeting erogsterol — either by binding to it, resulting in devastating cellular leakage, or preventing the synthesis of ergosterol from lanosterol.
References
February 2020
9-oxo-11R,15S-dihydroxy-5Z,13E-prostadienoic acid(PGE2) is an oxytocic prostaglandin. PGE2's synthetic equivalent, the drug "dinoprostone", is commonly administered during childbirth — to induce labor by stimulating uterine contraction; to promote cervical ripening; and to control postpartum hemorrhage (PPH). PGE2 has also been shown to be involved in stem cell development via modulation of the Wnt pathway.
References
-
Nobiletin: efficient and large quantity isolation fromorange peel extract
Biomed. Chromatogr
20: 133 –138 (2006)
DOI 10.1002/bmc.540
-
Nobiletin restoring beta-amyloid-impaired CREB phosphorylation rescues memory deterioration in Alzheimer's disease model rats.
Neurosci Lett.
2006 Jun 12,400(3):230-4. Epub 2006 Apr 3.
DOI 10.1016/j.neulet.2006.02.077
-
Nobiletin and Derivatives: Functional Compounds from Citrus Fruit Peel for Colon Cancer Chemoprevention
Cancers
2019, 11(6), 867
DOI 10.3390/cancers11060867
-
The citrus flavonoid nobiletin confers protection from metabolic dysregulation in high-fat-fed mice independent of AMPK
J. Lipid Res.
First Published on January 21, 2020
DOI 10.1194/jlr.RA119000542
January 2020
4Z,7Z,10Z,13Z,16Z,19Z-docosahexaenoic acid (DHA) - Fatty Acids and Conjugates [FA01]-> Unsaturated fatty acids [FA0306] - is a long-chain omega-3 polyunsaturated fatty acid [PUFA] commonly found in brain and retina[1,2]. DHA is thought to play a critical role in brain developent, to be involved in multiple aspects of cardiovascular function[3], and is implicated in multiple diseases and developmental disorders when deficient[1,3].
While DHA levels can be measured using isotope tracers, recent research demonstrates that it can be measured by natural abundance carbon isotope ratio analysis, which is less costly and generally safer[4].
References
-
Essential Fatty Acids: the Importance of n-3 Fatty Acids in the Retina and Brain
Nutrition Reviews
Volume 50, Issue 4, April 1992, Pages 21–29
DOI 10.1111/j.1753-4887.1992.tb01286.x
-
Essential role of docosahexaenoic acid towards development of a smarter brain
Neurochem Int
2015 Oct;89:51-62. Epub 2015 Aug 28
DOI 10.1016/j.neuint.2015.08.014
-
Omega-3 fatty acids EPA and DHA: health benefits throughout life
Adv Nutr
2012 Jan;3(1):1-7. Epub 2012 Jan 5
DOI 10.3945/an.111.000893
-
Turnover of brain DHA in mice is accurately determined by tracer-free natural abundance carbon isotope ratio analysis
Journal of Lipid Research
January 2020, 61, 116-126
DOI 10.1194/jlr.D119000518
December 2019
5-oxo-6E,8Z,11Z,14Z-eicosatetraenoic acid (5-Oxo-ETE) — Eicosanoids [FA03] -> Hydroxy/hydroperoxyeicosatetraenoic acids [FA0306] — is a G-protein-coupled receptor (GPCR)1 metabolite of the Arachadonic Acid Cascade2 involved in both autocrine and paracrine signaling. 5-Oxo-ETE is thought to be involved in inflammation3 and recent research suggests that it may play a role in the disorder granulomatosis with polyangiitis4.
References
-
International Union of Pharmacology XLIV. Nomenclature for the Oxoeicosanoid Receptor
Pharmacological Reviews
March 2004, 56 (1) 149-157
DOI 10.1124/pr.56.1.4
-
Biochemistry, biology and chemistry of the 5-lipoxygenase product 5-oxo-ETE
Prog Lipid Res.
2005 Mar-May;44(2-3):154-83
DOI 10.1016/j.plipres.2005.04.002
-
5-Oxo-6,8,11,14-eicosatetraenoic acid induces the infiltration of granulocytes into human skin
Journal of Allergy and Clinical Immunology
October 2003 Volume 112, Issue 4, Pages 768–774
DOI 10.1016/S0091-6749(03)01888-8
-
LTB4 and 5-oxo-ETE from extracellular vesicles stimulate neutrophils in granulomatosis with polyangiitis
J Lipid Res.
2019 Nov 8
DOI 10.1194/jlr.M092072
November 2019
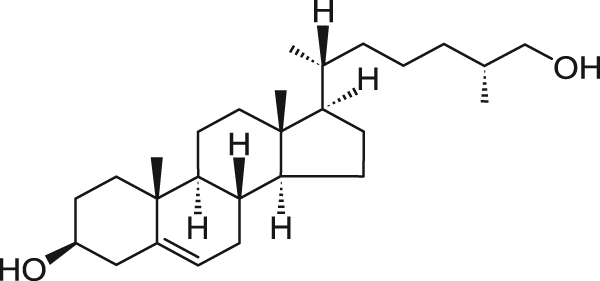
27-hydroxy-cholesterol is a sterol lipid metabolite of cholesterol1 that has been found to be involved in many biological functions, including metastasis of breast cancer2 and alzheimer's disease3. 27-hydroxy-cholesterol has also recently been shown to be present in human breast milk — at a notably elevated concentrations in colustrum — possibly serving an important role in shielding newborn babies from rotavirus and rhinovirus infection4.
References
-
LIPID MAPS® WikiPathways: Cholesterol metabolism
https://www.lipidmaps.org/resources/pathways/wikipathways.php?ID=WP4346
-
27-Hydroxycholesterol Links Hypercholesterolemia and Breast Cancer Pathophysiology
Science
2013 Nov 29; 342(6162): 1094–1098.
DOI 10.1126/science.1241908
-
A Crosstalk Between Brain Cholesterol Oxidation and Glucose Metabolism in Alzheimer's Disease.
Front Neurosci.
2019 May 31;13:556
DOI 10.3389/fnins.2019.00556
-
Antiviral oxysterols are present in human milk at diverse stages of lactation
The Journal of Steroid Biochemistry and Molecular Biology
Volume 193, October 2019, 105424
DOI 10.1016/j.jsbmb.2019.105424
October 2019
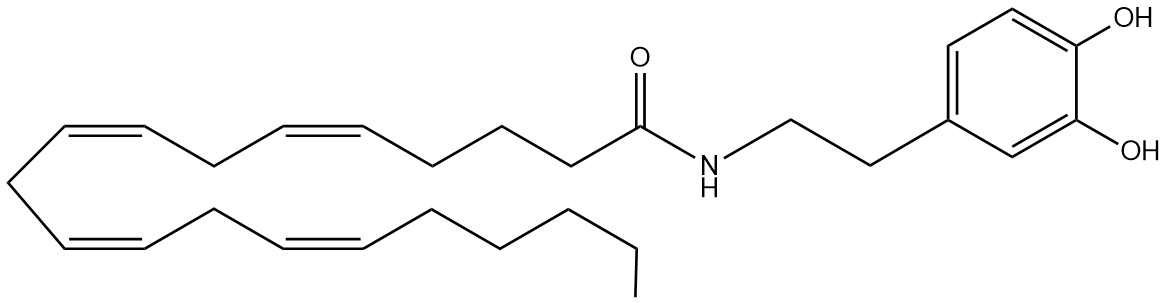
N-Arachidonoyl dopamine or NADA is a fatty acyl that belongs to the fatty amide class and fatty acyl amine sub class. It is an endogenous lipid that is found primarily in brain tissue in mammals1. Due to its structural similarity to N-acyl ethanolamines, NADA may also function as endocannabinoid2 and plays a regulatory role in both the peripheral and central nervous systems, and shows antioxidant and neuroprotectant properties 2. NADA, as ligand of the endocannabinoid receptor system, is involved in all aspects of mammalian physiology and pathology, and for this reason it represents a potential target for the design and development of new therapeutic drugs 3.
References
-
Endocannabinoids and endocannabinoid-related mediators: Targets, metabolism and role in neurological disorders.
Progress in Lipid Research
Volume 62, April 2016, Pages 107-128
DOI 10.1016/j.plipres.2016.02.002
-
The Endocannabinoid/Endovanilloid N-Arachidonoyl Dopamine (NADA) and Synthetic Cannabinoid WIN55,212-2 Abate the Inflammatory Activation of Human Endothelial Cells
Journal of Biological Chemistry
Volume 289, Issue 19 pp.13079–13100
DOI 10.1074/jbc.M113.536953
-
N-Arachidonoyl Dopamine: A Novel Endocannabinoid and Endovanilloid with Widespread Physiological and Pharmacological Activities.
Cannabis and Cannabinoid Research
Volume 2, Issue 1, 2017, pp. 183-196
DOI 10.1089/can.2017.0015
September 2019
Cortisol or 11β,17,21-trihydroxypregn-4-ene-3,20-dione (LMST02030001) is a sterol lipid member of the class of steroid hormones known as glucocorticoids. It is produced by the adrenal glands and regulated by the hypothalamus and pituitary in humans, and many other animals1. It is well known for helping our body’s “fight-or-flight” instinct in a crisis2, but it is also important for many other physiological processes such as: regulating blood pressure and the use of nutrients (carbohydrates, fats, and proteins2,3, preventing inflammation4, increasing glucose levels4, controlling the sleep cycle and balancing energy in order to handle stress3,5. Cortisol depletion or excess affects the body’s functions and can lead to many health problems if not monitored regularly5.
