Betaine Lipids
 Glycerolipids containing an ether-linked betaine (amino acid) moiety occur naturally
in lower eukaryotic organisms such as algae, bryophytes, fungi, and some primitive protozoa and photosynthetic bacteria.
They are not in general found in flowering plants (Rambutan seed oils from China may be an exception), but they have been detected in
some spore-producing plants, such as ferns and species belonging to the Equisetophyta and related genera.
In these lipids, the polar betaine group is linked by an ether bond at the sn‑3 position of the glycerol moiety, with the
fatty acids esterified in the sn-1 and sn-2 positions.
There is no phosphorus or carbohydrate group, and while some might prefer to classify such lipids with the
complex lipoamino acids, they are treated separately here because of their distinctive occurrence and function.
The term 'betaine' was originally applied to trimethylglycine (illustrated), first isolated from sugar beet,
but it is now used generically for other N‑trimethylated amino acids.
Occasional reports of their occurrence in marine invertebrates is attributed to digestion of algae or symbiotic bacteria.
Glycerolipids containing an ether-linked betaine (amino acid) moiety occur naturally
in lower eukaryotic organisms such as algae, bryophytes, fungi, and some primitive protozoa and photosynthetic bacteria.
They are not in general found in flowering plants (Rambutan seed oils from China may be an exception), but they have been detected in
some spore-producing plants, such as ferns and species belonging to the Equisetophyta and related genera.
In these lipids, the polar betaine group is linked by an ether bond at the sn‑3 position of the glycerol moiety, with the
fatty acids esterified in the sn-1 and sn-2 positions.
There is no phosphorus or carbohydrate group, and while some might prefer to classify such lipids with the
complex lipoamino acids, they are treated separately here because of their distinctive occurrence and function.
The term 'betaine' was originally applied to trimethylglycine (illustrated), first isolated from sugar beet,
but it is now used generically for other N‑trimethylated amino acids.
Occasional reports of their occurrence in marine invertebrates is attributed to digestion of algae or symbiotic bacteria.
1. Structure and Composition
Three related lipids of this type have been described with differing trimethylated hydroxyamino acids linked to diacylglycerols through an ether bond, i.e., 1,2‑diacylglyceryl-3-O-4'-(N,N,N-trimethyl)-homoserine (sometimes abbreviated to DGTS), 1,2‑diacylglyceryl-3-O-2'-(hydroxymethyl)-(N,N,N-trimethyl)-β-alanine (DGTA) and 1,2‑diacylglyceryl-3-O-carboxy-(hydroxymethyl)-choline (DGCC). They each have a positively charged trimethylammonium group and a negatively charged carboxyl group, and they are therefore zwitterionic at neutral pH.
Of these, DGTS is by far the most common in nature and the only one in green algae, and taxonomic and phytogenetic studies suggest that it may have been the first lipid of this type to be formed during evolution. The alanine-derived lipid DHTA can be a major constituent of the lipids of brown algae (sea weeds), e.g., Ochromonas danica and Fucus vesiculosus, sometimes together with DGCC, as in marine algae of the genus Haptophyceae, e.g., Pavlova lutheri (or other species from the Chromalveolata supergroup). DGCC is only rarely detected in other than trace amounts except when phosphate is limiting as in the model diatom Thalassiosira pseudonana, but it does occur normally in greater amounts in symbiotic dinoflagellates of the family Symbiodiniaceae and their coral hosts where it is the only betaine lipid (though sometimes with its lyso form). The fungus Heterospora chenopodii contains a monoacylglyceryltrimethylhomoserine in which the acyl moiety is a novel 3‑keto fatty acid, while a comparable lipid from Penicillium glaucoroseum contains an unusual branched-chain fatty acid.