References
-
The use of hair cortisol for the assessment of stress in animals
General and Comparative Endocrinology.
Volume 270, 2019, pp. 10-17.
DOI 10.1016/j.ygcen.2018.09.016
-
Investigating associations between momentary stress and cortisol in daily life: What have we learned so far?
Psychoneuroendocrinology.
Volume 105, 2019, pp. 105-116.
DOI 10.1016/j.psyneuen.2018.11.038
-
Exercise, the diurnal cycle of cortisol and cognitive impairment in older adults
Neurobiology of Stress.
Volume 9, November 2018, Pages 40-47
DOI 10.1016/j.ynstr.2018.08.004
-
Fructose-induced inflammation and increased cortisol: A new mechanism for how sugar induces visceral adiposity
Progress in Cardiovascular Diseases
Volume 61, Issue 1, 2018, pp. 3-9.
DOI 10.1016/j.pcad.2017.12.001
-
Redefining neuroendocrinology: Epigenetics of brain-body communication over the life course
Frontiers in Neuroendocrinology
Volume 49, 2018, pp. 8-30.
DOI 10.1016/j.yfrne.2017.11.001
August 2019
α-N-(3-hydroxyhexadecanoyl) L-ornithine(LMFA08020242) is an ornithine lipid (OL) that belongs to the fatty acyl category, fatty amide class and N-acyl amine sub-class. OL do not contain either glycerol or phosphate1,3 in their chemical structure and are found in many biological species such as mammals, plants and bacteria2. α-N-(3-hydroxyhexadecanoyl) L-ornithine in particular could be hydroxylated by hydroxylases to form much larger analogous forms3. This particular phenomena, that happens in some bacterial species, is considered a stress response to changing environmental conditions occurring in the membrane (e.g. phosphate-limiting growth conditions, high temperature or acid tolerance)1 that results in simply modifying already existing lipids without the need to synthesize new ones.
References
-
Characterization of Ornithine and Glutamine Lipids Extracted from Cell Membranes of Rhodobacter sphaeroides
Journal of the American Society for Mass Spectrometry
Volume 20, Issue 2,2009, 198-212
DOI 10.1016/j.jasms.2008.08.017
-
The LipidWeb: Simple N-Acylamides and Lipoamino Acids
https://www.lipidmaps.org/resources/lipidweb/index.php?page=lipids/simple/lipamino/index.htm
-
Ornithine lipids and their structural modifications: from A to E and beyond.
FEMS Microbiology Letters
Volume 335, 2012, pp. 1–10.
DOI 10.1111/j.1574-6968.2012.02623.x
July 2019
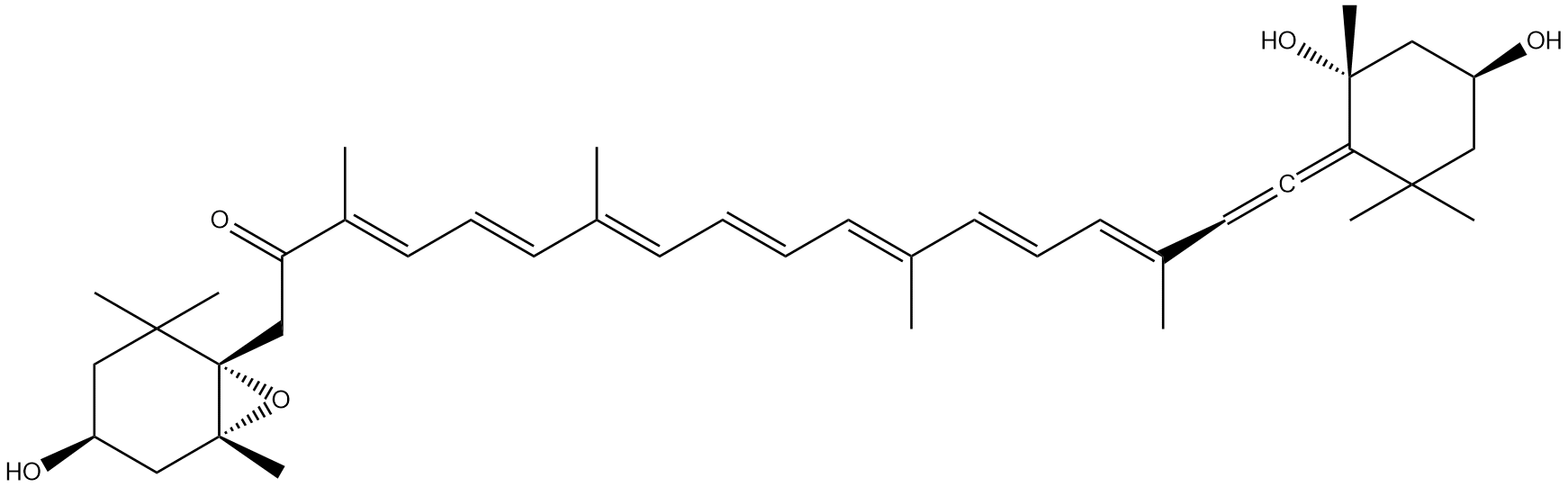
Fucoxanthinol(LMPR01070055) is a prenol lipid that belongs to the isoprenoid class and tetraterpenoid sub-class in LIPID MAPS®. It is also a type of carotenoid found typically in algae, higher plants and photosynthetic bacteria 1. In mammals, fucoxanthinol is obtained from metabolism of fucoxanthin by digestive enzymes (hydrolysis) in the gastrointestinal tract. This is then absorbed directly in the intestine and transported into the circulation and the liver2. Fucoxanthinol has been proposed to have beneficial characteristics such as antioxidant, anticancer, anti-obesity, antidiabetic, and anti-photo-aging properties1,3.
References
-
Pharmacokinetics of fucoxanthinol in human plasma after the oral administration of kombu extract.
British Journal of Nutrition
Volume 107, Issue 11, 2012 , pp. 1566-1569
DOI 10.1017/S0007114511004879
-
A novel strategy for isolation and purification of fucoxanthinol and fucoxanthin from the diatom Nitzschia laevis
Food Chemistry.
Volume 277, 30 March 2019, Pages 566-572
DOI 10.1016/j.foodchem.2018.10.133
-
Fucoxanthin and Its Metabolite Fucoxanthinol in Cancer Prevention and Treatment
Mar Drugs.
Volume 13, Issue 8, pp. 4784–4798.
DOI 10.3390/md13084784
June 2019
Arachidonic acid (LMFA01030001) or 5Z,8Z,11Z,14Z-eicosatetraenoic acid is an unsaturated fatty acid that can be found in plasma membranes where it is esterified to phospholipids (especially phosphatidylethanolamine, phosphatidylcholine, and phosphatidylinositols)1,2. Arachidonic acid (AA) and its metabolite by-products (eicosanoids) are important mediators of physiological processes, and these include prostaglandins, prostacyclin, thromboxane, and leukotrienes1,2,3,4. Arachidonic acid is obtained from food or by desaturation/chain elongation of the plant-rich (i.e. green and red algae) essential fatty acid, linoleic acid2,4.
References
-
Pathobiology of CNS Human Immunodeficiency Virus Infection
Neurobiology of Brain Disorders
Biological Basis of Neurological and Psychiatric Disorders 2015, Pages 444-466
DOI 10.1016/B978-0-12-398270-4.00028-8
-
Prostaglandins and Leukotrienes
Encyclopaedia of Food Sciences and Nutrition.
S. Katayama and J. B. Lee. (Second Edition). 2003, Pages 4798-4804.
-
Arachidonic acid: Physiological roles and potential health benefits – A review.
Journal of Advanced Research
Volume 11, May 2018, Pages 33-41.
DOI 10.1016/j.jare.2017.11.004
-
A review on algae and plants as potential source of arachidonic acid
Journal of Advanced Research
Volume 11, May 2018, Pages 3-13
DOI 10.1016/j.jare.2018.03.004
May 2019
17β-Estradiol (LMST02010001) is a sterol lipid classified as a C18 steroid estrogen in LIPID MAPS®. It is the most potent type of the four existent estrogens (also known as E2)1 and it is produced within the follicles of the ovaries and therefore considered as the most important female hormone that regulates menstrual cycles and reproduction. 17β-estradiol can be found in other tissues including the testicles, adrenal glands, fat, liver, breasts and brain1,2,3 and in other species such as crustaceans, insects and fish4. Its synthetic form has been widely used for menopause and hormonal disbalance treatments1,3. However, when disposed through wastewater discharges to natural environments like rivers and ponds, and found in large concentrations it could alter the aquatic ecosystem4.
References
-
The structural biology of oestrogen metabolism
Journal of Steroid Biochemistry & Molecular Biology.
Volume 137 (2013) pp. 27–49
DOI 10.1016/j.jsbmb.2012.12.014
-
PubChem Estradiol (Compound)
https://pubchem.ncbi.nlm.nih.gov/compound/estradiol#section=CAS
-
Follicle-stimulating hormone promotes the transformation of cholesterol to estrogen in mouse adipose tissue
Biochemical and Biophysical Research Communications.
Volume 495, Issue 3, 15 January 2018, Pages 2331-2337
DOI 10.1016/j.bbrc.2017.12.120
-
Pharmaceuticals in freshwater aquatic environments: A comparison of the African and European challeng
Science of The Total Environment.
Volume 654, March 2019, Pages 324-337
DOI 10.1016/j.scitotenv.2018.11.072
April 2019
Testosterone is a sterol lipid classified as a C19 androgen. It is excreted by testicular Leydig cells in males (1,2) and plays and important role in sexual development, body composition (bone mass, fat distribution, muscle mass and strength) and regulating general health and wellbeing (1,2,3). As a natural hormone, it is found in males and females but at different concentrations (7:1 male to female ratio). In men, testosterone decreases with age, and in younger males, a clinical deficit can be treated with a drug version (3). For many years, controversy on whether high or low levels of testosterone are related to prostate cancer has been a topic of much discussion and more research is required to provide a better picture on the real causes of developing this disease (1,3,4).
References
-
Role of androgens in energy metabolism affecting on body composition, metabolic syndrome, type 2 diabetes, cardiovascular disease, and longevity: lessons from a meta-analysis and rodent studies
Journal of Bioscience, Biotechnology, and Biochemistry
Volume 82, 2018 - Issue 10. Pages 1667-1682.
DOI 10.1080/09168451.2018.1490172
-
Understanding How Testosterone Affects Men
https://www.nih.gov/news-events/nih-research-matters/understanding-how-testosterone-affects-men
-
Low Free Testosterone and Prostate Cancer Risk: A Collaborative Analysis of 20 Prospective Studies.
European Urology.
Volume 74, Issue 5, November 2018, Pages 585-594.
DOI 10.1016/j.eururo.2018.07.024
-
The testosterone conundrum: The putative relationship between testosterone levels and prostate cancer
Seminars and Original Investigations.
Volume 34, Issue 11, November 2016, Pages 482.e1-482e4.
DOI 10.1016/j.urolonc.2016.05.023
March 2019
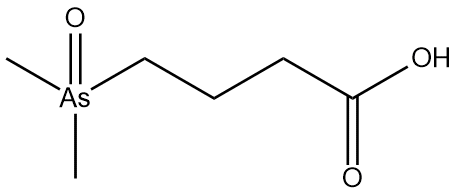
4-Dimethylarsenoyl butanoic acid also known as DMAB is an organometallic fatty acid that can be found in human urine as a metabolite of larger arsenolipids species containing up to 20 carbons and consumed in seafood (1,3). Exposure to soluble inorganic forms of arsenic has been widely reported as highly toxic and its consumption through drinking water is linked to chronic human health problems including cancer (skin and bladder)(1). Over the last 30 years over 40 naturally occurring water and fat soluble organo-arsenicals such DMAB have been identified in fish, algae and crustaceans at very high concentrations (over 100 µgAs/g of wet mass)(1-4). Although the majority of DMBA and other arsenolipid metabolites are excreted through urine after consumption (85%), their toxicity and fate of the residual amount of arsenic content in the body remains unknown(1,4).
References
-
Arsenic Fatty Acids Are Human Urinary Metabolites of Arsenolipids Present in Cod Liver
Angewandte Chemie International Edition.
Volume 45, Issue 1, December 16, 2005, pp. 150-154.
DOI 10.1002/anie.200502706
-
Chronic health effects in people exposed to arsenic via the drinking water: dose–response relationships in review
Toxicology and Applied Pharmacology.
Volume 198, Issue 3, 1 August 2004, pp. 243-252.
DOI 10.1016/j.taap.2003.10.022
-
Arsenolipids in marine oils and fats: A review of occurrence, chemistry and future research needs
Food Chemistry
Volume 133, Issue 3, 1 August 2012, Pages 618-630
DOI 10.1016/j.foodchem.2012.02.004
-
Arsenic species in seafood: Origin and human health implications
Pure Applied Chemistry
Volume 82, No. 2, 2010. pp. 373–381
DOI 10.1351/PAC-CON-09-07-01
February 2019
R-(-)-Linalool is a prenol lipid found naturally in many flowers and spices. Prenol lipids are known as terpenoids which are one of the largest groups of naturally occurring compounds synthesised by plants (1,3). (-)-Linalool is one of the major constituents of lavender (Lavandula angustifolia) and coriander (Coriandrum sativum L.) essential oils. Similar to other terpenoids such geraniol (geraniol) and citronellol (citronellol), (-)-Linalool has been reported to have bioactive properties in vitro and in vivo (3), including anti-inflammatory, antifungal, antimicrobial, anti-depressive (2,3), anticancer, antinociceptive, analgesic, anxiolytic and neuroprotective (3). These properties are being commercially exploited by several industries including agronomic, food, sanitary, cosmetic and pharmaceutical (2,3).
References
-
Metabolism of plant terpenoids
Progress in the Chemistry of Fats and other Lipids
Volume 10, 1970, Pages 153-212, IN1-IN2, 213-238
DOI 10.1016/0079-6832(70)90006-6
-
Biological effects of essential oils – A review
Food and Chemical Toxicology
Volume 46, Issue 2, February 2008, Pages 446-475
DOI 10.1016/j.fct.2007.09.106
-
Linalool bioactive properties and potential applicability in drug delivery systems
Colloids and Surfaces B: Biointerfaces
Volume 171, 1 November 2018, Pages 566-578
DOI 10.1016/j.colsurfb.2018.08.001
January 2019
3-carboxy-4-methyl-5-propyl-2-furan propanoic acid or CMPF is a heterocyclic fatty acid, and a major metabolite of furan fatty acids. Furan fatty acids naturally occur in algae, plants, organic dairy products, fish and some bacteria(1,2,3,4). Increased levels of CMPF have been found in patients with diabetes type 2 and recent studies have shown some evidence indicating that CMPF may be a biomarker for diabetes T2 progression(1,3) and renal impairment(1). Humans acquire furan fatty acids from the consumption of a wide range of foods such as vegetables, fruits, seed oil, dairy products, fish and fish oil(1,2,4). These compounds are scavengers of free radicals and can protect polyunsaturated fatty acids from oxidation(1,4)and at the same time they can be mainly metabolised to CMPF(1) raising the debate whether furan fatty acids intake could be harmful or beneficial for health.
References
-
Furan fatty acids – Beneficial or harmful to health?
Progress in Lipid Research.
Volume 68, 2017, Pages 119-137.
DOI 10.1016/j.plipres.2017.10.002
-
High Concentrations of Furan Fatty Acids in Organic Butter Samples from the German Market.
J. Agric. Food Chem.
2014, 62 (34), pp 8740–8744
DOI 10.1021/jf502975b
-
Detailed Study of Furan Fatty Acids in Total Lipids and the Cholesteryl Ester Fraction of Fish Liver.
Food Analytical Methods
Volume 9, Issue 2, 2016, pp 459–468
DOI 10.1007/s12161-015-0211-x
-
Synthesis and scavenging role of furan fatty acids
Proceedings of the National Academy of Sciences of the United States of America.
Volume 111, Issue 33, 2014, pp. 3450-57.
DOI 10.1073/pnas.1405520111 2018
December 2018
2-Arachidonoylglycerol also known as 2-AG is a monoacylglycerol neurotransmitter that binds to cannabinoid receptors located in the mammalian central nervous system (including the brain) and peripheral nervous system 1. 2-AG and anandamide (LMFA08040001) are involved in regulating a variety of physiological and cognitive processes as part of the endocannabinoid system (ECS) which includes fertility, pregnancy, appetite, pain-sensation, mood (depression and memory) and in mediating the pharmacological effects of cannabis (1,2,3). In contrast, ECS is up-regulated in numerous pathophysiological states such as inflammatory, neurodegenerative, gastrointestinal, metabolic and cardiovascular diseases, pain, and cancer therefore enhancement of endogenous endocannabinoid tone by inhibition of endocannabinoid degradation represents a promising therapeutic approach for the treatment of many diseases (4).
References
-
Endocannabinoids, exercise, pain, and a path to health with aging
Molecular Aspects of Medicine.
Volume 64, December 2018, pp. 68-78.
DOI 10.1016/j.mam.2018.10.001
-
The endocannabinoid system in mental disorders: Evidence from human brain studies
Biochemical Pharmacology
Volume 157, 2018, pp. 97-107
DOI 10.1016/j.bcp.2018.07.009
-
Chronic pain and cannabinoids. Great expectations or a Christmas carol.
Biochemical Pharmacology,
Volume 157, 2018, pp.33-42.
DOI 10.1016/j.bcp.2018.07.033
-
Enhanced endocannabinoid tone as a potential target of pharmacotherapy
Life Sciences
Volume 204, 1 July 2018, pp. 20-45.
DOI 10.1016/j.lfs.2018.04.054
November 2018
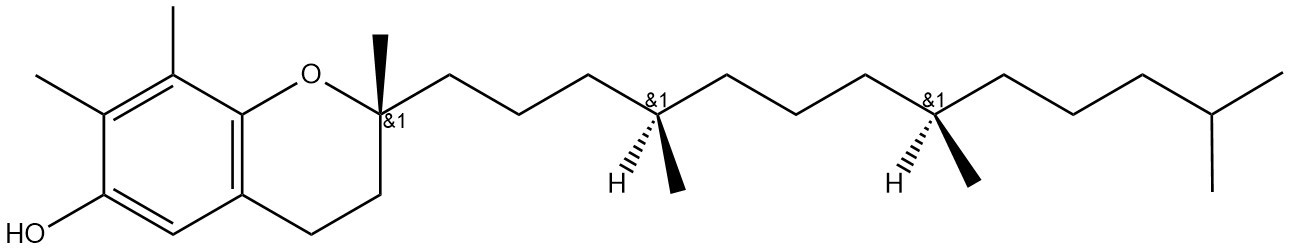 γ-Tocopherol
is one of the eight types of vitamin E and abundantly found in seeds (almond, pumpkin, walnuts, pecans, etc.) It has antioxidant properties due to its capability to trap nitrogen-based free radicals or reactive nitrogen oxide species (RNOS). However, when γ-tocopherol is metabolised to 2,7,8-trimethyl-2-(β-carboxyethyl)-6-hydroxychroman (γ-CEHC) it inhibits cyclooxygenase (COX-2) and prostaglandin E2 activity exhibiting also anti-inflammatory properties(1). Many serious degenerative conditions, including atherosclerosis, various types of cancer and Alzheimer's disease, appear to be promoted by chronic inflammation and COX-2 inhibitors show great potential in the prevention of these diseases.