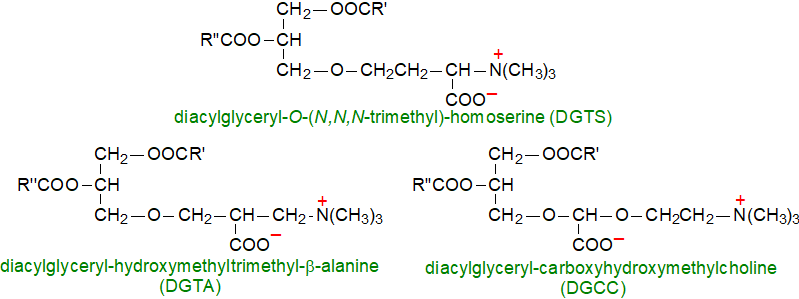 |
| Figure 1. Structures of the three main betaine lipids. |
In the diacylglyceryltrimethylhomoserines of most algae studied, the fatty acids in position sn-1 of the glycerol moiety tend to be saturated (mainly 14:0 and 16:0), while those in position 2 are C18 unsaturated (predominantly 18:2(n-6) and 18:3(n-3)). DGCC in the Symbiodiniaceae contains high proportions of long-chain and very-long chain polyunsaturated fatty acids (28:7 and 28:8), the latter not present in phosphatidylcholine from the same organism. The fatty acid compositions and positional distributions within the glycerol moiety can be somewhat different from those in other glycerolipids such as phosphatidylcholine, as can be seen from the results in Table 1 for a Chlorella sp. Of the fatty acids in DGTS of Acanthamoeba castellanii, 87% is oleate (9‑18:1). In O. danica (algae), DGTS contains mainly C14 and C18 fatty acids, while DGTA has C20 and C22 fatty acids. Some marine algae, including the microalga Nannochloropsis oceanica, have appreciable amounts of eicosapentaenoic acid (EPA or 20:5(n-3)) in both positions of their DGTS, although it is a minor component only in the phospholipids.
Table 1. Stereospecific distribution of acyl moieties of phosphatidylcholine (PC) and DGTS between positions sn-1 and 2 of the glycerol backbone in a Chlorella sp. |
||||||||||
| Lipid class |
Position | Fatty acid composition (Mol %) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 16:0 | 16:1 | 16:2 | 16:3 | 18:0 | 18:1 | 18:2 | 18:3 | other | ||
| PC | sn-1 | 14 | 2 | 2 | 1 | 2 | 60 | 8 | 9 | 2 |
| sn-2 | 2 | 5 | trace | 4 | trace | 52 | 20 | 15 | 2 | |
| DGTS | sn-1 | 46 | 5 | trace | 1 | 1 | 36 | 5 | 3 | 3 |
| sn-2 | 14 | 3 | 7 | 6 | 3 | 34 | 17 | 14 | 2 | |
| Adapted from Weber, N. et al. J. Lipid Mediators, 1, 37-48 (1989); PMID: 2519885. | ||||||||||
In the extra-chloroplastid space of Chlamydomonas reinhardtii (microalgae) growing in a nutrient replete culture, position sn-2 of the DGTS contains mainly C18 polyunsaturated fatty acids, whereas the glycerolipids in the chloroplasts have mainly C16 polyunsaturated fatty acids in this position. It is noteworthy that this difference in specificity is characteristic of the distinction between the ‘eukaryotic’ and ‘prokaryotic’ types (now known to be a misnomer) of glycerolipid synthesis seen in the glycerolipids of higher plants (see our web page on galactosyldiacylglycerols for further details).
 There is an
obvious similarity between the structures of betaine lipids and that of the zwitterionic glycerophospholipid
phosphatidylcholine.
Although the phase transition temperature for DGTS is slightly higher than that of phosphatidylcholine with an identical fatty acid composition,
the physical phase properties of both lipids in mixtures with water are in general alike.
There is evidence for an inverse relationship between the presence of betaine lipids and phosphatidylcholine in the
membranes of some algae, phytoplankton, fungi and bacteria (but not in all) to indicate that they can substitute for each other to a
substantial extent in relation to membrane biophysics at least, although the molecular species compositions can be very different.
DGCC and its deacylated derivative, obtained from digestion of symbiotic algae, are present in all tissues and organs, including algae-free
sperm and eggs, of the giant clam Tridacna crocea, where it may have the same role in membranes as phosphatidylcholine.
Indeed, the yeast Saccharomyces cerevisiae, in which the enzymes for DGTS biosynthesis are normally absent, has been genetically
engineered so that phosphatidylcholine is completely replaced by DGTS with all the essential functions continuing to operate.
There is an
obvious similarity between the structures of betaine lipids and that of the zwitterionic glycerophospholipid
phosphatidylcholine.
Although the phase transition temperature for DGTS is slightly higher than that of phosphatidylcholine with an identical fatty acid composition,
the physical phase properties of both lipids in mixtures with water are in general alike.
There is evidence for an inverse relationship between the presence of betaine lipids and phosphatidylcholine in the
membranes of some algae, phytoplankton, fungi and bacteria (but not in all) to indicate that they can substitute for each other to a
substantial extent in relation to membrane biophysics at least, although the molecular species compositions can be very different.
DGCC and its deacylated derivative, obtained from digestion of symbiotic algae, are present in all tissues and organs, including algae-free
sperm and eggs, of the giant clam Tridacna crocea, where it may have the same role in membranes as phosphatidylcholine.
Indeed, the yeast Saccharomyces cerevisiae, in which the enzymes for DGTS biosynthesis are normally absent, has been genetically
engineered so that phosphatidylcholine is completely replaced by DGTS with all the essential functions continuing to operate.
When phosphorus is a limiting nutrient, synthesis of DGTS to replace phospholipids is a common strategy in many algae and fungi. For example, the opportunistic fungal pathogen Cryptococcus neoformans can cause life-threatening meningitis, and it must acquire sufficient phosphate from its host to sustain its growth and tolerate host-derived stress, but it can produce DGTS as a defence mechanism; in so doing, it retains its virulence. In the plant pathogen Agrobacterium tumefaciens, the increased formation of DGTS in this circumstance is accompanied by increases in the concentration of cyclopropyl fatty acid constituents in all lipids. N. oceanica requires DGTS not only for normal cell proliferation under phosphate-starved conditions, but also for adaptation to low temperatures. Some data for the bacterium Mesorhizobium loti (Rhizobiales) grown in phosphorus-replete and phosphorus-depleted conditions are listed in Table 2.
Table 2. Polar lipid composition (%) of Mesorhizobium loti grown under phosphate-replete (+ Pi) and phosphate-depleted (- Pi) conditions |
||
| Lipid class | + Pi | - Pi |
|---|---|---|
| Glycosyldiacylglycerols | - | 10 |
| Diacylglycerol-trimethylhomoserine | - | 24 |
| Ornithine lipid | 11 | 16 |
| Cardiolipin (+ unknown) | 5 | 11 |
| Phosphatidylcholine | 30 | 15 |
| Phosphatidylglycerol | 17 | 14 |
| Mono- and dimethyl-phosphatidylethanolamine | 38 | 10 |
| Adapted from Devers, E.A. et al. J. Bact., 193, 1377-1384 (2011); DOI. | ||
2. Biosynthesis and Metabolism
All betaine lipids are of extra-plastidial origin, i.e., they are synthesised in the endoplasmic reticulum and not in the chloroplasts. The biosynthesis of diacylglyceryl-N,N,N-trimethylhomoserine was first studied in phosphate-starved cells of the purple bacterium Rhodobacter sphaeroides. Two enzyme systems were identified as essential to the process, with the first (BtaA) transferring the 3‑amino-3-carboxypropyl group of S‑adenosylmethionine to the 3-hydroxyl of a 1,2-diacyl-sn-glycerol to form the intermediate diacylglycerylhomoserine. The second enzyme system (BtaB) transfers methyl groups from S-adenosylmethionine in three successive steps to form the final product diacylglyceryl-N,N,N-trimethylhomoserine. This mechanism is common in prokaryotes. However, in the algal model Chlamydomonas reinhardtii, which produces DGTS in the endoplasmic reticulum to the exclusion of phosphatidylcholine regardless of phosphorus availability, a single bifunctional enzyme, betaine lipid synthase 1 (BTA1), which contains both BtaA- and BtaB-like domains, can carry out the complete synthesis. This mechanism predominates in eukaryotes, although the enzymes in other algae may differ somewhat in structure and domain location.