γ-Tocopherol
is one of the eight types of vitamin E and abundantly found in seeds (almond, pumpkin, walnuts, pecans, etc.) It has antioxidant properties due to its capability to trap nitrogen-based free radicals or reactive nitrogen oxide species (RNOS). However, when γ-tocopherol is metabolised to 2,7,8-trimethyl-2-(β-carboxyethyl)-6-hydroxychroman (γ-CEHC) it inhibits cyclooxygenase (COX-2) and prostaglandin E2 activity exhibiting also anti-inflammatory properties(1). Many serious degenerative conditions, including atherosclerosis, various types of cancer and Alzheimer's disease, appear to be promoted by chronic inflammation and COX-2 inhibitors show great potential in the prevention of these diseases.
References
-
N/A
October 2018
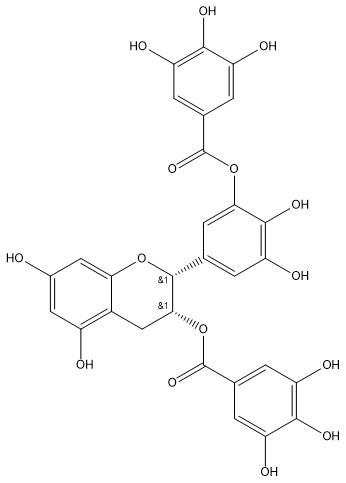
Epigallocatechin 3,3',-di-O-gallate, also known as EGCG is a polyketide lipid that belongs to the main class of flavonoids known as catechins which can be mainly derived or extracted naturally from green and black tea(1). However, as a flavonoid, this compound can be found in many other food products such cocoa, onions, plums, pecan and hazelnuts but in trace amounts. The widespread consumption of green tea around the world has been promoted due to its positive health effects attributed to the antioxidant and anti-inflammatory properties of compounds such EGCG contained within(2. However, other studies have highlighted that EGCG might protect from oxidative damages and could reduce cancer risk or other diseases caused by free radicals(3).
References
-
Medicinal Plants: Their Use in Anticancer Treatment.
Int Journal of Pharmacological Science Research
Volume 6. Issue 10, 2005, pp. 4103–4112
DOI 10.13040/IJPSR.0975-8232.6(10).4103-12
-
Green tea polyphenol epigallocatechin-3-gallate differentially modulates oxidative stress in PC12 cell compartments
Toxicology and Applied Pharmacology.
Volume 15. Issue 207(3), pp. 212-20.
DOI 10.1016/j.taap.2005.01.004
-
Effect of increased tea consumption on oxidative DNA damage among smokers: a randomized controlled study
Journal of Nutrition
Volume 133. Issue 10, 2003, pp. 3303S–3309S
DOI 10.1093/jn/133.10.3303S
September 2018
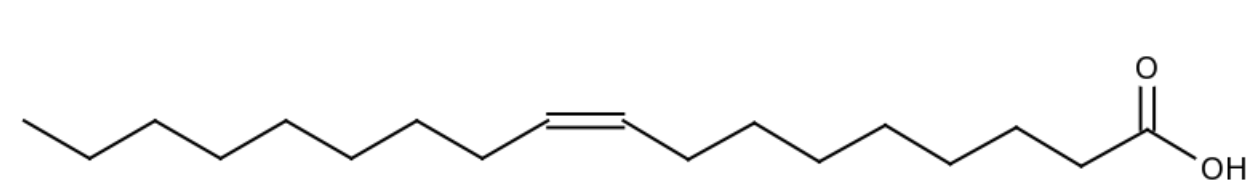
Oleic acid (Systematic Name:9Z-octadecenoic acid) is a monounaturated fatty acid whose name derives from the latin word 'oleum', which means oil. It is the most common fatty acid in nature, typically occurring in esterified form in glycerolipids and phospholipids. The biosynthesis of oleic acid involves the action of the enzyme stearoyl-CoA 9-desaturase acting on the saturated precursor stearoyl-CoA.
References
-
N/A
August 2018
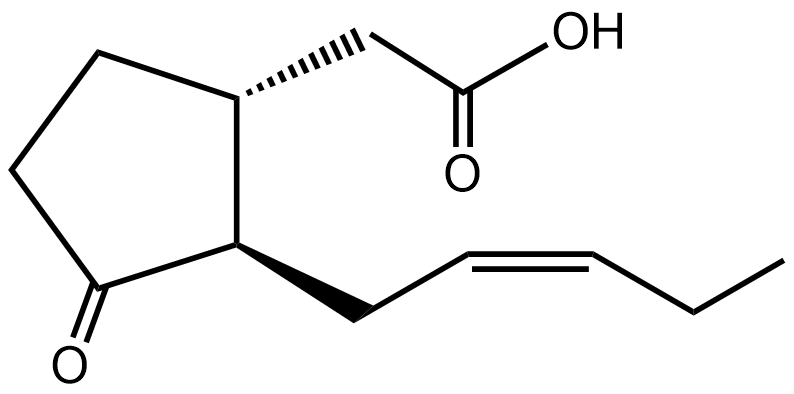
Jasmonic acid (systematic name: (1R,2R)-3-oxo-2-(pent-2Z-enyl)-cyclopentaneacetic acid) is an truncated octadecanoid present in several plants including jasmine. Jasmonic acids are a class of plant hormones which are involved in development, abiotic stress responses and plant-microbes interactions in defence and symbiosis. Derivates such as methyl-jasmonate are volatile and participate in long range signalling between plants. Jasmonic acid is synthesized from alpha linolenic acid which is first oxygenated by Lipoxygenase (13-LOX) and subsequently modified by beta oxidation to yield a C12 cyclic product.
References
-
N/A
July 2018
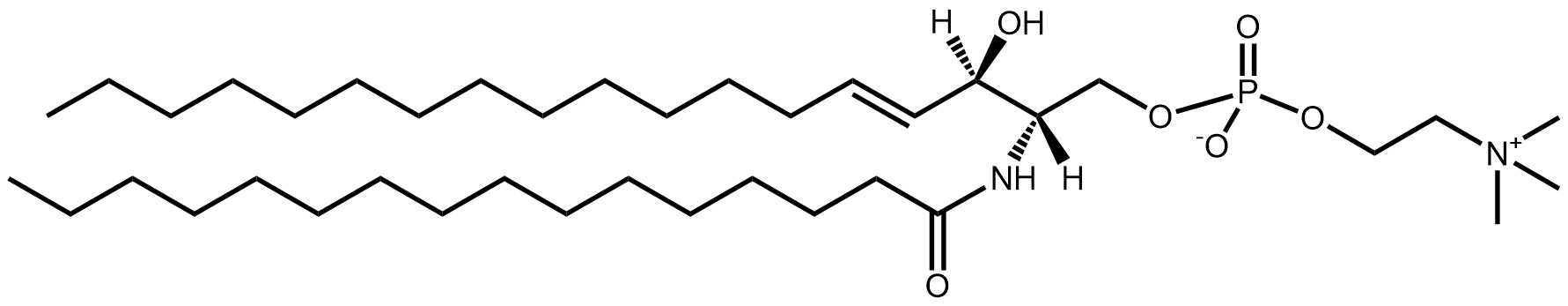
Sphingomyelins, such as SM(d18:1/16:0) (systematic name: N-(hexadecanoyl)-sphing-4-enine-1-phosphocholine) are a class of sphingolipids composed of the long-chain sphingosine base attached to a fatty acid via an N-acyl linkage and a phosphocholine head group. In humans, sphingomyelins represent ~85% of all sphingolipids, and typically make up 10-20 mol % of plasma membrane lipids. They resemble phosphatidylcholines in their general properties and three-dimensional structure, and in having no net charge on their head groups.
References
-
N/A
May 2018
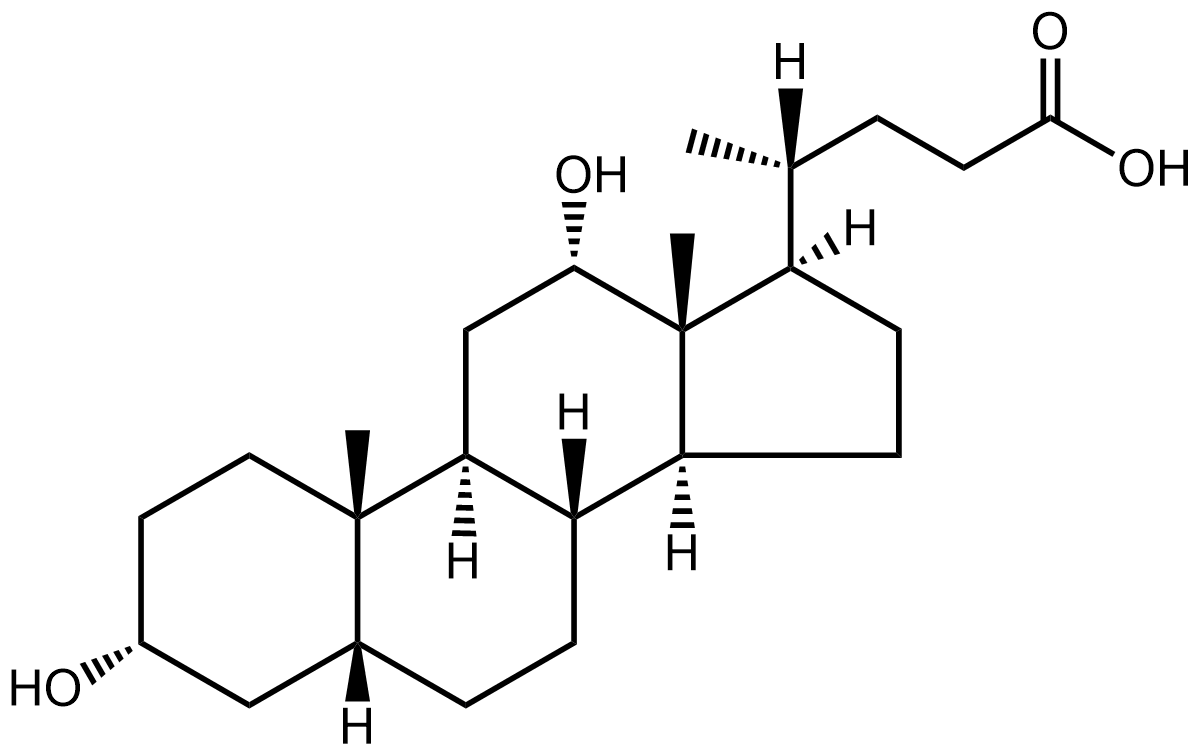 Deoxycholic acid (systematic name:3alpha,12alpha-Dihydroxy-5beta-cholan-24-oic acid), a C24 bile acid, is one of the secondary bile acids, which are metabolic byproducts of intestinal bacteria. The two primary bile acids secreted by the liver are cholic acid and chenodeoxycholic acid.
Bacteria metabolize chenodeoxycholic acid into the secondary bile acid lithocholic acid, and they metabolize cholic acid into deoxycholic acid. Sodium deoxycholate, the sodium salt of deoxycholic acid, is often used as a biological detergent to lyse cells and solubilise cellular and membrane components.
Deoxycholic acid (systematic name:3alpha,12alpha-Dihydroxy-5beta-cholan-24-oic acid), a C24 bile acid, is one of the secondary bile acids, which are metabolic byproducts of intestinal bacteria. The two primary bile acids secreted by the liver are cholic acid and chenodeoxycholic acid.
Bacteria metabolize chenodeoxycholic acid into the secondary bile acid lithocholic acid, and they metabolize cholic acid into deoxycholic acid. Sodium deoxycholate, the sodium salt of deoxycholic acid, is often used as a biological detergent to lyse cells and solubilise cellular and membrane components.
References
-
N/A
April 2018
Anandamide (Systematic Name: N-(5Z,8Z,11Z,14Z-eicosatetraenoyl)-ethanolamine), also known as N-arachidonoylethanolamine or AEA, is a fatty acid neurotransmitter derived from the non-oxidative metabolism of arachidonic acid an essential w-6 polyunsaturated fatty acid. It is synthesized from N-arachidonoyl phosphatidylethanolamine (NAPE) by multiple pathways and is degraded primarily by the fatty acid amide hydrolase (FAAH) enzyme, which converts anandamide into ethanolamine and arachidonic acid.
References
-
N/A
March 2018
Palmitelaidic acid (systematic name: 9E-hexadecenoic acid) is a trans fatty acid (the trans isomer of palmitoleic acid). Trans fatty acids are known to cause changes in plasma lipids and lipoprotein phenotypes, but the mechanisms involved are unknown. The major dietary sources of trans fatty acids are partly hydrogenated vegetable oils, mainly elaidic acid (9E-octadecenoic acid). Additional sources are animal and dairy fats (palmitelaidic acid and trans-vaccenic acid (11E-octadecenoic acid)) and partly hydrogenated fish oils. Palmitelaidic acid has been reported as the predominant trans-16:1 isomer in cheeses made with goat and sheep milk.
References
-
N/A
February 2018

ELV-N34, (Systematic Name:(16Z,19Z,22R,23E,25E,27Z,29S,31Z)-22,29-dihydroxydotetratriaconta-16,19,23,25, 27,31-hexaenoic acid) represents a novel class of lipid mediators biosynthesized in human retinal pigment epithelial (RPE) cells that are oxygenated derivatives of very long-chain polyunsaturated fatty acids (VLC-PUFAs,n-3). These mediators have been named elovanoids (ELV) and are necessary for neuroprotective signaling for photoreceptor cell integrity. Photoreceptor cells express the elongase enzyme ELOVL4 which catalyzes the biosynthesis of elovanoids from 22:6 fatty acids derived from DHA or EPA.
References
-
N/A
January 2018

Coniferonic acid (Systematic name:5Z,9Z,12Z,15Z-octadecatetraenoic acid) is a C18 Delta(5)-unsaturated bis-methylene-interrupted fatty acid commonly found in pine seed oil. It is assumed to be synthesized from alpha-linolenic acid (ALA; 18:3Delta(9,12,15)) by Delta(5)-desaturation. A unicellular green microalga Chlamydomonas reinhardtii also accumulates this fatty acid in a betaine lipid.
References
-
N/A
December 2017
Ursodeoxycholic acid or ursodiol (systematic name: 3alpha,7beta-dihydroxy-5beta-cholan-24-oic acid) is a 24-carbon secondary bile acid, which are metabolic byproducts of intestinal bacteria. Ursodeoxycholic acid helps regulate cholesterol by reducing the rate at which the intestine absorbs cholesterol molecules while breaking up micelles containing cholesterol. Because of this property, ursodeoxycholic acid is used to treat (cholesterol) gallstones non-surgically. Ursodeoxycholic acid has also been shown experimentally to suppress immune response such as immune cell phagocytosis.
References
-
N/A
November 2017
2-oleoyl-glycerol (Systematic Name:2-(9Z-octadecenoyl)-sn-glycerol) is a monoacylglycerol that is found in biological tissues. Monoacylglycerols or monoglycerides are a class of glycerolipids which are composed of a molecule of glycerol linked to a fatty acid via an ester bond. They are typically present at low levels in cell extracts but are intermediates in the degradation of triacylglycerols and diacylglycerols (lipolysis). 2-oleoyl-glycerol was found to be an endogenous ligand to the G protein-coupled receptor GPR119 and has been shown to increase glucagon-like peptide-1 (GLP-1) and gastric inhibitory polypeptide (GIP) levels following administration to the small intestine.
References
-
N/A
October 2017
The eicosanoid Leukotriene C4 (LTC4) is the parent cysteinyl leukotriene produced by the LTC4 synthase (glutathione S-transferase II) catalyzed conjugation of glutathione to LTA4. LTC4 is produced by neutrophils, macrophages, mast cells, and by transcellular metabolism in platelets. It is one of the constituents of slow-reacting substance of anaphylaxis (SRS-A) and exhibits potent smooth muscle contracting activity. LTC4-induced bronchoconstriction and enhanced vascular permeability contribute to the pathogenesis of asthma and acute allergic hypersensitivity.
References
-
N/A
September 2017
Diplotene (systematic name:Hop-22(29)-ene) is a commonly occurring member of the Hopanoid class of Prenol lipids. Hopanoids are natural pentacyclic compounds based on the chemical structure of hopane and have been found to be present in nature in vast amounts as components of bacteria and other primitive organisms. A range of hopanoids are found in petroleum reservoirs, where they are used as biological markers and they have also been found some terrestrial ferns. Hopanoids are one of the most abundant natural products on earth and are present in the organic matter of all sediments, independent of their age, origin or nature and are useful molecular fossil biomarkers in reconstruction of early evolution and geology.
References
-
N/A
August 2017
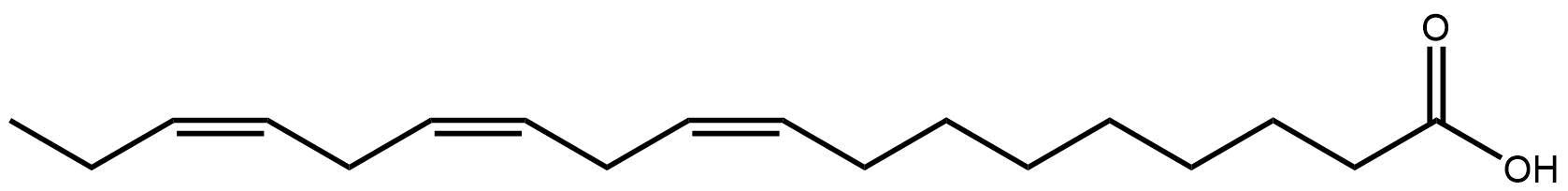
Alpha-linolenic acid or ALA (systematic Name: 9Z,12Z,15Z-octadecatrienoic acid) is an unsaturated fatty acid found primarily in seeds and vegetable oils. ALA is categorized as an omega-3 fatty acid, and is an isomer of gamma-linolenic acid, which is a polyunsaturated omega-6 fatty acid. It is an essential fatty acid because it is necessary for health and cannot be produced within the human body. ALA can only be obtained by humans through their diets because the absence of the required 12- and 15-desaturase enzymes makes de novo synthesis from stearic acid or oleic acid impossible.