 |
| Figure 2. Biosynthesis of betaine lipids in R. sphaeroides. |
In O. danica and some other algae, it has been established that the glyceryltrimethylhomoserine part of DGTS is the precursor of the polar group of DGTA by de-carboxylation and re-carboxylation reactions, but with simultaneous deacylation and reacylation of the glycerol moiety (remodelling) as the two have very different fatty acid distributions. The same mechanism operates in fungi. Very little appears to be known of the biosynthesis of 1,2‑diacylglyceryl-3-O-carboxy-(hydroxymethyl)-choline (DGCC).
During phosphate deprivation in the green alga Chlorella kessleri, phosphatidylcholine and phosphatidylethanolamine are almost completely degraded to release 18.1% cellular phosphorus, and the diacylglycerol moieties are utilized in part for DGTS synthesis in the endoplasmic reticulum for incorporation into extrachloroplast membranes. DGTS is not normally present in this organism, and induction of the expression of the BTA1 gene is required during phosphorus deficiency for synthesis to occur.
The ether bond linking the head group to the diacylglycerol moiety in betaine lipids is much stronger than the phosphoryl ester bond in phosphatidylcholine and is impervious to the phospholipases C and D, so it is unclear whether betaine lipids have any metabolic role in addition to their function in membranes, for example as a source of diacylglycerols. No enzyme that cleaves the ether bond has yet been identified, although presumably one must exist. On the other hand, there is evidence from experiments with algae that the betaine lipids are involved in the transfer of fatty acids from the extra-plastidial membranes to the chloroplast, and that they may be the primary acceptor of fatty acids formed de novo before they are processed and redistributed to other lipids including triacylglycerols. During nitrogen starvation, the acyl groups of betaine lipids may be utilized for triacylglycerol synthesis in the algae Phaeodactylum tricornutum, but 1,2-diacyl-sn-glycerols are not produced from them for this purpose in algae. In C. reinhardtii, oleic acid esterified to DGTS can be desaturated to linoleic and linolenic acids outwith the chloroplast (cf., the web page on linolenate biosynthesis in phosphatidylcholine of higher plants), while in N. oceanica, EPA may be esterified first to phospholipids and then rapidly transferred to DGTS in the endoplasmic reticulum and to monogalactosyldiacylglycerols in the plastids.
3. Analysis
Like the choline-containing lipids, betaine lipids display a blue coloration when sprayed with Dragendorff reagent, but they are not stained by the typical reagents used to detect lipid-bound phosphorus. They are usually identified by this means on examination by thin-layer chromatography. Modern mass spectrometric methods greatly facilitate analysis.
4. Related Lipids linked to Amino Acids other than Betaine
A similar type of lipid but in which the amino acid lysine is linked to 1,2-diacyl-sn-glycerol via an ester rather than an ether bond, i.e., lysyl-diacylglycerol, was first isolated from Mycobacterium phlei strain IST, with palmitic and tuberculostearic acids as the fatty acid constituents, and it is now known to be widely distributed among actinobacterial pathogens, including Mycobacterium and Corynebacterium sp., where it contributes to antibiotic resistance and virulence (one of the many unusual lipids from such genera). An enzyme designated 'multiple peptide resistance factor' (a LysX homologue) from Corynebacterium pseudotuberculosis and other mycobacteria is responsible for its synthesis, although the mechanism may be more complex in M. tuberculosis.