References
-
N/A
July 2017
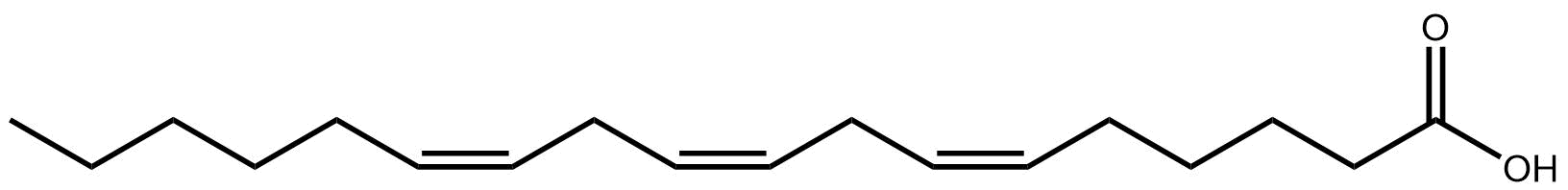
Gamma-linolenic acid or GLA (systematic Name: 6Z,9Z,12Z-octadecatrienoic acid) is an unsaturated fatty acid found primarily in vegetable oils. GLA is categorized as an omega-6 fatty acid, and is an isomer of alpha-linolenic acid, which is a polyunsaturated omega-3 fatty acid, found in rapeseed canola oil, soy beans, walnuts, flax seed (linseed oil) and hemp seed. The human body produces GLA from linoleic acid (LA). This reaction is catalyzed by the enzyme delta6-desaturase (D6D). LA is consumed sufficiently in most diets, from such abundant sources as cooking oils and meats.
References
-
N/A
June 2017
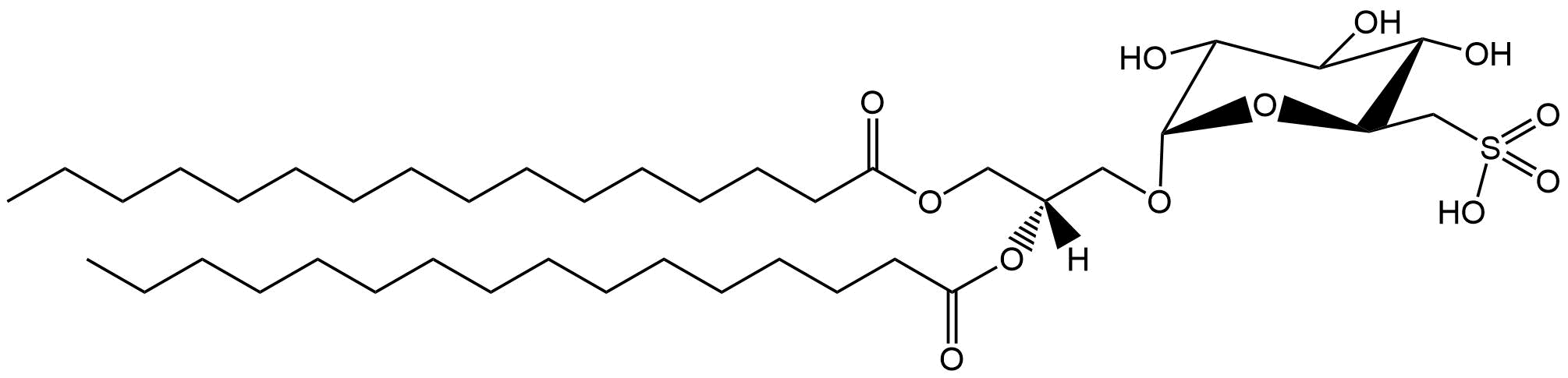
Sulfoquinovosyl diacylglycerols, (SQDG), are a class of sulfur-containing but phosphorus-free lipids (sulfolipids) found in many photosynthetic organisms. SQDG has been found in all photosynthetic plants, algae, cyanobacteria, purple sulfur and non-sulfur bacteria and is localised in the thylakoid membranes, where it appears to be important for membrane structure and function and for optimal activity of photosynthetic protein complexes. It has been estimated to be one of the most abundant organosulfur species in the biosphere and thus plays a major role in the global biogeochemical sulfur cycle. A structure examples is SQDG(16:0/16:0) (1,2-dihexadecanoyl-3-(6'-sulfo-a-D-quinovosyl)-sn-glycerol).
References
-
N/A
April 2017
15R-hydroxy-12S,14R-dioxolane-EET (15R-hydroxy-12S,14R-dioxolane-5Z,8Z,10E-eicosatrienoic acid) is an eicosanoid containing a 1,2-dioxolane (cyclic peroxide) group. Dioxolanes of this type have been generated from enantiomers of 14,15-EET by two mammalian LOX enzymes, 15-LOX-1 and platelet-type 12-LOX. This type of transformation could occur naturally with the co-occurrence of LOX and cytochrome P450 or peroxygenase enzymes.
References
-
N/A
March 2017
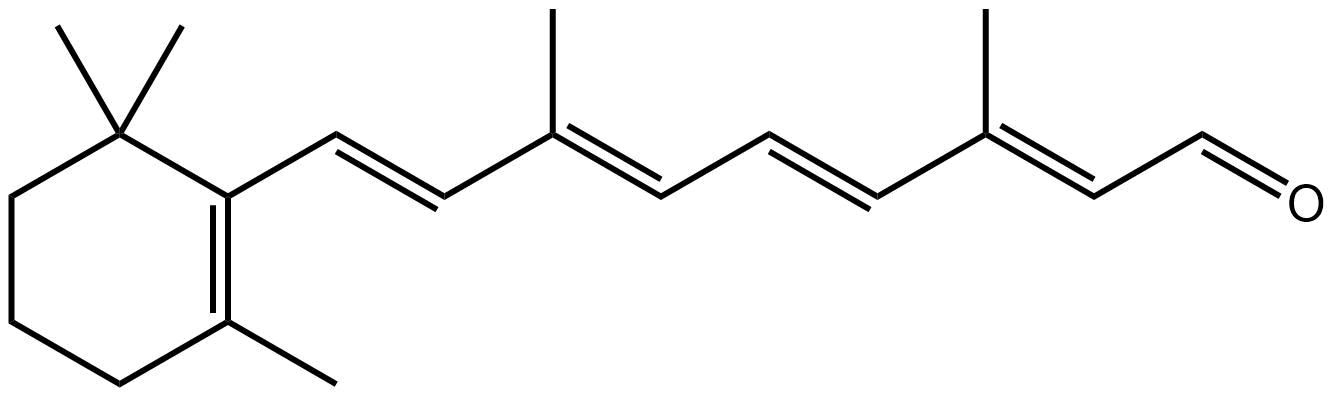
Retinal (all-trans-retinal) is a polyene isoprenoid chromophore, bound to proteins called opsins, and is the chemical basis of animal vision. Retinal exists in two forms, a cis and a trans isomer. On illumination with white light, the visual pigment, cis-retinal is converted to trans-retinal. This isomer must be transformed back into the cis form by retinal isomerase before it combines again with opsin (dark phase). Both isomers can be reduced to retinol (vitamin A) by a NADH-dependent alcohol dehydrogenase.
References
-
N/A
February 2017
 Hyodeoxycholic acid (HDCA) or 3alpha,6alpha-Dihydroxy-5beta-cholan-24-oic acid is a C-24 secondary bile acid, one of the metabolic byproducts of intestinal bacteria. HDCA is present in mammalian species in different proportions and is the main acid constituent of hog bile. Hyodeoxycholic acid undergoes glucuronidation in human liver and kidneys via the enzyme UDP-glucuronosyltransferase.
Hyodeoxycholic acid (HDCA) or 3alpha,6alpha-Dihydroxy-5beta-cholan-24-oic acid is a C-24 secondary bile acid, one of the metabolic byproducts of intestinal bacteria. HDCA is present in mammalian species in different proportions and is the main acid constituent of hog bile. Hyodeoxycholic acid undergoes glucuronidation in human liver and kidneys via the enzyme UDP-glucuronosyltransferase.
References
-
N/A
January 2017
Myristic acid, or tetradecanoic acid, is a common saturated fatty acid named after the nutmeg Myristica fragrans. Myristic acid is also found in palm kernel oil, coconut oil, butter fat and is a minor component of many other animal fats. It is used to synthesize flavor and as an ingredient in soaps and cosmetics. Myristoylation is a co-translational or post-translational modification where a myristoyl group, derived from myristic acid, is covalently attached by an amide bond to the alpha-amino group of an N-terminal glycine residue. Myristoylation plays an essential role in membrane targeting, protein-protein interactions and functions widely in a variety of signal transduction pathways.
References
-
N/A
December 2016
Phosphatidylinositols (for example PI(16:0/18:1)) consist of a class of glycerophospholipids containing a myo-inositol group headgroup. Typically phosphatidylinositols form a minor component on the cytosolic side of eukaryotic cell membranes. Biosynthesis of phosphatidylinositol from CDP-diacylglycerol and myo-inositol is catalyzed by phosphatidylinositol synthase in eukaryotes. Phosphatidylinositols can be phosphorylated by a number of different kinases to form phosphatidylinositol phosphates (PIP), phosphatidylinositol bisphosphates (PIP2) and phosphatidylinositol trisphosphates (PIP3). These molecules play important roles in lipid signaling, cell signaling and membrane trafficking.
References
-
N/A
November 2016
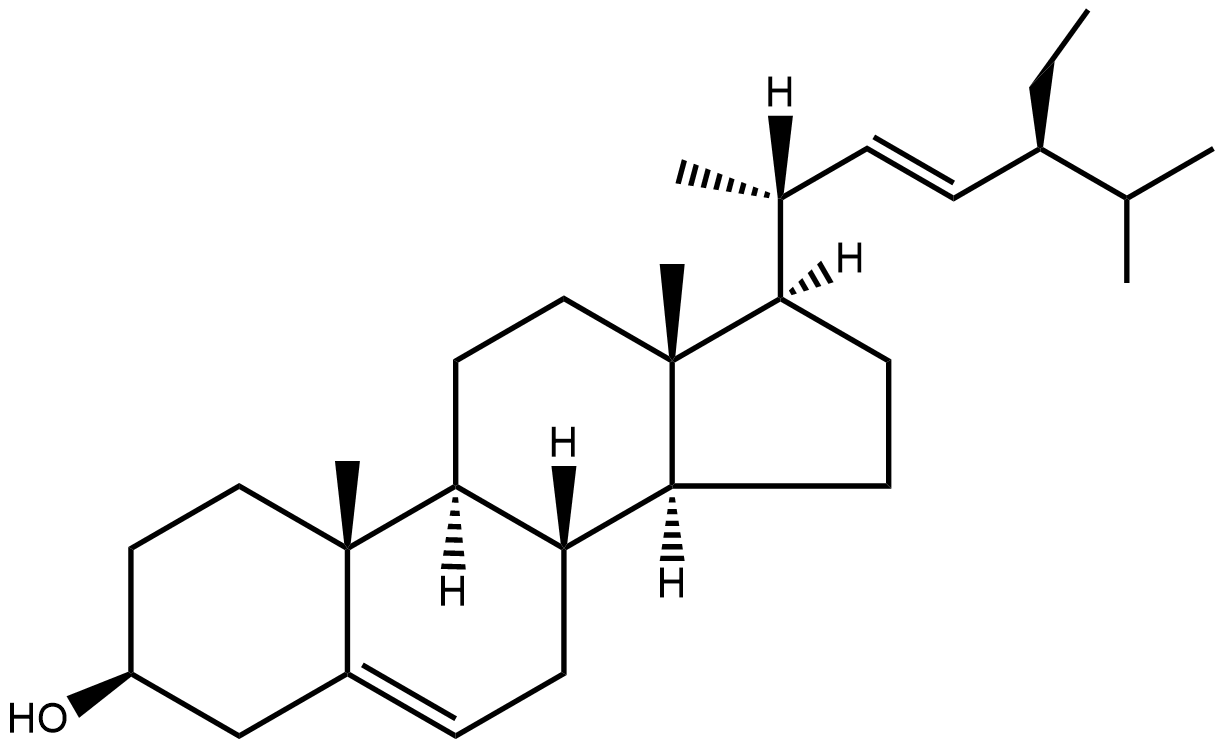
Stigmasterol (stigmasta-5,22E-dien-3beta-ol) is an unsaturated phytosterol (plant sterol) occurring in the plant fats or oils of soybean, rape seed, and in a number of medicinal herbs. It differs structurally from cholesterol due to the presence of a side-chain double bond and ethyl group. Stigmasterol is also found in various vegetables, legumes, nuts, seeds, and unpasteurized milk. Phytosterols normally are broken down in the bile.
References
-
N/A
October 2016
C16-Sphingomyelin or SM(d18:1/16:0) ( systematic name: N-(hexadecanoyl)-sphing-4-enine-1-phosphocholine) is a commonly occurring member of the sphingomyelin class of sphingolipids. Sphingomyelins are present in the plasma membranes of animal cells and are especially prominent in myelin, a membranous sheath that surrounds and insulates the axons of some neurons. Sphingomyelins contain a phosphocholine polar head group attached to a ceramide backbone and resemble phosphatidylcholines in their general properties and three-dimensional structure.
References
-
N/A
September 2016
Leukotriene B4 (5S,12R-dihydroxy-6Z,8E,10E,14Z-eicosatetraenoic acid) is a pro-inflammatory eicosanoid mediator synthesised in myeloid cells from arachidonic acid. Synthesis is catalysed by 5-lipoxygenase and leukotriene A4 hydrolase and is increased by inflammatory mediators including endotoxin, complement fragments, tumor necrosis factor and interleukins. Leukotriene B4 is able to induce the adhesion and activation of leukocytes on the endothelium, allowing them to bind to and cross it into the tissue. In neutrophils, it is also a potent chemoattractant, and is able to induce the formation of reactive oxygen species and the release of lysosomal enzymes by these cells.
References
-
N/A
August 2016
 Chenodeoxycholic acid (3alpha,7alpha-dihydroxy-5ß-cholan-24-oic acid) is a C24 bile acid and one of the main bile acids produced by the liver.
Chenodeoxycholic acid is synthesized in the liver from cholesterol and can be conjugated in the liver with taurine or glycine, forming taurochenodeoxycholic acid or glycochenodeoxycholic acid. Chenodeoxycholic acid is the most potent natural bile acid for stimulating the nuclear bile acid receptor, farnesoid X receptor. The transcription of many genes is activated by FXR.
Chenodeoxycholic acid (3alpha,7alpha-dihydroxy-5ß-cholan-24-oic acid) is a C24 bile acid and one of the main bile acids produced by the liver.
Chenodeoxycholic acid is synthesized in the liver from cholesterol and can be conjugated in the liver with taurine or glycine, forming taurochenodeoxycholic acid or glycochenodeoxycholic acid. Chenodeoxycholic acid is the most potent natural bile acid for stimulating the nuclear bile acid receptor, farnesoid X receptor. The transcription of many genes is activated by FXR.
References
-
N/A
July 2016
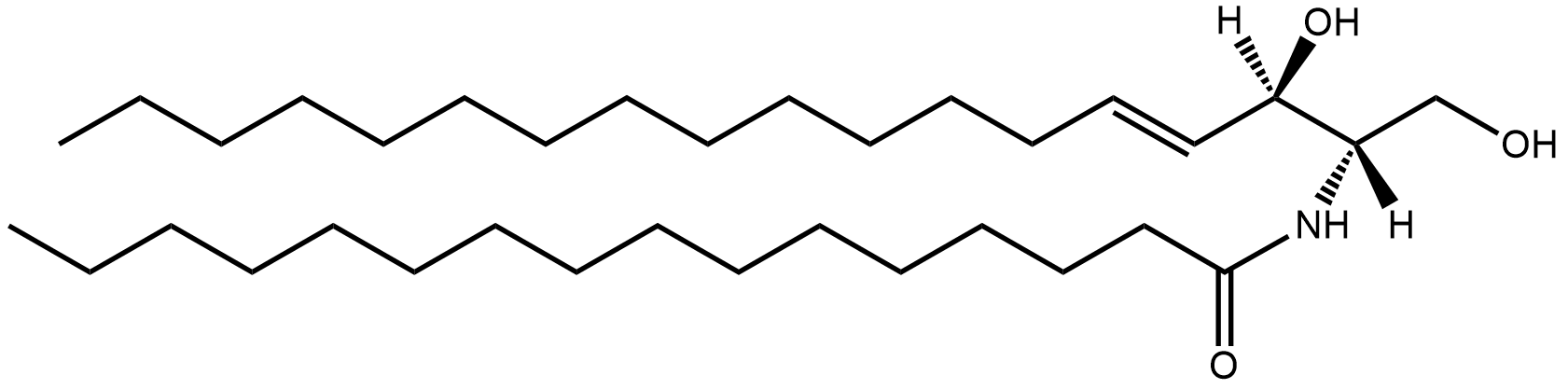
Ceramides, such as Cer(d18:1/16:0) (systematic name: N-(hexadecanoyl)-sphing-4-enine) are a class of sphingolipids composed of the long-chain base sphingosine attached to a fatty acid via an N-acyl linkage. Ceramides are found in high concentrations within the cell membrane of cells. De novo synthesis of ceramide occurs in the endoplasmic reticulum. Ceramide is subsequently transported to the Golgi apparatus by either vesicular trafficking or the ceramide transfer protein CERT. Once in the Golgi apparatus, ceramide can be further metabolized to other sphingolipids, such as sphingomyelin (a key component of cell membranes) and the complex glycosphingolipids.
References
-
N/A
June 2016
Cyanidin (2-(3,4-Dihydroxyphenyl) chromenylium-3,5,7-triol) is a polyketide metabolite belonging to the anthocyanidin subclass of flavonoids. It is a pigment found in many red berries including grapes, bilberry, blackberry and blueberry. Cyanidin, like other anthocyanidins, has putative antioxidant and radical-scavenging effects which may protect cells from oxidative damage and reduce risk of cardiovascular diseases and cancer.
References
-
N/A
May 2016
Oleanolic acid ((3beta-hydroxyolean-12-en-28-oic acid)) is a naturally occurring pentacyclic triterpenoid related to betulinic acid. It is widely distributed in food and plants where it exists as a free acid or as an aglycone of triterpenoid saponins. Oleanolic acid is relatively non-toxic, hepatoprotective, and exhibits antitumor and antiviral properties.
References
-
N/A
April 2016
Palmitic acid (LMFA01010001), or hexadecanoic acid, is the most common saturated fatty acid found in animals, plants and microorganisms. As its name indicates, it is a major component of the oil from palm trees (palm oil), but can also be found in meats, cheeses, butter, and dairy products. Palmitic acid is the first fatty acid produced during fatty acid synthesis and the precursor to longer chain fatty acids. Palmitic acid is used to produce soaps and cosmetics. These applications utilize sodium palmitate, which is commonly obtained by saponification of palm oil.