Various Corynebacteria possess an alanyl-diacylglycerol synthase (AlaDAGS), an enzyme related to those involved in the synthesis of complex lipoamino acids, that uses Ala-tRNA and 1,2-diacyl-sn-glycerols as substrates. Anaerobic sulfate-reducing Desulfatibacillum alkenivorans produces butyramide-cysteine-dialkyl- or acylalkylglycerols when grown under conditions of phosphorus limitation. 1,2-Diacyl-sn-glycero-3-dehydrophenylalanine (ester-linked also) has been isolated from larvae of the bruchid beetle Bruchidius dorsalis. Such lipids could also be classified as a subset of triacylglycerols or of complex lipoamino acids.
Suggested Reading
- Cañavate, J.P., Armada, I., Ríos, J.L. and Hachero-Cruzado, I. Exploring occurrence and molecular diversity of betaine lipids across taxonomy of marine microalgae. Phytochemistry, 124, 68-78 (2016); DOI.
- Geiger, O., González-Silva, N. López-Lara, I.M. and Sohlenkamp, C. Amino acid-containing membrane lipids in bacteria. Prog. Lipid Res., 49, 46-60 (2010); DOI.
- Gill, C.P., Phan, C., Platt, V., Worrell, D., Andl, T. and Roy, H. The MprF homolog LysX synthesizes lysyl-diacylglycerol contributing to antibiotic resistance and virulence. Microbiol. Spectrum, 11, e01429-23 (2023); DOI.
- Hoffmann, D.Y. and Shachar-Hill, Y. Do betaine lipids replace phosphatidylcholine as fatty acid editing hubs in microalgae? Front. Plant Sci., 14, 1077347 (2023); DOI.
- Kunzler, K. and Eichenberger, W. Betaine lipids and zwitterionic phospholipids in plants and fungi. Phytochemistry, 46, 883-892 (1997); DOI.
- Li-Beisson, Y., Beisson, F. and Riekhof, W. Metabolism of acyl-lipids in Chlamydomonas reinhardtii. Plant J., 82, 504-522 (2015); DOI.
- Oishi, Y., Otaki, R., Iijima, Y., Kumagai, E., Aoki, M., Tsuzuki, M., Fujiwara, S. and Sato, N. Diacylglyceryl-N,N,N-trimethylhomoserine-dependent lipid remodeling in a green alga, Chlorella kessleri. Commun. Biol., 5, 19 (2022); DOI.
- Riekhof, W.R., Andre, C. and Benning, C. Two enzymes, BtaA and BtaB, are sufficient for betaine lipid biosynthesis in bacteria. Arch. Biochem. Biophys., 441, 96-105 (2005); DOI.
- Riekhof, W.R., Naik, S., Bertrand, H., Benning, C. and Voelker, D.R. Phosphate starvation in fungi induces the replacement of phosphatidylcholine with the phosphorus-free betaine lipid diacylglyceryl-N,N,N-trimethylhomoserine. Eukaryotic Cell, 13, 749-757 (2014); DOI.
- Riekhof, W.R., Sears, B.B. and Benning, C. Annotation of genes involved in glycerolipid biosynthesis in Chlamydomonas reinhardtii: discovery of the betaine lipid synthase BTA1Cr. Eukaryotic Cell, 4, 242-252 (2005); DOI.
- Rozentsvet, O.A., Dembitsky, V.M. and Saksonov, S.V. Occurrence of diacylglyceryltrimethylhomoserines and major phospholipids in some plants. Phytochemistry, 54, 401-407 (2000); DOI.
- Salomon, S., Oliva, O., Amato, A., Bastien, O., Michaud, M. and Jouhet, J. Betaine lipids: biosynthesis, functional diversity and evolutionary perspectives. Prog. Lipid Res., 97, 101320 (2025); DOI.
- Sikorskaya, T.V., Ermolenko, EV. and Long, P.Q. Betaine lipids of Symbiodiniaceae hosted by Indo-Pacific corals. Phycol. Res., 71, 193-199 (2023); DOI.
 |
© Author: William W. Christie |  |
|
| Contact/credits/disclaimer | Updated: August 2025 | ||
© The LipidWeb is open access and fair use is encouraged but not text and data mining, AI training, and similar technologies.