References
-
N/A
February 2016

Docosahexaenoic acid (DHA) is an omega-3 fatty acid that is a primary structural component of the human brain, cerebral cortex, skin, sperm, testicles and retina. It can be synthesized from alpha-linolenic acid or obtained directly from maternal milk or fish oil. It's systematic name is 4Z,7Z,10Z,13Z,16Z,19Z-docosahexaenoic acid. DHA is the most abundant omega-3 fatty acid in the brain and retina. DHA comprises 40% of the polyunsaturated fatty acids (PUFAs) in the brain and 60% of the PUFAs in the retina. Fifty percent of the weight of a neuron's plasma membrane is composed of DHA.
References
-
N/A
January 2016
Sphingosine-1-phosphate (S1P) is an signaling sphingolipid and is composed of a C18 sphingoid base with a phosphate group at the C1 position. Phosphorylation of sphingosine to S1P is catalyzed by sphingosine kinase, an enzyme ubiquitously found in the cytosol and endoplasmic reticulum of various types of cells. Although S1P is of importance in the entire human body, it is a major regulator of vascular and immune systems. In the vascular system, S1P regulates angiogenesis, vascular stability, and permeability. In the immune system, it is now recognized as a major regulator of trafficking of T- and B-cells.
References
-
N/A
December 2015
Resolvin D2 (7S,16R,17S-trihydroxy-4Z,8E,10Z,12E,14E,19Z-docosahexaenoic acid) is a member of class of eicosanoids and docosanoids known as resolvins. Resolvins are compounds that are made by the human body from the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). They are produced by the COX-2 pathway especially in the presence of aspirin. Experimental evidence indicates that resolvins reduce cellular inflammation by inhibiting the production and transportation of inflammatory cells and chemicals to the sites of inflammation.
References
-
N/A
November 2015
Taurocholic acid ,(N-(3alpha,73alpha,123alpha-trihydroxy-5beta-cholan-24-oyl)-taurine), known also as cholyltaurine, is a bile acid conjugate involved in the emulsification of fats. It occurs as a sodium salt in the bile of mammals. It is a conjugate of cholic acid with taurine.
References
-
N/A
October 2015
12-oxo Phytodienoic acid (OPDA) or 12-oxo-PDA LMFA02010001 is an octadecanoid and a biologically active, immediate precursor of 7-epi jasmonic acid. In addition to its link with jasmonic acid activity, OPDA appears to play an independent role in mediating resistance to pathogens and pests.
References
-
N/A
September 2015
 Geranial, or 3,7-dimethyl-2E,6-octadienal is a monoterpenoid with the molecular formula C10H16O. The two compounds are double bond isomers. The 2E-isomer is also called citral A. The 2Z-isomer is known as neral or citral B.
Geranial is present in the oils of several plants, including limes, lemons and oranges and has a strong lemon odor. It also has strong antimicrobial qualities and pheromonal effects in insects.
Geranial, or 3,7-dimethyl-2E,6-octadienal is a monoterpenoid with the molecular formula C10H16O. The two compounds are double bond isomers. The 2E-isomer is also called citral A. The 2Z-isomer is known as neral or citral B.
Geranial is present in the oils of several plants, including limes, lemons and oranges and has a strong lemon odor. It also has strong antimicrobial qualities and pheromonal effects in insects.
References
-
N/A
August 2015
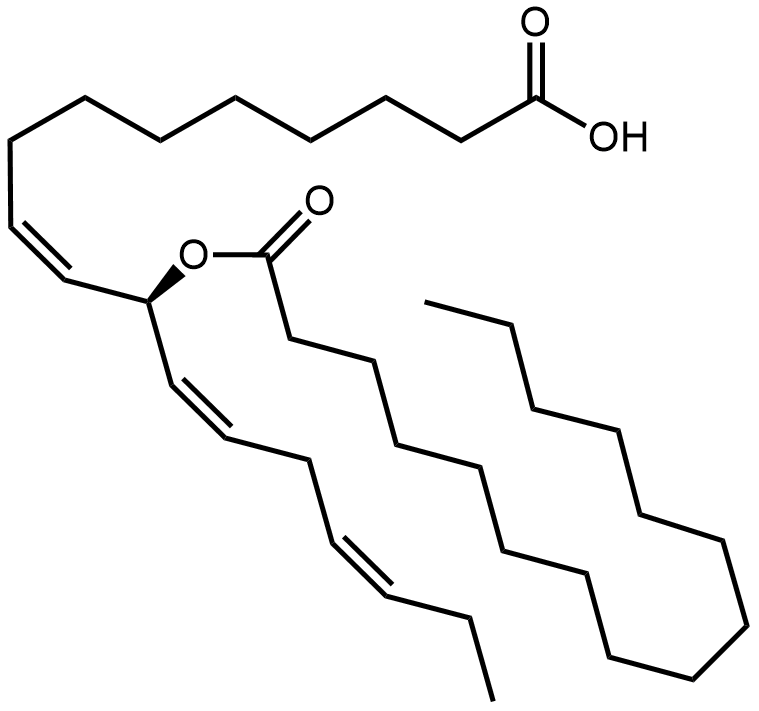
Mayolene-16 or 11R-hexadecanoyloxyoctadeca-9Z,12Z,15Z-trienoic acid is a member of a class of fatty esters (mayolenes) found in the larvae of the European cabbage butterfly, Pieris rapae. They are composed of esters between straight-chain fatty acids and 11-hydroxy alpha-linolenic acid. The mayolenes have been shown to act as potent chemical deterrents to larval predators such as ants.
References
-
N/A
July 2015
Myristoleic acid, or 9Z-tetradecenoic acid, is an omega-5 fatty acid. It is biosynthesized from myristic acid by the enzyme delta-9 desaturase, but it is uncommon in nature. One of the major sources of this fatty acid is the seed oil from plants of the family Myristicaceae, comprising up to 30 per cent of the oil in some species.
References
-
N/A
June 2015
Prostaglandin D2 (or PGD2) is a prostaglandin that binds to the receptor PTGDR, as well as CRTH2. It is a major prostaglandin produced by mast cells and recruits Th2 cells, eosinophils, and basophils. In mammalian organs, large amounts of PGD2 are found only in the brain and in mast cells. It is critical to development of allergic diseases such as asthma. Cellular synthesis occurs through the arachidonic acid cascade with the final conversion from PGH2 performed by PGD2 synthase (PTGDS).
References
-
N/A
May 2015
Lauric acid, or dodecanoic acid, is a saturated fatty acid with a 12-carbon atom chain. Lauric acid, as a component of triglycerides, comprises about half of the fatty acid content in coconut oil, laurel oil, and palm kernel oil. It is also found in human breast milk (6.2% of total fat), cow's milk and goat's milk. Industrially, it is mainly used for the production of soaps and cosmetics.
References
-
N/A
April 2015
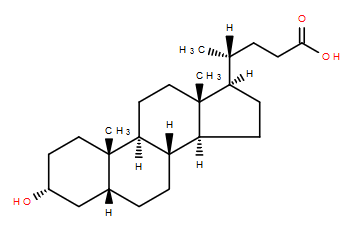 Lithocholic acid, or 3alpha-hydroxy-5beta-cholan-24-oic acid is a monohydroxy secondary bile acid that acts as a detergent to solubilize fats for absorption. Bacterial action in the colon produces lithocholic acid from chenodeoxycholic acid by reduction of the C7 hydroxyl group in the B ring.
Lithocholic acid can activate the vitamin D receptor without raising calcium levels as much as vitamin D itself.
Lithocholic acid, or 3alpha-hydroxy-5beta-cholan-24-oic acid is a monohydroxy secondary bile acid that acts as a detergent to solubilize fats for absorption. Bacterial action in the colon produces lithocholic acid from chenodeoxycholic acid by reduction of the C7 hydroxyl group in the B ring.
Lithocholic acid can activate the vitamin D receptor without raising calcium levels as much as vitamin D itself.
References
-
N/A
March 2015

Aldosterone (LMST02030026) is a steroid hormone of the mineralocorticoid family synthesized from cholesterol in the adrenal gland. It plays a central role in the regulation of blood pressure, mainly by acting on the distal tubules and collecting ducts of the nephron, increasing reabsorption of ions and water. When dysregulated, aldosterone is pathogenic and contributes to the development and progression of cardiovascular and renal disease. Aldosterone is part of the renin-angiotensin system and tends to promote Na+ and water retention, and lower plasma K+ concentration.
References
-
N/A
February 2015
Jasmonic Acid (LMFA02020001), is a C12 fatty acid which is is biosynthesized from linolenic acid by the octadecanoid pathway. It is a plant growth regulator involved in the signaling mechanisms for a variety of conditions including plant defense, wound healing, tuberization, fruit ripening, and senescence Jasmonic acid is also converted to a variety of derivatives including esters such as methyl jasmonate and may also be conjugated to amino acids.
References
-
N/A
January 2015
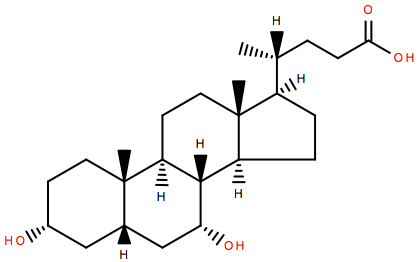
Chenodeoxycholic Acid (LMST04010032), along with cholic acid, is one of two primary bile acids found in humans. Chenodeoxycholic Acid is synthesized in the liver from cholesterol. It is known to aid digestion and can be used to dissolve gallstones and in the treatment of cerebral cholesterosis.
References
-
N/A